Introduction
Child obesity is a public-health issue of increasing global concern. In 2013, the age-standardised prevalence of overweight and obesity among children and adolescents aged <20 years was estimated to be 23.8% and 22.6% in high-income countries, and 12.9% and 13.4% in low- and middle-income countries, among boys and girls, respectively [Reference de Onis, Blössner and Borghi1–3]. The same year, over 42 million (7%) children under 5 years of age were estimated to be overweight or obese, with prevalence expected to increase to 11% by 2025 if current trends remain constant [Reference de Onis, Blössner and Borghi1–3]. Obesity during childhood is a risk factor for short- and long-term adverse outcomes, including type-2 diabetes, hypertension and dyslipidaemia [Reference Steinberger and Daniels4]. Given the anticipated burden on individuals, health systems and economies, effective evidence-based strategies to prevent and manage child obesity are essential.
Obesity risk is understood to result from a complex interplay of environmental and genetic factors, including well-studied contributions from diet, physical activity and maternal factors such as smoking and breastfeeding [Reference Mozaffarian, Hao, Rimm, Willett and Hu5–Reference Monasta, Batty, Cattaneo, Lutje, Ronfani, Van Lenthe and Brug7]. Additionally, increasing evidence has implicated alterations in the intestinal microbiome in obesity development [Reference Bäckhed, Ding, Wang, Hooper, Koh, Nagy, Semenkovich and Gordon8, Reference Turnbaugh, Ley, Mahowald, Magrini, Mardis and Gordon9]. In particular, antibiotic use in early life has been hypothesised to promote obesity through metabolic dysregulation caused by changes in the intestinal microbial composition [Reference Zeissig and Blumberg10, Reference Cox and Blaser11]. This dysregulation is thought to encompass multiple potential pathways, including increases in microbiota-derived energy, decreased energy requirements due to lowered intestinal defence, changes in hepatic function and altered metabolic signalling [Reference Cox and Blaser11].
Global antibiotic consumption was 34.8 billion daily defined doses (DDDs) in 2015, a 65% increase compared with 2000 levels [Reference Klein, Van Boeckel, Martinez, Pant, Gandra, Levin, Goossens and Laxminarayan12, Reference Van Boeckel, Gandra, Ashok, Caudron, Grenfell, Levin and Laxminarayan13]. Evidence among children suggests notable but varied prevalence across populations and population subgroups [Reference Gharbi, Doerholt, Vergnano, Bielicki, Paulus, Menson, Riordan, Lyall, Patel, Bernatoniene, Versporten, Heginbothom, Goossens and Sharland14–Reference Versporten, Sharland, Bielicki, Drapier, Vankerckhoven and Goossens20]. For example, antibiotic use among children up to 2 years of age was estimated to be 1365 prescriptions per 1000 persons in the USA in 2010 [Reference Hicks, Taylor and Hunkler16], and 462 prescriptions per 1000 persons in Sweden in 2012 [Reference Ternhag and Hellman19]. In both cases, these were among the highest ratios across all age groups [Reference Hicks, Taylor and Hunkler16, Reference Ternhag and Hellman19]. This widespread use of antibiotics among children, and their hypothesised metabolic effects [Reference Cox and Blaser11], suggests the need to more clearly determine the potential link between antibiotic use and obesity in this age group.
A number of studies have assessed associations between antibiotic exposure and measures of body mass in children, with inconsistent results regarding the existence and strength of associations [Reference Scott, Horton, Mamtani, Haynes, Goldberg, Lee and Lewis21–Reference Saari, Virta, Sankilampi, Dunkel and Saxen33]. A more detailed understanding of these studies is required to better identify and define future research aims and inform public-health intervention. In this narrative review, we aim to synthesise and summarise the current evidence on the relation between early life antibiotic use and measures of child body mass.
Methods
We searched PubMed, Web of Science and the Cochrane Library in order to identify studies published before October 2017 assessing the relation between early childhood antibiotic use and measures of child body mass. Key words for antibiotics and body mass-related outcomes were searched and combined using Boolean operators as appropriate (online Supplementary Methods).
Definitions of exposure and outcome
Early childhood antibiotic use was defined as exposure to any pharmaceutical agent with antibacterial action between birth and 3 years of age. This time period was selected as the microbiome is thought to be dynamic and in development until it matures at this age [Reference Yatsunenko, Rey, Manary, Trehan, Dominguez-Bello, Contreras, Magris, Hidalgo, Baldassano, Anokhin, Heath, Warner, Reeder, Kuczynski, Caporaso, Lozupone, Lauber, Clemente, Knights, Knight and Gordon34], with antibiotic administration during this period hypothesised to have a pronounced impact on the development of the microbiome composition and related metabolic functioning. No restrictions were placed on the nature or frequency of the antibiotic exposure, or on the method of ascertainment of antibiotic exposure.
Given the wide variation in definition of overweight and obesity among children in research and clinical practice [Reference de Onis, Blössner and Borghi1, Reference de Onis and Lobstein35–Reference de Onis, Onyango, Borghi, Siyam, Nishida and Siekmann37], measures of body mass included any outcome based on absolute or relative weight or body mass index (BMI) between 2 and 18 years of age. This included BMI-for-age, weight-for-height or binary measures such as overweight or obesity based upon any of these indices. Studies examining self-reported or parental-reported outcomes were included. The upper age limit of the outcome was chosen given the interest in examining long-term implications of early life antibiotic use across childhood.
Inclusion and exclusion criteria
We placed no restrictions on study design, publication date or status in our selection criteria. Only English language publications were reviewed. Studies based on children who were extremely malnourished or with an underlying disease, or in children born low birth weight or small for gestational age, were excluded, given potential differences in their microbiome profile relative to generally healthy, normal weight children [Reference Blanton, Charbonneau, Salih, Barratt, Venkatesh, Ilkaveya, Subramanian, Manary, Trehan, Jorgensen, Fan, Henrissat, Leyn, Rodionov, Osterman, Maleta, Newgard, Ashorn, Dewey and Gordon38, Reference Subramanian, Huq, Yatsunenko, Haque, Mahfuz, Alam, Benezra, DeStefano, Meier and Muegge39].
Literature search and synthesis
An initial search was performed by one author (EP) to identify studies published before March 2016. This was repeated in October 2017 by a second author (UP) to confirm the initial search results and identify more recently published studies (Fig. 1). Titles and abstracts of retrieved records were first screened for inclusion in a full text review. The full texts of potentially relevant studies were then examined to confirm inclusion based on eligibility criteria. Additionally, the references of included studies were also examined to assess the sensitivity of the search strategy and identify any studies that may have been missed. No further studies were identified using this method. Studies published before March 2016 that were chosen for inclusion during the second literature search (conducted by UP) were consistent with those identified during the first literature search (conducted by EP). Results of this second search are presented in the online Supplementary Methods, and in Fig. 1.

