Individuals with schizophrenia die 15–20 years earlier than the general population, with 60% or more of this premature mortality due to physical illness, predominantly cardiovascular disease. Reference Beary, Hodgson and Wildgust1–Reference Osby, Correia, Brandt, Ekbom and Sparen6 The metabolic syndrome describes a cluster of risk factors for cardiovascular disease, including dyslipidaemia, hypertension, impaired glucose tolerance/type 2 diabetes mellitus and central obesity. 7–Reference Papanastasiou12 A recent meta-analysis demonstrated across 93 studies that a third of people with schizophrenia have metabolic syndrome, with rates as high as 69% in those with chronic illness. Reference Vancampfort, Stubbs, Mitchell, De Hert, Wampers and Ward13 The odds ratio (OR) of metabolic syndrome in chronic schizophrenia compared with the general population is 2.35 (95% CI 1.68–2.39). Reference Vancampfort, Wampers, Mitchell, Correll, De Herdt and Probst14 Chronic schizophrenia is associated with lipid disorder, specifically reduced high-density lipoprotein (HDL) cholesterol compared with population controls (OR = 2.35, 95% CI 1.78–3.10) and raised triglyceride levels (OR = 2.73, 95% CI 1.95–3.83). Reference Vancampfort, Wampers, Mitchell, Correll, De Herdt and Probst14 Although antipsychotic treatment could contribute to this, several studies have found evidence for alterations in lipids at illness onset, Reference Chen, Du, Yin, Yang, Nie and Wang15–Reference Misiak, Laczmanski, Sloka, Szmida, Piotrowski and Loska20 suggesting that alterations might be intrinsic to schizophrenia. Perry et al recently conducted a meta-analysis examining glucose dysregulation in patients with first-episode psychosis (FEP) and in subgroup analyses explored differences in lipid parameters between FEP and healthy control groups. Reference Perry, Mcintosh, Weich, Singh and Rees21 However, their meta-analysis did not systematically search for studies examining lipid parameters, so key studies may have been missed. Moreover, sensitivity analyses examining parameters known to affect dyslipidaemia were not performed. Furthermore, their meta-analysis did not consider adipocytokines in FEP. In view of this we set out to investigate whether alterations in lipid and adipocytokine parameters are evident in individuals at the onset of psychotic illness, with no or minimal antipsychotic exposure.
Method
A systematic review was performed according to Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) and Meta-Analysis of Observational Studies in Epidemiology (MOOSE) guidelines (online supplement DS1). Reference Moher, Liberati, Tetzlaff and Altman22,Reference Stroup, Berlin, Morton, Olkin, Williamson and Rennie23 Two reviewers (T.P. and K.B.) independently searched Medline (from 1946 to 14 December 2016), EMBASE (from 1947 to 2016, week 50) and PsycINFO (from 1806 to December 2016, week 1). These databases were selected because they comprehensively cover psychiatric and medical journals likely to publish studies on this topic, and are consistent with previous meta-analyses in the field focusing on metabolic dysfunction and cardiovascular disease risk factors in psychotic illness. Reference Gurillo, Jauhar, Murray and MacCabe24–Reference Mitchell, Vancampfort, De Herdt, Yu and De Hert26 The following keywords were used: (‘schizophrenia’ OR ‘schizo-affective’ OR ‘psychosis’) AND (‘early’ OR ‘first episode’ OR ‘risk’ OR ‘prodrome’) AND (‘metabolic’ OR ‘lipid’ OR ‘cholesterol’ OR ‘HDL’ OR ‘LDL’ OR ‘lipoprotein’ OR ‘triglyceride’ OR ‘adiponectin’ OR ‘ghrelin’ OR ‘leptin’ OR ‘resistin’ OR ‘chemerin’ OR ‘omentin’ OR ‘apelin’ or ‘adipocytokine’ OR ‘adipokine’). This was complemented by hand-searching of meta-analyses and review articles, which are listed in online supplement DS1, together with an a priori protocol. Inclusion criteria were:
-
(a) a DSM or ICD diagnosis of schizophrenia, schizoaffective disorder, schizophreniform disorder, schizophrenia spectrum or psychotic disorder not otherwise specified or an at-risk mental state for psychosis according to research criteria; Reference Miller, McGlashan, Woods, Stein, Driesen and Corcoran27,Reference Yung, Yuen, McGorry, Phillips, Kelly and Dell'Olio28
-
(b) a first episode of illness, defined as either first treatment contact (in-patient or out-patient) or duration of illness up to 5 years following illness onset; Reference Breitborde, Srihari and Woods29
-
(c) less than 2 weeks of antipsychotic treatment;
-
(d) a healthy control group;
-
(e) lipid profile assessment including one or more of the following measures: total cholesterol, HDL cholesterol, low-density lipoprotein (LDL) cholesterol, triglycerides, adiponectin, ghrelin, leptin, resistin, chemerin, omentin or apelin. Both fasting and non-fasting values were accepted, although all studies included in the analysis recorded fasting parameters.
