Introduction
Ménière's disease is an inner ear disorder that is characterised by recurrent spontaneous episodes of a range of symptoms that can include vertigo, fluctuating low-frequency sensorineural hearing loss, tinnitus and aural fullness.Reference Surgery, Monsell and Balkany1 The prevalence of Ménière's disease in the US population in 2010 was 190 per 100 000.Reference Harris and Alexander2 Furthermore, it is a disease more common among adults, especially for those who are in their fourth decade of life, and patients usually first experience symptoms between the ages of 20 and 60 years.Reference Da Costa, De Sousa and De Toledo Piza3 Patients initially have symptoms on one side with some patients developing symptom bilaterality after many years.Reference Harcourt, Barraclough and Bronstein4 The male to female ratio varies from nearly equal for both sexes to a slight female preponderance that can go up to 1:1.3.Reference Da Costa, De Sousa and De Toledo Piza3 The diagnosis of Ménière's disease is not always straightforward as there are no widely accepted confirmatory diagnostic tests. Patients with Ménière's disease may not present with all of the classical symptoms as described above during the first encounter. Therefore, Ménière's disease can only be diagnosed at a later stage when the patient already has more than one episode of disease flare-up.Reference Surgery, Monsell and Balkany1–Reference Lingam, Connor, Casselman and Beale6 Currently, most studies are using the diagnostic criteria for Ménière's disease set by the American Academy of Otolaryngology–Head and Neck Surgery (AAO–HNS) criteria in 1995, which take into account the natural history of the disease; Ménière's disease is classified as certain, definite, probable or possible.Reference Surgery, Monsell and Balkany1
The pathological hallmark of this disease is believed to be endolymphatic hydrops, which was first observed in post-mortem examination of patients with Ménière's disease in 1938; however, a causal relationship has never been proven.Reference Harcourt, Barraclough and Bronstein4 Endolymphatic hydrops is the distension of the membranous labyrinth of the inner ear, which can occur in both the vestibule and the cochlea.Reference Harcourt, Barraclough and Bronstein4 This hydrops was conventionally thought to cause the symptoms experienced by the patients affected with the disorder because the sensory and neural elements of the inner ear are damaged as a result of the release of the endolymphatic fluid into the perilymph.Reference Harcourt, Barraclough and Bronstein4,Reference Ghossaini and Miller5 Thus, visualising endolymphatic hydrops could be a potential tool in confirming the diagnosis and monitoring the progress of Ménière's disease in vivo. However, endolymphatic hydrops can also be caused by trauma, viral infections, autoimmune diseases and electrolyte imbalance, which can potentially confound the findings of magnetic resonance imaging (MRI).
The potential to visualise the endolymphatic hydrops in vivo was first established in an animal study using post-intratympanic gadolinium MRI.Reference Niyazov, Andrews, Strelioff, Sinha and Lufkin7 Since then, various projects have studied the use of MRI to visualise endolymphatic hydrops after intravenous (IV) or intratympanic administration of gadolinium.Reference Fiorino, Pizzini, Beltramello, Mattellini and Barbieri8,Reference Carfrae, Holtzman, Eames, Parnes and Lupinetti9 In an additional study, three-dimensional (3D) fluid-attenuated inversion recovery (FLAIR) and 3D-real IR (three-dimensional real inversion recovery) sequences are used in post-gadolinium endolymphatic hydrops imaging because they can produce images that differentiate between the endolymphatic space and the perilymph.Reference Connor and Pai10 The delayed acquisition of MRI scan post-gadolinium is also important because the perilymph is most enhanced after around 3.5 to 4.5 hours, appearing as a bright signal in the image of 3D-FLAIR.Reference Conte, Lo Russo, Calloni, Sina, Barozzi and Berardino11 Conversely, the endolymph is non-enhanced, appearing as a dark signal in the image of 3D-FLAIR.Reference Conte, Lo Russo, Calloni, Sina, Barozzi and Berardino11 This is because the perilymph is more permeable to the gadolinium contrast compared with the endolymphatic space and the presence of the blood-labyrinth barrier does not allow the contrast to easily enter the endolymph.Reference Connor and Pai10
In order to assess the presence and severity of endolymphatic hydrops using MRI, Nakashima et al. have developed a grading system to categorise the endolymphatic hydrops in both the vestibule and cochlea as none, mild or significant.Reference Nakashima, Naganawa, Pyykkö, Gibson and Sone12 Vestibular endolymphatic hydrops is graded as none when the ratio is less than 1:3, mild when the ratio is between 1:3 and 1:2, or significant when the ratio is more than 1:2.Reference Nakashima, Naganawa, Pyykkö, Gibson and Sone12 As for cochlear endolymphatic hydrops, the grading is dependent on the displacement of the Reissner's membrane.Reference Nakashima, Naganawa, Pyykkö, Gibson and Sone12 Cochlear endolymphatic hydrops is graded as none if no displacement of the Reissner's membrane can be identified, mild if the Reissner's membrane is displaced but not exceeding the space occupied by the scala vestibuli or significant if the Reissner's membrane is displaced to the point of exceeding the scala vestibuli.Reference Nakashima, Naganawa, Pyykkö, Gibson and Sone12 An alternative to this grading system is to evaluate the morphology of the saccule and compare this with the utricle to form the inversion of saccule and utricle ratio as it was noted that the saccule is more commonly affected in endolymphatic hydrops compared with the utricle.Reference Attyé, Eliezer, Boudiaf, Tropres, Chechin and Schmerber13
Despite the recent reports on delayed-acquisition post-gadolinium MRI and endolymphatic hydrops, a systematic review of the current literature with a focus on the techniques used and their clinical implications is missing. On these grounds, our objectives were to assess imaging methods that are optimal for visualising endolymphatic hydrops in patients with Ménière's disease and to evaluate the use of visualising endolymphatic hydrops in aiding the diagnosis of Ménière's disease.