Fig. 1. Schematic of literature search and exclusion for final review.
Data on study design, sampling framework, inclusion and exclusion criteria, sample characteristics, exposure and outcome definition and ascertainment, covariates, statistical methodology and main results were extracted from included studies. Results of studies were grouped and summarised in terms of overall associations between antibiotic exposure and measures of child body mass, and associations by sex, window of antibiotic exposure, repeated antibiotic exposure, antibiotic class and maternal BMI. Qualitative review and evidence synthesis included an examination of the presence of associations, and their magnitude and statistical strength. The consistency in trends of associations across strata was also considered when examining windows of exposure and repeated exposure to antibiotics. We assessed whether studies adjusted for a number of confounders that may plausibly influence associations between antibiotic use and obesity among children (Box 1; online Supplementary Table S1). Other extracted data on key study characteristics were also assessed in order to understand individual studies’ strengths and limitations. Given the wide variation in study characteristics, we did not perform a meta-analysis of study results.
Box 1. Potential confounders of the association between early life antibiotic use and child obesity
Early childhood infections
Delivery mode
Exclusive breastfeeding
Preterm birth and birth weight
Maternal smoking
Socio-economic status
Maternal BMI
Current wheeze/asthma
Lifestyle (diet and physical activity)
Siblingship
See Supplementary Table S1 for hypothesised pathways.
Results
Study characteristics
We identified 13 observational studies, based on a total of 6 81 332 individuals, which permitted assessment of antibiotic exposure before 3 years of age and subsequent measures of body mass or risk of overweight or obesity in childhood (Fig. 1, Table 1) [Reference Scott, Horton, Mamtani, Haynes, Goldberg, Lee and Lewis21–Reference Saari, Virta, Sankilampi, Dunkel and Saxen33]. None of the examined studies included assessment of participants’ intestinal microbiota. Study designs included both prospective [Reference Ajslev, Andersen, Gamborg, Sorensen and Jess22, Reference Trasande, Blustein, Liu, Corwin, Cox and Blaser26–Reference Rogawski, Platts-Mills, Seidman, John, Mahfuz, Ulak, Shrestha, Soofi, Yori, Mduma, Svensen, Ahmed, Lima, Bhutta, Kosek, Lang, Gottlieb, Zaidi, Kang, Bessong, Houpt and Guerrant28, Reference Mbakwa, Scheres, Penders, Mommers, Thijs and Arts31, Reference Saari, Virta, Sankilampi, Dunkel and Saxen33] and retrospective [Reference Scott, Horton, Mamtani, Haynes, Goldberg, Lee and Lewis21, Reference Schwartz, Pollak, Bailey-Davis, Hirsch, Cosgrove, Nau, Kress, Glass and Bandeen-Roche25, Reference Gerber, Bryan, Ross, Daymont, Parks, Localio, Grundmeier, Stallings and Zaoutis29, Reference Bailey, Forrest, Zhang, Richards, Livshits and DeRusso30, Reference Li, Chen, Ferber and Odouli32] cohort studies, and cross-sectional [Reference Murphy, Stewart, Braithwaite, Beasley, Hancox and Mitchell24] and case–control studies [Reference Azad, Bridgman, Becker and Kozyrskyj23]. Apart from one study based in India [Reference Rogawski, Westreich, Adair, Becker-Dreps, Sandler, Sarkar, Kattula, Ward, Meshnick and Kang27] and two multi-site studies [Reference Murphy, Stewart, Braithwaite, Beasley, Hancox and Mitchell24, Reference Rogawski, Platts-Mills, Seidman, John, Mahfuz, Ulak, Shrestha, Soofi, Yori, Mduma, Svensen, Ahmed, Lima, Bhutta, Kosek, Lang, Gottlieb, Zaidi, Kang, Bessong, Houpt and Guerrant28], all research was based on North American [Reference Azad, Bridgman, Becker and Kozyrskyj23, Reference Schwartz, Pollak, Bailey-Davis, Hirsch, Cosgrove, Nau, Kress, Glass and Bandeen-Roche25, Reference Gerber, Bryan, Ross, Daymont, Parks, Localio, Grundmeier, Stallings and Zaoutis29, Reference Bailey, Forrest, Zhang, Richards, Livshits and DeRusso30, Reference Li, Chen, Ferber and Odouli32] and European [Reference Scott, Horton, Mamtani, Haynes, Goldberg, Lee and Lewis21, Reference Ajslev, Andersen, Gamborg, Sorensen and Jess22, Reference Trasande, Blustein, Liu, Corwin, Cox and Blaser26, Reference Mbakwa, Scheres, Penders, Mommers, Thijs and Arts31, Reference Saari, Virta, Sankilampi, Dunkel and Saxen33] populations (Table 1; online Supplementary Tables S2 and S3).
Table 1. Summary of studies included in final review

zBMI, body mass index-for-age z-score; WAZ, weight-for-age z-score; aOR, adjusted odds ratio.
*p < 0.05.
See Supplementary Tables S2 and S3 for full details on characteristics of studies.
Blank cells for anthropometric reference column indicate that none was used.
a Authors reported that questionnaires were only able to capture antibiotics prescribed for ear and lung infection.
b British 1990 growth reference used as part of the UK WHO Term Growth Reference.