Further information regarding rationale for the inclusion and exclusion criteria is provided in online supplement DS1. Studies in any language were considered. No restriction was made with regard to study design. Exclusion criteria were:
-
(a) study not reporting absolute values (i.e. studies that only provided data regarding the dichotomous presence or absence of raised lipid parameters as defined by diagnostic criteria); 7–10
-
(b) sample including patients with multiple episodes of schizophrenia;
-
(c) chronic antipsychotic treatment (more than 2 weeks' lifetime exposure);
-
(d) substance- or medication-induced psychotic disorder;
-
(e) physical comorbidity that might affect lipid homeostasis (e.g. familial hypercholesterolaemia, thyroid dysfunction, nephrotic syndrome);
-
(f) absence of measures in a healthy control group.
Data extraction
Screening based on title and abstract was performed independently by two authors (T.P. and K.B.). Where full texts, abstracts or group estimate data were not available, authors were contacted and articles or data requested. We allowed 4 weeks for authors to respond, with repeat contact attempts made after 2 weeks. Data extraction was performed independently (by T.P. and K.B.) and any disagreements resolved by rechecking original articles. Data were extracted according to the following model: author, year of publication, country, type of publication (i.e. prospective, cross-sectional, case–control, retrospective), matching criteria for patients and controls (confirmed by review of study methodology, or by confirmation of non-significance between mean parameter levels of patient and control groups); whether or not patient groups were antipsychotic-naïve (and if not, duration of treatment); and mean (with standard deviation) measure of lipid or adipocytokine parameter in patient and control groups. Where there were multiple publications for the same data-set, data were extracted from the study with the largest data-set. Study quality was assessed using the Newcastle-Ottawa Scale. Reference Stang30
Statistical analysis