Materials and methods
Review question
The topic of interest for this review was the strength of delayed acquisition of post-gadolinium MRI in diagnosing Ménière's disease. The key question that arose from this topic was ‘Is visualisation of endolymphatic hydrops using post-gadolinium MRI adequate to diagnose Ménière's disease?’
Inclusion and exclusion criteria
We searched for studies assessing the strengths and limitations of delayed-gadolinium MRI for detecting endolymphatic hydrops. We specifically focused on whether delayed-gadolinium MRI can detect endolymphatic hydrops and what the clinical implications of such findings are. We also documented the technical parameters used by each study with focus on the route of gadolinium administration and the delay between gadolinium administration and MRI acquisition. Table 1 demonstrates the inclusion and exclusion criteria used to perform the literature search in this review.
Table 1. Inclusion and exclusion criteria
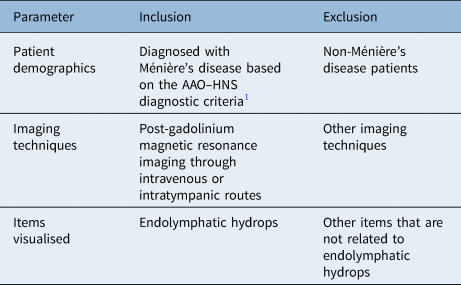
AAO–HNS = American Academy of Otolaryngology–Head and Neck Surgery
Search strategy
Medline and Embase were used in the systematic search. In addition to that, the reference sections listed in relevant studies were manually searched to find other potential studies that met the inclusion criteria. The literature search included the criteria: Ménière's disease, post-gadolinium MRI and endolymphatic hydrops, with articles in the English language and without a time limit; case reports were excluded. Preferred Reporting Items for Systematic Reviews and Meta-analyses (‘PRISMA’) guidelines were used for the methodology of this review.Reference Moher, Liberati, Tetzlaff and Altman14
Out of a total of 30 results, we ruled out 17 based on their title and abstracts. Finally, a total of 11 studies were included for review. Any duplicates that were found during the manual search were excluded from this review. The methodology of the search strategy carried out is summarised in Figure 1.

Fig. 1. Flow diagram adapted from Preferred Reporting Items for Systematic Reviews and Meta-Analyses (‘PRISMA’) 2009.Reference Moher, Liberati, Tetzlaff and Altman14
Data extraction
Two investigators conducted the data extraction independently. The following information was extracted from the articles that met the eligibility criteria: number of patients with Ménière's disease, how the diagnosis of Ménière's disease (or no Ménière's disease) was set, type (strength of magnetic field) of scan and sequences used, dose and route of gadolinium, number of hours post-gadolinium administration that the MRI was acquired, items or areas of interest assessed on the scans, assessment of endolymphatic hydrops and where exactly in the inner ear this was assessed, the person(s) who carried out the assessment and whether the assessment was blinded or not (including information on whether the scan assessment was performed by multiple assessors independently and in such case reports on inter-observer reliability), and side effects. The data were extracted and then tabulated in the form of an Excel® spreadsheet.
Assessment of bias
We used the Quality Assessment of Diagnostic Accuracy Studies-2 tool to assess the bias in the included studies.Reference Whiting, Rutjes, Westwood, Mallett, Reitsma and Leeflang15 Because of the predominantly experimental nature of the studies, it was difficult to identify a bias tool that suited the needs of the current review; however, as we were primarily looking into diagnostic accuracy, the four-domain Quality Assessment of Diagnostic Accuracy Studies-2 tool was the most appropriate one. Additionally, we used screening of the studies by two independent investigators to eliminate any bias. The results of the Quality Assessment of Diagnostic Accuracy Studies-2 tool are highlighted in Table 2 and Figure 2.