Studies varied considerably with respect to a range of methodological aspects, including exposure and outcome definition and ascertainment, analytical strategy and inclusion of covariates (Table 1, online Supplementary Tables S2 and S3). While all studies examined any antibiotic exposure in early life, one study was able to capture only antibiotics prescribed for ear and lung infections [Reference Ajslev, Andersen, Gamborg, Sorensen and Jess22]. Overall exposure windows of studies ranged from up to 6 months of age [Reference Ajslev, Andersen, Gamborg, Sorensen and Jess22, Reference Rogawski, Westreich, Adair, Becker-Dreps, Sandler, Sarkar, Kattula, Ward, Meshnick and Kang27–Reference Gerber, Bryan, Ross, Daymont, Parks, Localio, Grundmeier, Stallings and Zaoutis29] to up to ⩾3 years of age [Reference Schwartz, Pollak, Bailey-Davis, Hirsch, Cosgrove, Nau, Kress, Glass and Bandeen-Roche25, Reference Mbakwa, Scheres, Penders, Mommers, Thijs and Arts31]. Methods of antibiotic exposure ascertainment included parental recall [Reference Ajslev, Andersen, Gamborg, Sorensen and Jess22, Reference Murphy, Stewart, Braithwaite, Beasley, Hancox and Mitchell24, Reference Trasande, Blustein, Liu, Corwin, Cox and Blaser26–Reference Rogawski, Platts-Mills, Seidman, John, Mahfuz, Ulak, Shrestha, Soofi, Yori, Mduma, Svensen, Ahmed, Lima, Bhutta, Kosek, Lang, Gottlieb, Zaidi, Kang, Bessong, Houpt and Guerrant28], prescription data [Reference Scott, Horton, Mamtani, Haynes, Goldberg, Lee and Lewis21, Reference Azad, Bridgman, Becker and Kozyrskyj23, Reference Schwartz, Pollak, Bailey-Davis, Hirsch, Cosgrove, Nau, Kress, Glass and Bandeen-Roche25, Reference Rogawski, Platts-Mills, Seidman, John, Mahfuz, Ulak, Shrestha, Soofi, Yori, Mduma, Svensen, Ahmed, Lima, Bhutta, Kosek, Lang, Gottlieb, Zaidi, Kang, Bessong, Houpt and Guerrant28–Reference Mbakwa, Scheres, Penders, Mommers, Thijs and Arts31, Reference Saari, Virta, Sankilampi, Dunkel and Saxen33] and pharmacy data [Reference Li, Chen, Ferber and Odouli32]. Outcomes reported ranged from continuous measures including BMI [Reference Murphy, Stewart, Braithwaite, Beasley, Hancox and Mitchell24, Reference Schwartz, Pollak, Bailey-Davis, Hirsch, Cosgrove, Nau, Kress, Glass and Bandeen-Roche25] or BMI-for-age z-score [Reference Trasande, Blustein, Liu, Corwin, Cox and Blaser26, Reference Mbakwa, Scheres, Penders, Mommers, Thijs and Arts31, Reference Saari, Virta, Sankilampi, Dunkel and Saxen33], weight-for-length z-score [Reference Rogawski, Westreich, Adair, Becker-Dreps, Sandler, Sarkar, Kattula, Ward, Meshnick and Kang27] or weight-for-age z-score [Reference Rogawski, Platts-Mills, Seidman, John, Mahfuz, Ulak, Shrestha, Soofi, Yori, Mduma, Svensen, Ahmed, Lima, Bhutta, Kosek, Lang, Gottlieb, Zaidi, Kang, Bessong, Houpt and Guerrant28] and rate of weight gain [Reference Gerber, Bryan, Ross, Daymont, Parks, Localio, Grundmeier, Stallings and Zaoutis29] to binary measures of overweight [Reference Ajslev, Andersen, Gamborg, Sorensen and Jess22, Reference Azad, Bridgman, Becker and Kozyrskyj23, Reference Trasande, Blustein, Liu, Corwin, Cox and Blaser26, Reference Mbakwa, Scheres, Penders, Mommers, Thijs and Arts31, Reference Saari, Virta, Sankilampi, Dunkel and Saxen33] or obesity [Reference Scott, Horton, Mamtani, Haynes, Goldberg, Lee and Lewis21, Reference Bailey, Forrest, Zhang, Richards, Livshits and DeRusso30, Reference Li, Chen, Ferber and Odouli32]. Outcomes of relative body mass, including BMI-for-age, weight-for-length, weight-for-age and binary classifications of overweight and obesity based on these measures, were expressed using a number of different references (Table 1). Outcome measures were by parental report in three studies [Reference Ajslev, Andersen, Gamborg, Sorensen and Jess22, Reference Murphy, Stewart, Braithwaite, Beasley, Hancox and Mitchell24, Reference Mbakwa, Scheres, Penders, Mommers, Thijs and Arts31], and measured in clinical or research settings in the rest [Reference Scott, Horton, Mamtani, Haynes, Goldberg, Lee and Lewis21, Reference Azad, Bridgman, Becker and Kozyrskyj23, Reference Schwartz, Pollak, Bailey-Davis, Hirsch, Cosgrove, Nau, Kress, Glass and Bandeen-Roche25–Reference Bailey, Forrest, Zhang, Richards, Livshits and DeRusso30, Reference Li, Chen, Ferber and Odouli32, Reference Saari, Virta, Sankilampi, Dunkel and Saxen33]. Studies employed a variety of analytical methods to assess associations, including those based upon covariance analysis [Reference Saari, Virta, Sankilampi, Dunkel and Saxen33], linear regression [Reference Murphy, Stewart, Braithwaite, Beasley, Hancox and Mitchell24–Reference Rogawski, Westreich, Adair, Becker-Dreps, Sandler, Sarkar, Kattula, Ward, Meshnick and Kang27, Reference Gerber, Bryan, Ross, Daymont, Parks, Localio, Grundmeier, Stallings and Zaoutis29], logistic regression [Reference Scott, Horton, Mamtani, Haynes, Goldberg, Lee and Lewis21–Reference Azad, Bridgman, Becker and Kozyrskyj23, Reference Trasande, Blustein, Liu, Corwin, Cox and Blaser26, Reference Li, Chen, Ferber and Odouli32, Reference Saari, Virta, Sankilampi, Dunkel and Saxen33], Cox proportional hazards models [Reference Bailey, Forrest, Zhang, Richards, Livshits and DeRusso30], generalised estimating equations [Reference Rogawski, Platts-Mills, Seidman, John, Mahfuz, Ulak, Shrestha, Soofi, Yori, Mduma, Svensen, Ahmed, Lima, Bhutta, Kosek, Lang, Gottlieb, Zaidi, Kang, Bessong, Houpt and Guerrant28, Reference Mbakwa, Scheres, Penders, Mommers, Thijs and Arts31] and longitudinal rate regression [Reference Gerber, Bryan, Ross, Daymont, Parks, Localio, Grundmeier, Stallings and Zaoutis29] (Table 1, online Supplementary Tables S2 and S3). None of the studies adjusted for all variables identified as potentially important confounders (Box 1, online Supplementary Table S3, Table 1).
Overall associations
Of the 13 studies identified, six reported overall measures of association. Of these, only one reported notable associations, between antibiotic use before 12 months of age and overweight risk at 9 and 12 years of age [adjusted odds ratio (aOR) for overweight at 12 years: 2.56, 95% confidence interval (95% CI) 1.36–4.79] [Reference Azad, Bridgman, Becker and Kozyrskyj23]. Five studies, of which two were based on populations of over 38 000 individuals, reported no overall association between antibiotic use and child body mass (Table 2) [Reference Ajslev, Andersen, Gamborg, Sorensen and Jess22, Reference Rogawski, Westreich, Adair, Becker-Dreps, Sandler, Sarkar, Kattula, Ward, Meshnick and Kang27–Reference Gerber, Bryan, Ross, Daymont, Parks, Localio, Grundmeier, Stallings and Zaoutis29, Reference Li, Chen, Ferber and Odouli32]. While all studies involved unmatched cohorts, the two larger studies additionally included matched longitudinal sub-studies of twins discordant for antibiotic exposure, with both confirming the null associations observed in their main analyses [Reference Gerber, Bryan, Ross, Daymont, Parks, Localio, Grundmeier, Stallings and Zaoutis29, Reference Li, Chen, Ferber and Odouli32].