Comprehensive Meta-Analysis software version 3.0 was used in all analyses. A two-tailed P value less than 0.05 was deemed significant. A random effects model was used in all analyses owing to an expectation of heterogeneity of data across studies. Reference Stroup, Berlin, Morton, Olkin, Williamson and Rennie23 Standardised mean differences in lipid parameters and adipocytokine levels between patient and control cohorts were used as the effect size, using Hedges' adjusted g and 95% confidence intervals. The direction of the effect size was positive if participants with schizophrenia demonstrated higher values of lipid parameters compared with controls. Where significant differences in lipid parameters were demonstrated, absolute differences were also calculated to allow clinical interpretation. Mean differences were calculated using SI units (mmol/L), which for certain studies required conversion from conventional units (mg/dL). To do so we used standardised conversion factors. Reference Kratz and Lewandrowski31 Heterogeneity across studies was assessed using Cochran's Q. Reference Bowden, Tierney, Copas and Burdett32 Inconsistency across studies was assessed with the I 2 statistic, Reference Higgins, Thompson, Deeks and Altman33 with a value below 25% deemed to have low heterogeneity, 25–75% medium heterogeneity and above 75% high heterogeneity. Publication bias and selective reporting were assessed using Egger's test of the intercept, Reference Egger, Zellweger-Zahner, Schneider, Junker, Lengeler and Antes34 although this was not calculated when fewer than ten studies were analysed, as recommended by the Cochrane Collaboration, and represented diagrammatically with funnel plots, again as recommended by the Cochrane Collaboration. Reference Higgins and Green35
The most common cause of dyslipidaemia is secondary to obesity and high fat intake, particularly saturated fat. Reference Durrington36 Age, gender and ethnicity are also recognised to influence lipid parameters. Reference Mozaffarian, Benjamin, Go, Arnett, Blaha and Cushman37 To address whether differences in body mass index (BMI), dietary intake, age, gender and ethnicity between patient and control groups influenced results, separate sensitivity analyses examining groups matched for these factors were performed. Matching was confirmed either by review of study method or by calculation of no significant difference between mean parameters in patient and control