Fig. 2. Graph illustrating the proportion of studies with low, high or unclear risk of bias and concerns regarding applicability using the Quality Assessment of Diagnostic Accuracy Studies-2 tool.Reference Whiting, Rutjes, Westwood, Mallett, Reitsma and Leeflang15 QUADAS = Quality Assessment of Diagnostic Accuracy Studies-2 tool
Table 2. Results of Quality Assessment of Diagnostic Accuracy Studies-2 assessmentReference Whiting, Rutjes, Westwood, Mallett, Reitsma and Leeflang15

Results
Study characteristics and magnetic resonance imaging settings
The number of participants for each study varied, ranging from 6 to 68 participants; they were all diagnosed with Ménière's disease using the AAO–HNS diagnostic criteria.Reference Surgery, Monsell and Balkany1 The type of scanner used for all of the studies was the 3T MRI scanner, with 3D-FLAIR being the most common sequence used; 8 of 11 studies used 3D-FLAIR only, 1 of 11 studies used 3D-real IR only, and 2 of 11 studies used both 3D-FLAIR and 3D-real IR.
As for the route of gadolinium administration, most of the studies used IV only (8 of 11) with the rest being either intratympanic only (2 of 11), or both IV and intratympanic (1 of 11). The dose of gadolinium used for the IV route varied from 0.1 mmol/kg (6 of 11) to 0.2 mmol/kg (3 of 11), whereas the dose for the intratympanic route ranged from 5-fold dilution (1 of 11) to 8-fold dilution (2 of 11). In terms of the time of acquisition of MRI post-gadolinium, 4 hours was the most widely used (8 of 11) for the IV route. As for the intratympanic route, 24 hours was used (3 of 11). Shi et al. and Sano et al. used multiple times of acquisition, including 3 hours, 6 hours and 12 hours for Shi et al. and 10 minutes for Sano et al.Reference Sano, Teranishi, Yamazaki, Isoda, Naganawa and Sone22,Reference Shi, Li, Yin and Zou24 Additionally, Attyé et al.Reference Attyé, Eliezer, Medici, Tropres, Dumas and Krainik20 reported an acquisition time of between 4.5 and 5.5 hours. Table 3 demonstrates the number of patients and the imaging techniques used in each study.
Table 3. Number of patients and imaging techniques used for each study

All studies were performed in a 3T MRI scanner. MD = Ménière's disease; MRI = magnetic resonance imaging; 3D-FLAIR = three-dimensional fluid-attenuated inversion recovery; 3D-real IR = three-dimensional real inversion recovery
Radiological assessment and findings
Table 4 outlines the structures that were visualised in the MRI, measurements that were used to evaluate endolymphatic hydrops and the persons who evaluated the scans for each study. Endolymphatic hydrops of both the vestibuli and cochlea were evaluated for almost all of the studies (9 of 11), with two studies that only evaluated the vestibular endolymphatic hydrops. All of the studies that investigated cochlear endolymphatic hydrops determined its presence by observing if there was any dilatation of the cochlear duct. As for vestibular endolymphatic hydrops, the cut-off values for the vestibular endolymphatic space to the entire vestibule ratio included 33.3 per cent (7 of 11), 45 per cent (1 of 11) and 50 per cent (2 of 11). Additionally, Attyé et al. also described the use of saccule to utricle ratio inversion technique to determine the presence of endolymphatic hydrops in the vestibule.Reference Attyé, Eliezer, Boudiaf, Tropres, Chechin and Schmerber13,Reference Attyé, Eliezer, Medici, Tropres, Dumas and Krainik20
Table 4. Outline of items visualised on the scan, assessments used to identify EH, person who assessed the scans and whether the assessors are blinded or not for each study

EH = endolymphatic hydrops; VES = vestibular endolymphatic space; SURI = saccule to utricle ratio inversion
As for the number of radiologists or persons who evaluated the scans for each study, 6 of 11 had 2 people evaluating the scans, 3 of 11 had only one person evaluating the scans and 2 of 11 did not mention who evaluated the scans. A total of 8 of 11 studies stated that the evaluators were blinded to clinical data, and the rest (3 of 11) did not mention any form of blinding.