Table 2. Overall associations reported between any antibiotic exposure in early life and measures of child body mass

zBMI, body mass index-for-age z-score; WAZ, weight-for-age z-score; aOR, adjusted odds ratio.
*p < 0.05.
See Table 1 and online Supplementary Table S3 for adjustments relating to analyses in each study.
Measures of association are relative to unexposed group unless stated otherwise. Blank cells indicate that no overall association estimate was reported.
Modification of associations by sex
Sex-specific differences in susceptibility to infections and in drug pharmacokinetics have been previously observed in humans [Reference Klein40–Reference Soldin and Mattison42]. Potential differential effects of antibiotic use on child overweight and obesity risk would thus be important to clarify. Three of four studies stratifying analyses by sex reported associations persisting only in boys following adjustment for covariates such as maternal BMI, smoking or socioeconomic status [Reference Azad, Bridgman, Becker and Kozyrskyj23, Reference Murphy, Stewart, Braithwaite, Beasley, Hancox and Mitchell24, Reference Saari, Virta, Sankilampi, Dunkel and Saxen33] (Table 3).
Table 3. Sex-specific associations reported between any antibiotic exposure in early life and measures of child body mass

Measures of association are relative to unexposed group unless stated otherwise.
zBMI, body mass index-for-age z-score; aOR, adjusted odds ratio.
*p < 0.05.
See Table 1 and online Supplementary Table S3 for adjustments relating to analyses in each study.
a Two-tailed p values reported, as 95% CIs were not provided.
b p for interaction = 0.002.
c p for interaction = 0.05.
Window of antibiotic exposure
Data from animal models suggest that earlier antibiotic exposure may have the capacity to more notably alter the intestinal microbiome, inducing metabolic changes leading to increased adiposity [Reference Cox Laura, Yamanishi, Sohn, Alekseyenko Alexander, Leung Jacqueline, Cho, Kim Sungheon, Li, Gao, Mahana, Zárate Rodriguez Jorge, Rogers Arlin, Robine, Loke and Blaser Martin43]. Similar effects would be important to clarify in humans. Evidence supporting specific early life windows of antibiotic exposure among studies included in this review was limited. Studies defined windows of exposure differently, often up to 6, 12 or 24 months of age [Reference Scott, Horton, Mamtani, Haynes, Goldberg, Lee and Lewis21–Reference Saari, Virta, Sankilampi, Dunkel and Saxen33]. Eight out of 13 studies stratified by exposure window; of these, five reported some evidence of stronger associations with antibiotic exposure in the first 3 to 6 months compared with later periods [Reference Scott, Horton, Mamtani, Haynes, Goldberg, Lee and Lewis21, Reference Azad, Bridgman, Becker and Kozyrskyj23, Reference Trasande, Blustein, Liu, Corwin, Cox and Blaser26, Reference Li, Chen, Ferber and Odouli32, Reference Saari, Virta, Sankilampi, Dunkel and Saxen33] (Fig. 2, online Supplementary Table S4). However, this pattern was not always consistently observed across multiple outcome measures or time points within the same study [Reference Azad, Bridgman, Becker and Kozyrskyj23, Reference Trasande, Blustein, Liu, Corwin, Cox and Blaser26], and few studies reported notable or consistent trends in associations across consecutive exposure windows [Reference Scott, Horton, Mamtani, Haynes, Goldberg, Lee and Lewis21, Reference Azad, Bridgman, Becker and Kozyrskyj23, Reference Saari, Virta, Sankilampi, Dunkel and Saxen33]. No notable trends in association, or associations with exposure at <6 months of age was noted in two studies [Reference Bailey, Forrest, Zhang, Richards, Livshits and DeRusso30, Reference Mbakwa, Scheres, Penders, Mommers, Thijs and Arts31], whereas another study reported associations between antibiotic use up to 24 months of age, but not up to 6 months of age, and rate of weight gain [Reference Gerber, Bryan, Ross, Daymont, Parks, Localio, Grundmeier, Stallings and Zaoutis29] (online Supplementary Table S4). Statistical tests comparing differences between exposure window categories, or comparing trends across categories, were not performed in any study [Reference Scott, Horton, Mamtani, Haynes, Goldberg, Lee and Lewis21, Reference Azad, Bridgman, Becker and Kozyrskyj23, Reference Trasande, Blustein, Liu, Corwin, Cox and Blaser26, Reference Gerber, Bryan, Ross, Daymont, Parks, Localio, Grundmeier, Stallings and Zaoutis29–Reference Saari, Virta, Sankilampi, Dunkel and Saxen33].