groups. To investigate further the influence of difference in BMI between patient and control groups on lipid parameter effect size, random effects meta-regression analyses were performed, regressing lipid parameter effect size on difference in BMI between patient and control groups. Meta-regression was not performed when fewer than ten studies were analysed, as recommended by the Cochrane Collaboration. Reference Higgins and Green35 To ensure that findings were not secondary to physical illness in one or both cohorts, sensitivity analyses were performed after removing studies that failed to document whether participants were excluded on the basis of poor physical health.
Results
Of 2070 citations retrieved, 20 case-control studies met inclusion criteria. Reference Chen, Du, Yin, Yang, Nie and Wang15–Reference Misiak, Laczmanski, Sloka, Szmida, Piotrowski and Loska20,Reference Petrikis, Tigas, Tzallas, Papadopoulos, Skapinakis and Mavreas38–Reference Srihari, Phutane, Ozkan, Chwastiak, Ratliff and Woods51 The search process is shown in Fig. 1 and the studies finally selected are summarised in Table 1. The overall sample comprised 1167 patients and 1184 controls. The mean age of patients was 27.9 years. Of the 1167 patients, 746 (63.9%) were antipsychotic-naïve whereas 421 (36.1%) had received antipsychotic medication for up to 14 days. There was no study of people with an at-risk mental state. The online data supplement details the raw data for analyses (Tables DS1–8); further information regarding cohort characteristics is given in Table DS9. Forest plots for HDL cholesterol and leptin, funnel plots and scatter plots for the individual lipid parameters are shown in Figs DS1–11.
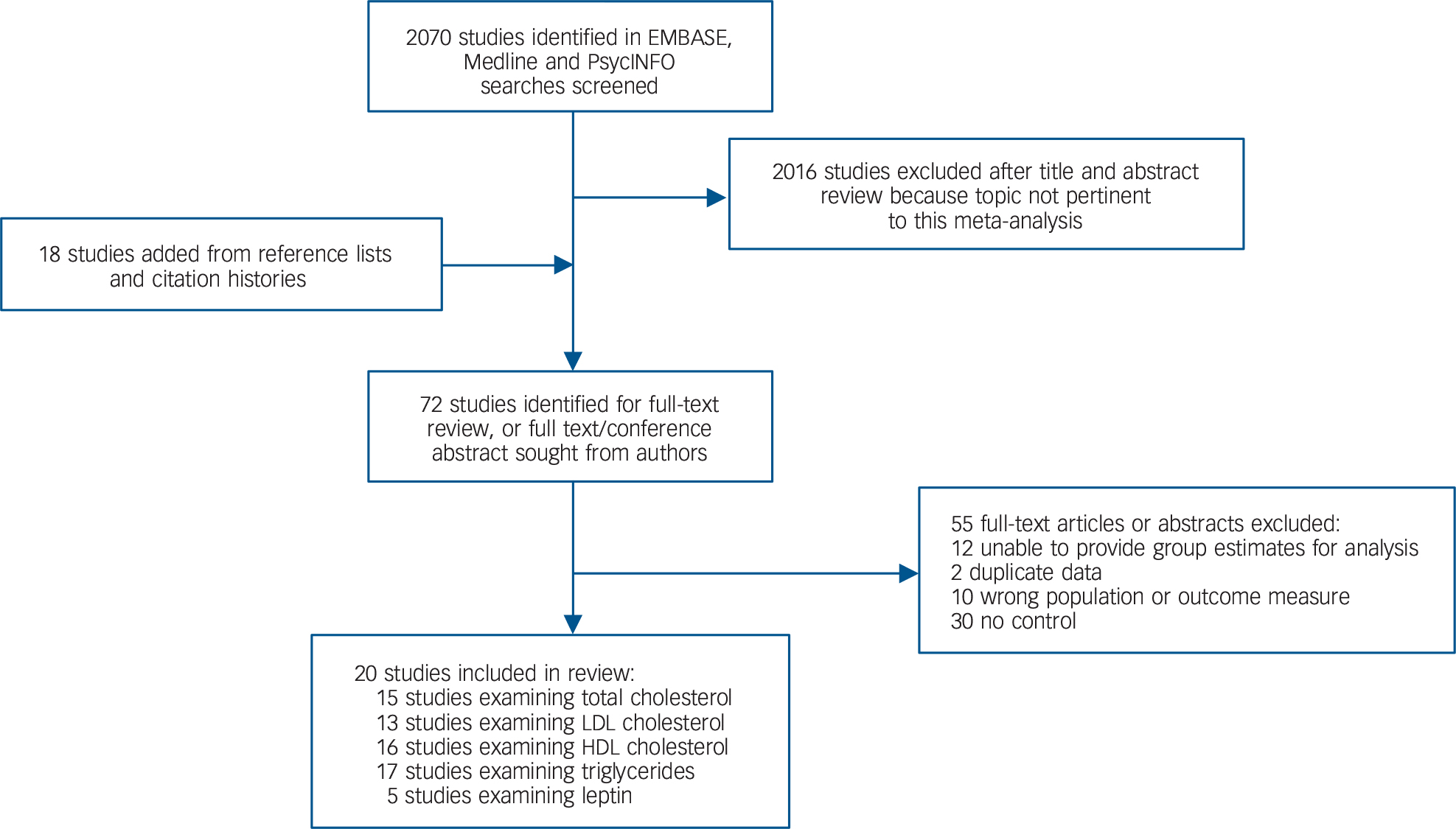
Fig. 1 Search process. HDL, high-density lipoprotein; LDL, low-density lipoprotein.
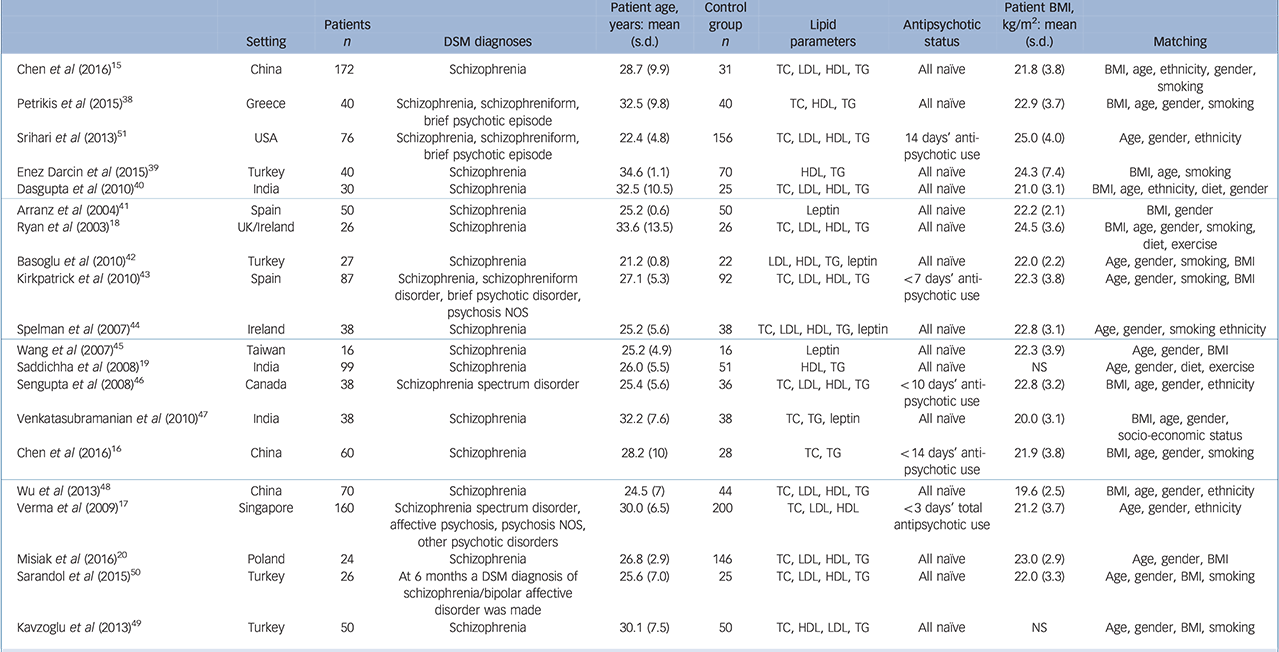
Table 1 Studies meeting inclusion criteria examining lipid and adipocytokine parameters in first-episode schizophrenia and related disorders
| Setting | Patients n |
DSM diagnoses | Patient age, years: mean (s.d.) |
Control group n |
Lipid parameters |
Antipsychotic status |
Patient BMI, kg/m2: mean (s.d) |
Matching | |
|---|---|---|---|---|---|---|---|---|---|