Discussion
Main findings
This is, to our knowledge, the first review to assess the potential strengths and biases associated with the evaluation of endolymphatic hydrops using the delayed gadolinium MRI, as well as the clinical applicability of this technique. We included a series of main findings of both radiological and clinical significance. Firstly, all studies used the 3T MRI scanner, with 3D-FLAIR being the most commonly used MRI sequence to assess endolymphatic hydrops. Secondly, the IV route of administering gadolinium was overall found to be more ubiquitous than the intratympanic route. Thirdly, all studies used some form of semi-quantitative grading systems in their assessment of endolymphatic hydrops. As for the timing of acquisition of MRI, four hours appeared to be the most commonly used for IV gadolinium. In contrast, 24 hours is more commonly used for intratympanic gadolinium. Moreover, most of the studies included experienced radiologists who were blinded to clinical data for assessing endolymphatic hydrops of both the vestibule and cochlea. Despite the relatively small number of studies carried out so far, the protocols used appear grossly uniform with only minor modifications.
However, we identified bias mostly in the patient selection but also in the methodology of scan assessment (lack of independent or blind review in some studies); to overcome these limitations further studies will be required to strengthen the evidence of using MRI for diagnostic purposes in Ménière's disease.
Technical considerations and clinical implications
There are several explanations as to why certain parameters were used more frequently in these studies. Firstly, 3D-FLAIR was used more frequently than 3D-real IR as it is more sensitive to gadolinium contrast and no post-imaging processing is required.Reference Connor and Pai10 Additionally, administering gadolinium through the IV route provides a shorter acquisition time than the intratympanic route and allows for visualising of both the inner ears.Reference Connor and Pai10 As for the dose of gadolinium used, it is dependent on the optimisation of the imaging sequence. Lower concentrations are needed for heavily T2-weighted 3D-FLAIR, which makes it more ideal in clinical settings as it could reduce any concerns about potential side effects of gadolinium on the patients.Reference Sano, Teranishi, Yamazaki, Isoda, Naganawa and Sone22 There are multiple semi-quantitative grading systems used in these studies, which demonstrates the lack of standardisation in assessing endolymphatic hydrops. Therefore, the objectivity of determining endolymphatic hydrops on MRI scans is difficult to examine.
Additionally, all published studies used 3T MRI, which, although more accessible in many places than in the past, is still not widely available. There are no studies using a weaker magnetic field; thus, delayed acquisition post-gadolinium MRI for identifying endolymphatic hydrops is not possible in many areas. This limits its applicability but also restricts the set-up of future studies.
With respect to the clinical implication of the available studies, while the endolymphatic spaces can well be visualised, it remains unclear to what extent the presence or absence of endolymphatic hydrops can confirm or exclude the diagnosis of Ménière's disease. Attyé et al. recently showed that endolymphatic hydrops can be related to sensorineural hearing loss rather than the presence of Ménière's disease itself.Reference Attyé, Eliezer, Medici, Tropres, Dumas and Krainik20 While the endolymphatic spaces are well visualised, the diagnosis of Ménière's disease cannot be set based on purely radiological grounds. Although MRI can facilitate the diagnosis, Ménière's disease still remains predominantly a clinical diagnosis.
Strengths and limitations of the included studies
There are several limitations of the studies that are included in this review. Firstly, the sample size of each study is small because of the low prevalence of Ménière's disease; additionally, the included studies did not comment on the power of the enrolled cohorts. The small sample size makes it more difficult to validate the effectiveness of these imaging techniques in identifying endolymphatic hydrops and its relation to Ménière's disease. Secondly, some studies only had one radiologist evaluating the scans, which methodologically gives rise to questions about the robustness and the reliability of the measurements. Moreover, some of the studies did not mention any form of blinding, which may lead to biases when it comes to the interpretation of scans in patients with Ménière's disease. Additionally, the majority of these studies did not use healthy controls with no inner ear pathologies to make comparisons with patients diagnosed with Ménière's disease.
We used the Quality Assessment of Diagnostic Accuracy Studies-2 tool to identify and highlight these limitations to include accurate data and meaningful conclusions that help with the clinical significance of such imaging techniques. According to the Quality Assessment of Diagnostic Accuracy Studies-2 tool, patient selection appeared to be at high risk of bias for most of the studies because the patients are not randomly selected; some studies did not include all of the patients recruited in the results for various reasons. Additionally, there is a high risk of bias for the index test domain of the Quality Assessment of Diagnostic Accuracy Studies-2 tool for 6 out of 11 studies in the review.
Conclusion
Recent advances in MRI techniques have enabled us to identify endolymphatic hydrops on delayed acquisition post-gadolinium MRI; this carries potential in aiding the diagnosis of Ménière's disease and also guiding the clinicians to decide on optimal treatment. However, these techniques need to be more thoroughly validated before they can be widely used in the management of patients with Ménière's disease; the availability of a 3T MRI scanner could also be an issue. We identified bias in most of the included studies; thus, future studies including larger number of patients and control groups should help with further improving this developing and promising technique.
Competing interests
None declared