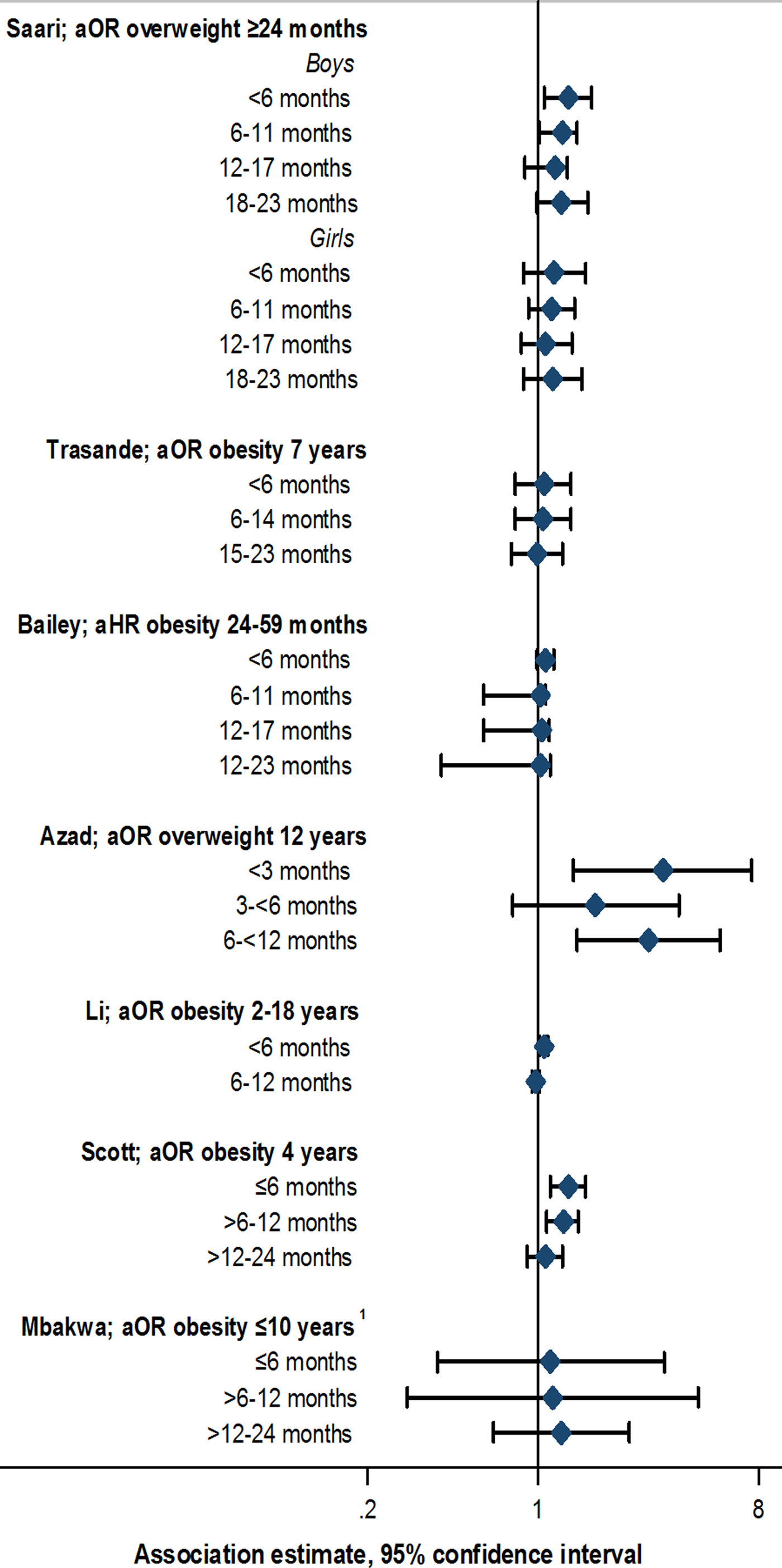
Fig. 2. Association between antibiotic exposure in specific age windows and measures of child body mass: selected risk measures reported in reviewed studies. aOR, adjusted odds ratio; aHR, adjusted hazard ratio. 1Estimates displayed for children exposed to two or more courses of antibiotics. See online Supplementary Table S3 for adjustments relating to analyses in each study, and online Supplementary Table S4 for additional estimates reported from other studies.
Repeated antibiotic exposure
Increased exposure to antibiotics in early life through repeated courses may be expected to result in more pronounced alterations in the intestinal microbiome, with potentially greater metabolic effects. The existence of a dose-dependent relationship between antibiotic use and childhood obesity risk would therefore be important to examine. Seven studies assessed the relation between the number of antibiotic courses and measures of child body mass [Reference Scott, Horton, Mamtani, Haynes, Goldberg, Lee and Lewis21, Reference Azad, Bridgman, Becker and Kozyrskyj23, Reference Rogawski, Westreich, Adair, Becker-Dreps, Sandler, Sarkar, Kattula, Ward, Meshnick and Kang27, Reference Gerber, Bryan, Ross, Daymont, Parks, Localio, Grundmeier, Stallings and Zaoutis29–Reference Mbakwa, Scheres, Penders, Mommers, Thijs and Arts31, Reference Saari, Virta, Sankilampi, Dunkel and Saxen33] (Fig. 3; online Supplementary Table S5). In four studies, there was some evidence of association between exposure to two or more antibiotic courses and increased measures of body mass, or increasing magnitude of associations with increasing course number [Reference Scott, Horton, Mamtani, Haynes, Goldberg, Lee and Lewis21, Reference Gerber, Bryan, Ross, Daymont, Parks, Localio, Grundmeier, Stallings and Zaoutis29, Reference Bailey, Forrest, Zhang, Richards, Livshits and DeRusso30, Reference Saari, Virta, Sankilampi, Dunkel and Saxen33]. However, trends were not consistent when examined across subgroups within these studies [Reference Scott, Horton, Mamtani, Haynes, Goldberg, Lee and Lewis21, Reference Gerber, Bryan, Ross, Daymont, Parks, Localio, Grundmeier, Stallings and Zaoutis29, Reference Bailey, Forrest, Zhang, Richards, Livshits and DeRusso30, Reference Saari, Virta, Sankilampi, Dunkel and Saxen33]. In two studies, no associations were observed across any category of number of antibiotic courses and measures of body mass [Reference Rogawski, Westreich, Adair, Becker-Dreps, Sandler, Sarkar, Kattula, Ward, Meshnick and Kang27, Reference Mbakwa, Scheres, Penders, Mommers, Thijs and Arts31], while one study reported decreased odds of overweight with exposure to an increasing number of antibiotic courses [Reference Azad, Bridgman, Becker and Kozyrskyj23]. Statistical tests for differences or trends across categories of exposure were reported only in this study, and were not notable (p for linear trend >0.1 in all cases; Figure 2, online Supplementary Table S5) [Reference Azad, Bridgman, Becker and Kozyrskyj23].