| Chen et al (2016) Reference Chen, Du, Yin, Yang, Nie and Wang15 | China | 172 | Schizophrenia | 28.7 (9.9) | 31 | TC, LDL, HDL, TG | All naïve | 21.8 (3.8) | BMI, age, ethnicity, gender, smoking |
| Petrikis et al (2015) Reference Petrikis, Tigas, Tzallas, Papadopoulos, Skapinakis and Mavreas38 | Greece | 40 | Schizophrenia, schizophreniform, brief psychotic episode |
32.5 (9.8) | 40 | TC, HDL, TG | All naïve | 22.9 (3.7) | BMI, age, gender, smoking |
| Srihari et al (2013) Reference Srihari, Phutane, Ozkan, Chwastiak, Ratliff and Woods51 | USA | 76 | Schizophrenia, schizophreniform, brief psychotic episode |
22.4 (4.8) | 156 | TC, LDL, HDL, TG | 14 days' antipsychotic use |
25.0 (4.0) | Age, gender, ethnicity |
| Enez Darcin et al (2015) Reference Enez Darcin, Yalcin Cavus, Dilbaz, Kaya and Dogan39 | Turkey | 40 | Schizophrenia | 34.6 (1.1) | 70 | HDL, TG | All naïve | 24.3 (7.4) | BMI, age, smoking |
| Dasgupta et al (2010) Reference Dasgupta, Singh, Rout, Saha and Mandal40 | India | 30 | Schizophrenia | 32.5 (10.5) | 25 | TC, LDL, HDL, TG | All naïve | 21.0 (3.1) | BMI, age, ethnicity, diet, gender |
| Arranz et al (2004) Reference Arranz, Rosel, Ramirez, Duenas, Fernandez and Sanchez41 | Spain | 50 | Schizophrenia | 25.2 (0.6) | 50 | Leptin | All naïve | 22.2 (2.1) | BMI, gender |
| Ryan et al (2003) Reference Ryan, Collins and Thakore18 | UK/Ireland | 26 | Schizophrenia | 33.6 (13.5) | 26 | TC, LDL, HDL, TG | All naïve | 24.5 (3.6) | BMI, age, gender, smoking, diet, exercise |
| Basoglu et al2010) Reference Basoglu, Oner, Gunes, Semiz, Ates and Algul42 | Turkey | 27 | Schizophrenia | 21.2 (0.8) | 22 | LDL, HDL, TG, leptin | All naïve | 22.0 (2.2) | Age, gender, smoking, BMI |
| Kirkpatrick et al (2010) Reference Kirkpatrick, Garcia-Rizo, Tang, Fernandez-Egea and Bernardo43 | Spain | 87 | Schizophrenia, schizophreniform disorder, brief psychotic disorder, psychosis NOS |
27.1 (5.3) | 92 | TC, LDL, HDL, TG | <7 days' antipsychotic use |
22.3 (3.8) | Age, gender, smoking, BMI |
| Spelman et al (2007) Reference Spelman, Walsh, Sharifi, Collins and Thakore44 | Ireland | 38 | Schizophrenia | 25.2 (5.6) | 38 | TC, LDL, HDL, TG, leptin | All naïve | 22.8 (3.1) | Age, gender, smoking ethnicity |
| Wang et al (2007) Reference Wang, Yang, Chen, Lee, Yeh and Lu45 | Taiwan | 16 | Schizophrenia | 25.2 (4.9) | 16 | Leptin | All naïve | 22.3 (3.9) | Age, gender, BMI |