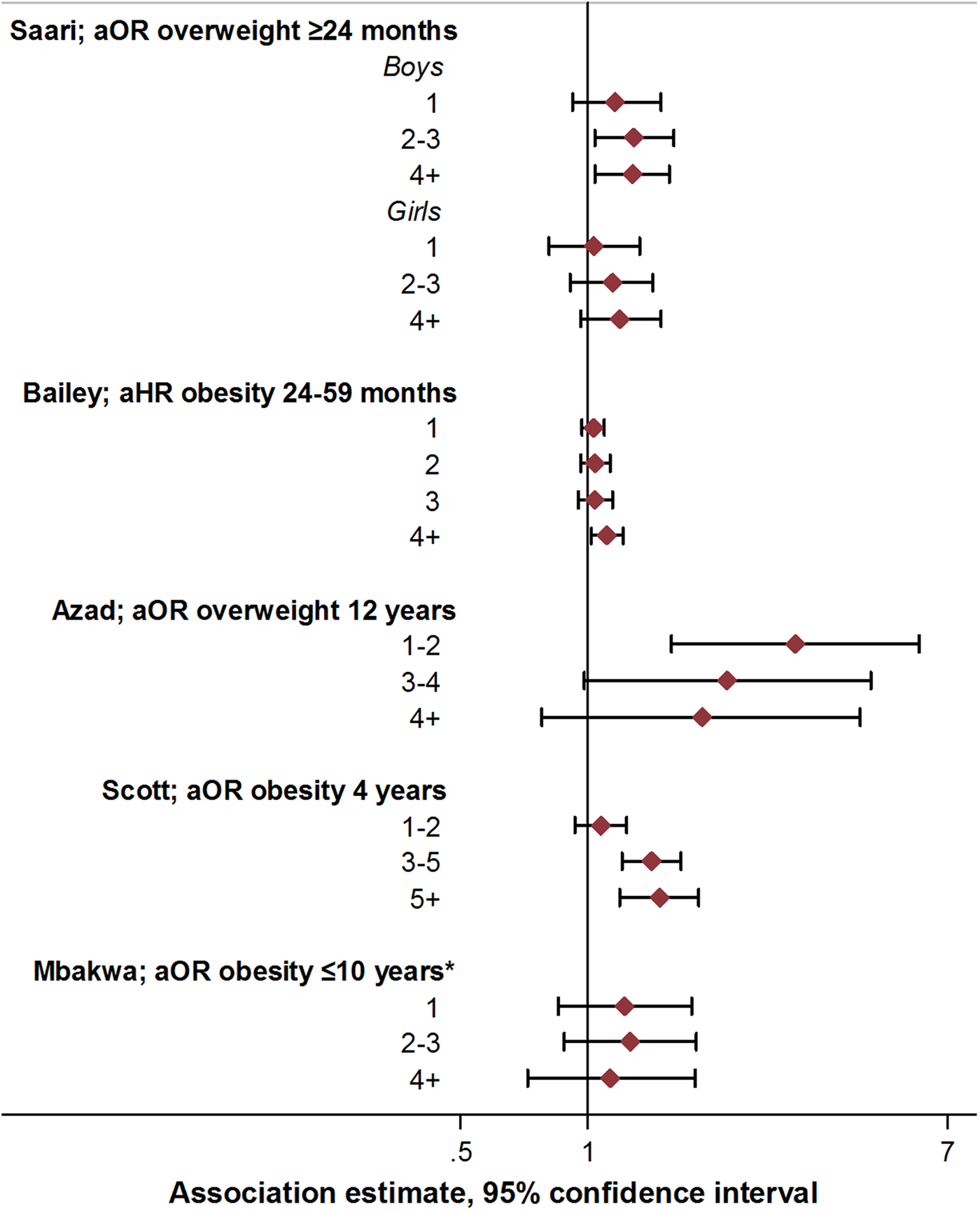
Fig. 3. Association between antibiotic course number and measures of child body mass: selected risk measures reported in reviewed studies. aOR, adjusted odds ratio; aHR, adjusted hazard ratio. See online Supplementary Table S3 for adjustments relating to analyses in each study, and online Supplementary Table S5 for additional estimates reported from other studies.
Antibiotic class
Broad spectrum antibiotics are thought to affect a wider range of intestinal microbiota, and thereby more notably alter intestinal microbial composition, with potential subsequent implications for metabolic dysregulation [Reference Modi, Collins and Relman44]. Evidence on effect modification of obesity risk by antibiotic class in the studies identified here was equivocal (Table 4). Of seven studies stratifying by antibiotic class, three reported stronger associations between broad v. narrow spectrum antibiotic use and later child body mass [Reference Rogawski, Platts-Mills, Seidman, John, Mahfuz, Ulak, Shrestha, Soofi, Yori, Mduma, Svensen, Ahmed, Lima, Bhutta, Kosek, Lang, Gottlieb, Zaidi, Kang, Bessong, Houpt and Guerrant28, Reference Bailey, Forrest, Zhang, Richards, Livshits and DeRusso30, Reference Saari, Virta, Sankilampi, Dunkel and Saxen33], whilst three suggested no differences in strength of associations [Reference Gerber, Bryan, Ross, Daymont, Parks, Localio, Grundmeier, Stallings and Zaoutis29, Reference Mbakwa, Scheres, Penders, Mommers, Thijs and Arts31, Reference Li, Chen, Ferber and Odouli32]. One study stratified by anti-anaerobic activity, suggesting increased obesity risk with greater than five courses of antibiotics with anti-anaerobic activity (aOR 1.46, 95% CI 1.09–1.96) [Reference Scott, Horton, Mamtani, Haynes, Goldberg, Lee and Lewis21].
Table 4. Association between antibiotic class and measures of child body mass

zBMI, body mass index-for-age z-score; WAZ, weight-for-age z-score; aOR, adjusted odds ratio.
Measures of association are relative to the unexposed group, and are presented with 95% CIs unless stated otherwise.
*p < 0.05.
See online Supplementary Table S3 for adjustments relating to analyses in each study.
a Estimates read off graph.
b Penicillin and amoxicillin classified as narrow spectrum operationally, all other agents considered broad.
c Antibiotics with anaerobic coverage: penicillins, imidazoles, lincosamides, tetracyclines; without anaerobic coverage: cephalosporins, macrolides, sulpha-containing agents, isoniazid, rifampicin, fluoroquinolones, aminoglycosides.
The effect of maternal BMI
Intestinal microbial composition in pregnant women has been shown to differ by weight status, and to influence the offspring intestinal microbiome [Reference Collado, Isolauri, Laitinen and Salminen45]. Such resulting variations are hypothesised to lead to differential weight-related responses to antibiotic exposure among children of overweight v. normal weight mothers [Reference Collado, Isolauri, Laitinen and Salminen45]. Two studies examined maternal weight as a potential effect modifier [Reference Ajslev, Andersen, Gamborg, Sorensen and Jess22, Reference Trasande, Blustein, Liu, Corwin, Cox and Blaser26]. One study reported associations between antibiotic use before 6 months of age and overweight at 7 years of age only among children of normal weight mothers (aOR 1.54, 95% CI 1.09–2.17) [Reference Ajslev, Andersen, Gamborg, Sorensen and Jess22]. The other study observed similar associations with child overweight at 38 months of age; however, these did not persist to overweight at 7 years [Reference Trasande, Blustein, Liu, Corwin, Cox and Blaser26].
Addressing confounding by clinical indication
Confounding by clinical indication occurs when the association between a clinical treatment and outcome is explained by the disease for which treatment was indicated [Reference Psaty, Koepsell, Lin, Weiss, Siscovick, Rosendaal, Pahor and Furberg46]. Measures to identify and address confounding by indication are thus crucial in studies examining clinical treatments [Reference Psaty, Koepsell, Lin, Weiss, Siscovick, Rosendaal, Pahor and Furberg46]. Four studies here considered infection status, with three stratifying by, or adjusting for, infection type or severity [Reference Rogawski, Westreich, Adair, Becker-Dreps, Sandler, Sarkar, Kattula, Ward, Meshnick and Kang27, Reference Rogawski, Platts-Mills, Seidman, John, Mahfuz, Ulak, Shrestha, Soofi, Yori, Mduma, Svensen, Ahmed, Lima, Bhutta, Kosek, Lang, Gottlieb, Zaidi, Kang, Bessong, Houpt and Guerrant28, Reference Bailey, Forrest, Zhang, Richards, Livshits and DeRusso30]. Only one study clearly examined the influence of the indication for antibiotic use on child obesity risk, reporting associations between the presence of early life infection and obesity (aOR 1.25, 95% CI 1.20–1.29), but not between antibiotic use and obesity among children with infection (aOR 1.01, 95% CI 0.98–1.04) [Reference Li, Chen, Ferber and Odouli32]. Notably, including both healthy children and those with untreated infection in the reference group resulted in associations between antibiotic use and child obesity that were comparable with those observed in other studies (aOR 1.20, 95% CI 1.17–1.22) [Reference Li, Chen, Ferber and Odouli32], suggesting that inappropriate accounting for confounding by indication may be an important limitation of other studies [Reference Scott, Horton, Mamtani, Haynes, Goldberg, Lee and Lewis21–Reference Trasande, Blustein, Liu, Corwin, Cox and Blaser26, Reference Gerber, Bryan, Ross, Daymont, Parks, Localio, Grundmeier, Stallings and Zaoutis29, Reference Mbakwa, Scheres, Penders, Mommers, Thijs and Arts31, Reference Saari, Virta, Sankilampi, Dunkel and Saxen33] (online Supplementary Tables S4 and S5).
Quantification of weight gain
Four studies examining measures of relative BMI or weight as outcomes (Table 1) quantified the absolute magnitude of weight gain associated with antibiotic exposure [Reference Schwartz, Pollak, Bailey-Davis, Hirsch, Cosgrove, Nau, Kress, Glass and Bandeen-Roche25–Reference Rogawski, Westreich, Adair, Becker-Dreps, Sandler, Sarkar, Kattula, Ward, Meshnick and Kang27, Reference Gerber, Bryan, Ross, Daymont, Parks, Localio, Grundmeier, Stallings and Zaoutis29]. Estimates ranged from increases of 0.001 and 0.032 kg at 2 years of age among boys and girls exposed to antibiotics in the first 6 months of life [Reference Rogawski, Westreich, Adair, Becker-Dreps, Sandler, Sarkar, Kattula, Ward, Meshnick and Kang27] to a maximum weight gain of 1.50 kg by 15 years of age associated with exposure to macrolides [Reference Schwartz, Pollak, Bailey-Davis, Hirsch, Cosgrove, Nau, Kress, Glass and Bandeen-Roche25]. One study estimated 0.09 kg increased weight among 38 month old children exposed to antibiotics up to 2 years of age [Reference Trasande, Blustein, Liu, Corwin, Cox and Blaser26], whilst another estimated a 0.15 kg increase between 2 and 5 years of age attributable to antibiotic use in the first 6 months of life [Reference Gerber, Bryan, Ross, Daymont, Parks, Localio, Grundmeier, Stallings and Zaoutis29]. In all cases, reported gains in weight approximated to <2.9% of the age-specific population median, proportions that would be unlikely to notably alter risk of adverse outcomes (online Supplementary Table S6).
Discussion
Clarifying the existence and nature of associations between antibiotic use in early life and subsequent risk of child obesity is essential to guide appropriate policies and strategies relating to child health and to use of antibiotic medications. We examined observational studies exploring the relation between early childhood antibiotic use and measures of child body mass. The totality of evidence was inconsistent, with key differences in study design potentially contributing to varying conclusions. Overall, there was no strong evidence supporting clinically relevant associations between antibiotic exposure and body mass in childhood. Further comprehensive and comparable studies using routine clinical data may help clarify this association.
Although the exact measures varied by study, associations reported between antibiotic use and child body mass were generally not appreciable in terms of effect size and statistical strength. In certain studies, associations were only apparent when examining exposure to four or more antibiotic courses [Reference Scott, Horton, Mamtani, Haynes, Goldberg, Lee and Lewis21, Reference Bailey, Forrest, Zhang, Richards, Livshits and DeRusso30]. Trends or patterns, such as those relating to consecutive exposure windows or antibiotic class, were often inconsistent across studies or study subgroups [Reference Scott, Horton, Mamtani, Haynes, Goldberg, Lee and Lewis21, Reference Azad, Bridgman, Becker and Kozyrskyj23, Reference Rogawski, Platts-Mills, Seidman, John, Mahfuz, Ulak, Shrestha, Soofi, Yori, Mduma, Svensen, Ahmed, Lima, Bhutta, Kosek, Lang, Gottlieb, Zaidi, Kang, Bessong, Houpt and Guerrant28–Reference Saari, Virta, Sankilampi, Dunkel and Saxen33]. Notably, studies quantifying observed associations did not indicate important absolute gains in weight related to antibiotic exposure, suggesting that if this association does exist, it is not likely to be clinically relevant [Reference Schwartz, Pollak, Bailey-Davis, Hirsch, Cosgrove, Nau, Kress, Glass and Bandeen-Roche25–Reference Rogawski, Westreich, Adair, Becker-Dreps, Sandler, Sarkar, Kattula, Ward, Meshnick and Kang27, Reference Gerber, Bryan, Ross, Daymont, Parks, Localio, Grundmeier, Stallings and Zaoutis29].
Findings reported in studies must also be considered within the context of their specific limitations. These limitations ranged from small population or subgroup sample sizes [Reference Azad, Bridgman, Becker and Kozyrskyj23, Reference Rogawski, Westreich, Adair, Becker-Dreps, Sandler, Sarkar, Kattula, Ward, Meshnick and Kang27, Reference Mbakwa, Scheres, Penders, Mommers, Thijs and Arts31] to errors in ascertainment of antibiotic exposure measured through parental recall [Reference Ajslev, Andersen, Gamborg, Sorensen and Jess22, Reference Murphy, Stewart, Braithwaite, Beasley, Hancox and Mitchell24, Reference Trasande, Blustein, Liu, Corwin, Cox and Blaser26, Reference Rogawski, Westreich, Adair, Becker-Dreps, Sandler, Sarkar, Kattula, Ward, Meshnick and Kang27] or by prescriptions [Reference Azad, Bridgman, Becker and Kozyrskyj23, Reference Schwartz, Pollak, Bailey-Davis, Hirsch, Cosgrove, Nau, Kress, Glass and Bandeen-Roche25, Reference Rogawski, Westreich, Adair, Becker-Dreps, Sandler, Sarkar, Kattula, Ward, Meshnick and Kang27, Reference Gerber, Bryan, Ross, Daymont, Parks, Localio, Grundmeier, Stallings and Zaoutis29, Reference Bailey, Forrest, Zhang, Richards, Livshits and DeRusso30] or examination of only specific kinds of antibiotics [Reference Ajslev, Andersen, Gamborg, Sorensen and Jess22], and the inability to measure adherence to treatment. Such errors in ascertainment of antibiotic exposure may limit the ability of studies to detect associations. Not all studies controlled for a comprehensive range of relevant confounders, and most importantly, most studies did not sufficiently examine confounding by clinical indication [Reference Scott, Horton, Mamtani, Haynes, Goldberg, Lee and Lewis21–Reference Mbakwa, Scheres, Penders, Mommers, Thijs and Arts31, Reference Saari, Virta, Sankilampi, Dunkel and Saxen33, Reference Psaty, Koepsell, Lin, Weiss, Siscovick, Rosendaal, Pahor and Furberg46]. We found only one study to have accounted for this, which reported no association between antibiotic use and obesity risk among children with infection, and demonstrated the potential for observing erroneous positive associations when including uninfected children in the reference group [Reference Li, Chen, Ferber and Odouli32]. These findings indicate that associations reported in other studies must be cautiously interpreted, and highlight the need for further, more robust evidence [Reference Scott, Horton, Mamtani, Haynes, Goldberg, Lee and Lewis21–Reference Mbakwa, Scheres, Penders, Mommers, Thijs and Arts31, Reference Saari, Virta, Sankilampi, Dunkel and Saxen33].