| Saddichha et al (2008) Reference Saddichha, Manjunatha, Ameen and Akhtar19 | India | 99 | Schizophrenia | 26.0 (5.5) | 51 | HDL, TG | All naïve | NS | Age, gender, diet, exercise |
| Sengupta et al (2008) Reference Sengupta, Parrilla-Escobar, Klink, Fathalli, Ying Kin and Stip46 | Canada | 38 | Schizophrenia spectrum disorder | 25.4 (5.6) | 36 | TC, LDL, HDL, TG | <10 days' antipsychotic use |
22.8 (3.2) | BMI, age, gender, ethnicity |
| Venkatasubramanian et al (2010) Reference Venkatasubramanian, Chittiprol, Neelakantachar, Shetty and Gangadhar47 | India | 38 | Schizophrenia | 32.2 (7.6) | 38 | TC, TG, leptin | All naïve | 20.0 (3.1) | BMI, age, gender, socio-economic status |
| Chen et al (2016) Reference Chen, Broqueres-You, Yang, Wang, Li and Yang16 | China | 60 | Schizophrenia | 28.2 (10) | 28 | TC, TG | <14 days' antipsychotic use |
21.9(3.8) | BMI, age, gender, smoking |
| Wu et al (2013) Reference Wu, Huang, Wu, Zhong, Wei and Wang48 | China | 70 | Schizophrenia | 24.5 (7) | 44 | TC, LDL, HDL, TG | All naïve | 19.6 (2.5) | BMI, age, gender, ethnicity |
| Verma et al (2009) Reference Verma, Subramaniam, Liew and Poon17 | Singapore | 160 | Schizophrenia spectrum disorder, affective psychosis, psychosis NOS, other psychotic disorders |
30.0 (6.5) | 200 | TC, LDL, HDL | <3 days' total antipsychotic use |
21.2 (3.7) | Age, gender, ethnicity |
| Misiak et al (2016) Reference Misiak, Laczmanski, Sloka, Szmida, Piotrowski and Loska20 | Poland | 24 | Schizophrenia | 26.8 (2.9) | 146 | TC, LDL, HDL, TG | All naïve | 23.0 (2.9) | Age, gender, BMI |
| Sarandol et al (2015) Reference Sarandol, Sarandol, Acikgoz, Eker, Akkaya and Dirican50 | Turkey | 26 | At 6 months a DSM diagnosis of schizophrenia/bipolar affective disorder was made |
25.6 (7.0) | 25 | TC, LDL, HDL, TG | All naïve | 22.0 (3.3) | Age, gender, BMI, smoking |
| Kavzoglu et al (2013) Reference Kavzoglu and Hariri49 | Turkey | 50 | Schizophrenia | 30.1 (7.5) | 50 | TC, HDL, LDL, TG | All naïve | NS | Age, gender, BMI, smoking |
BMI, body mass index; HDL, high-density lipoprotein cholesterol; LDL, low-density lipoprotein cholesterol; NOS, not otherwise specifed. TC, total cholesterol; TG, triglyceride.