In this review, we were unable to synthesise effect estimates meaningfully given the high degree of heterogeneity among studies, which included differences in study populations and settings, observation windows, definitions of exposures and outcomes, inclusion of potential confounders and effect modifiers and choice of analytic approaches. To improve comparability and enable synthesis of data in meta-analyses, future studies should ideally: (i) define all exposures and outcomes, including their respective time windows, consistently with previous studies, (ii) consider using multiple analytic methods as employed in previous research and (iii) control for all confounding factors included in previous studies, including accounting for confounding by indication. As stronger evidence emerges regarding the characterisation and contribution of related measures in childhood, such as waist circumference, to future disease risk, studies may also consider such measures as potentially important outcomes [Reference Azad, Bridgman, Becker and Kozyrskyj23, Reference Khadilkar, Ekbote, Chiplonkar, Khadilkar, Kajale, Kulkarni, Parthasarathy, Arya, Bhattacharya and Agarwal47]. Given the effect sizes observed here, future studies must be based on large populations in order to allow for more precise estimation of association. Given this required scale and complexity, such analyses would be best facilitated by the use of routine clinical data captured in electronic health records, as demonstrated in key studies reviewed here [Reference Scott, Horton, Mamtani, Haynes, Goldberg, Lee and Lewis21, Reference Azad, Bridgman, Becker and Kozyrskyj23, Reference Schwartz, Pollak, Bailey-Davis, Hirsch, Cosgrove, Nau, Kress, Glass and Bandeen-Roche25, Reference Gerber, Bryan, Ross, Daymont, Parks, Localio, Grundmeier, Stallings and Zaoutis29, Reference Bailey, Forrest, Zhang, Richards, Livshits and DeRusso30, Reference Li, Chen, Ferber and Odouli32]. Electronic health records provide a platform for obtaining large-scale, longitudinal patient data, as well as facilitating the collection of information on infection type, test results, treatment dose, duration and response and other potential confounding factors. They may additionally be utilised to prospectively recruit patients into future, large scale studies.
The evidence reviewed here is insufficient to justify the risk of child obesity as an important reason to limit antibiotic use. However, multiple other reasons exist for improving monitoring and management of antibiotic use, including the development of antibiotic resistance. The importance of antibiotic stewardship programmes to achieve appropriate use of antibiotics is increasingly being recognised, with recent evidence suggesting notable effects of such initiatives on overall antibiotic use, infection incidence and other patient outcomes in hospitals [Reference Baur, Gladstone, Burkert, Carrara, Foschi, Döbele and Tacconelli48–Reference Schuts, Hulscher, Mouton, Verduin, Stuart, Overdiek, van der Linden, Natsch, Hertogh, Wolfs, Schouten, Kullberg and Prins50]. The expansion of these programmes may help address antibiotic resistance on a wider scale, and thus contribute to reducing associated morbidity and mortality [Reference Goff, Kullar, Goldstein, Gilchrist, Nathwani, Cheng, Cairns, Escandón-Vargas, Villegas, Brink, van den Bergh and Mendelson51]. However, the evidence here does not support a tangible effect of such programmes on the current burden of child obesity, and suggests that resources aimed towards child obesity prevention would be better placed in existing strategies and initiatives targeting appropriate nutrition and physical activity in early life and later childhood [52].
We followed a systematic literature search and selection method to identify all relevant studies investigating the relation between early life antibiotic use and child obesity, and present here a detailed qualitative synthesis of identified studies. However, certain limitations must be considered. We included only English language publications in our search, and are unable to rule out publication bias, although the presence of such bias would support our interpretation of the current evidence. The inability to quantitatively synthesise effect estimates limits our ability to present more definitive conclusions, and points towards the need for more harmonised methods to assess this association. Finally, this review examined only exposure to antibiotics for medical use, and did not capture potential exposure through other methods, such as low-dose antibiotics present in food. Although this has been recognised as a potentially important source of exposure [Reference Cox and Blaser11, Reference Riley, Raphael and Faerstein53], few studies, to our knowledge, have quantified it and explored its association with outcomes in humans, and it remains a potentially important area for future research.
In this review, we observed no robust evidence implicating antibiotic use as an important risk factor for child obesity, with notable heterogeneity among studies limiting more conclusive interpretations. Further more detailed, comparable studies are required to clearly determine the existence of such associations. Regardless, there is a well-justified need to achieve more appropriate use of antibiotics [Reference Goff, Kullar, Goldstein, Gilchrist, Nathwani, Cheng, Cairns, Escandón-Vargas, Villegas, Brink, van den Bergh and Mendelson51]. Furthermore, multiple comprehensively researched risk factors may be targeted to address child obesity [52]. Efforts in these directions will be key for addressing both these concerning public-health issues on a large scale.
Supplementary material
The supplementary material for this article can be found at https://doi.org/10.1017/gheg.2018.16.
Financial support
This work was supported by the Wellcome Trust (grant number 206194), the African Partnership for Chronic Disease Research (Medical Research Council UK partnership grant number MR/K013491/1) and the National Institute for Health Research Cambridge Biomedical Research Centre (UK). EP is supported by the Gates Cambridge Trust.
Conflict of interest
None.
Ethical standards
The authors assert that all procedures contributing to this work comply with the ethical standards of the relevant national and institutional committees on human experimentation and with the Helsinki Declaration of 1975, as revised in 2008.