Total cholesterol
Total cholesterol concentration was analysed using data for 15 studies, comprising 927 patients and 959 controls. Reference Chen, Du, Yin, Yang, Nie and Wang15–Reference Ryan, Collins and Thakore18,Reference Misiak, Laczmanski, Sloka, Szmida, Piotrowski and Loska20,Reference Petrikis, Tigas, Tzallas, Papadopoulos, Skapinakis and Mavreas38,Reference Dasgupta, Singh, Rout, Saha and Mandal40,Reference Kirkpatrick, Garcia-Rizo, Tang, Fernandez-Egea and Bernardo43,Reference Spelman, Walsh, Sharifi, Collins and Thakore44,Reference Sengupta, Parrilla-Escobar, Klink, Fathalli, Ying Kin and Stip46–Reference Srihari, Phutane, Ozkan, Chwastiak, Ratliff and Woods51 Total cholesterol levels were significantly decreased in patients compared with controls: g = −0.19, 95% CI −0.32 to −0.06; P = 0.005 (Fig. 2). This corresponds to an absolute decrease in total cholesterol of 0.26 mmol/L. There was significant between-sample heterogeneity, with an I 2 of 42% (Q = 24.2, P = 0.04). Findings of Egger's test (P = 0.54) suggested that publication bias was not significant, and visual inspection of the funnel plot suggested symmetry (online Fig. DS3). After restricting analyses to 12 BMI-matched studies, Reference Chen, Du, Yin, Yang, Nie and Wang15,Reference Chen, Broqueres-You, Yang, Wang, Li and Yang16,Reference Ryan, Collins and Thakore18,Reference Misiak, Laczmanski, Sloka, Szmida, Piotrowski and Loska20,Reference Petrikis, Tigas, Tzallas, Papadopoulos, Skapinakis and Mavreas38,Reference Dasgupta, Singh, Rout, Saha and Mandal40,Reference Kirkpatrick, Garcia-Rizo, Tang, Fernandez-Egea and Bernardo43,Reference Sengupta, Parrilla-Escobar, Klink, Fathalli, Ying Kin and Stip46–Reference Sarandol, Sarandol, Acikgoz, Eker, Akkaya and Dirican50 total cholesterol concentration remained significantly decreased in patients (n = 813) compared with controls (n = 765); g = −0.21, 95% CI −0.36 to −0.06; P < 0.01. Meta-regression of effect size for total cholesterol on absolute difference in BMI between patient and control groups (scatter plot shown in online Fig. DS8) revealed that BMI difference between the two cohorts was not a significant moderator of the total cholesterol effect size (β = 0.05, 95% CI −0.30 to 0.40; P = 0.76). There were insufficient studies to perform a sensitivity analysis where groups were matched for dietary intake. After restricting analyses to seven studies matched for ethnicity, Reference Chen, Du, Yin, Yang, Nie and Wang15,Reference Verma, Subramaniam, Liew and Poon17,Reference Ryan, Collins and Thakore18,Reference Dasgupta, Singh, Rout, Saha and Mandal40,Reference Spelman, Walsh, Sharifi, Collins and Thakore44,Reference Sengupta, Parrilla-Escobar, Klink, Fathalli, Ying Kin and Stip46,Reference Srihari, Phutane, Ozkan, Chwastiak, Ratliff and Woods51 there was no longer a significant difference in total cholesterol between patients (n = 540) and controls (n = 512): g = −0.21, 95% CI −0.42 to 0.00; P = 0.05. After exclusion of two studies in which participants were not specifically documented as being physically healthy, Reference Verma, Subramaniam, Liew and Poon17,Reference Srihari, Phutane, Ozkan, Chwastiak, Ratliff and Woods51 total cholesterol concentration remained significantly decreased in patients (n = 691) and controls (n = 603): g = −0.17, 95% CI −0.32 to −0.02; P = 0.02. All studies involved in the total cholesterol analysis were matched for age and gender and therefore these sensitivity analyses were not required.
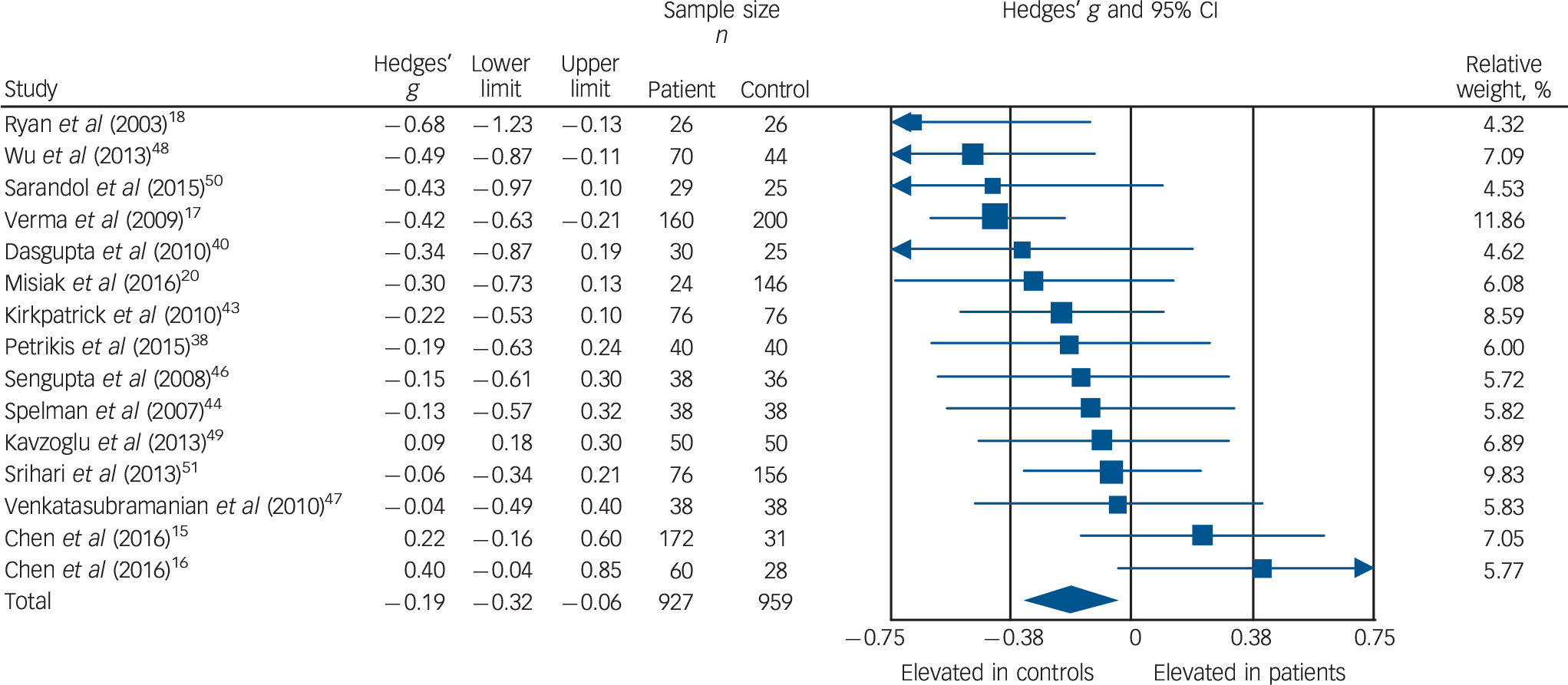
Fig. 2 Forest plot showing total cholesterol concentration in patients with first-episode psychosis and controls. There was a significant reduction in total cholesterol concentration in patients (Hedges' g = −0.19, 95% CI −0.32 to −0.06; P = 0.005).
LDL cholesterol
Low-density lipoprotein cholesterol concentration was analysed using data from 13 studies, comprising 806 patients and 875 controls. Reference Chen, Du, Yin, Yang, Nie and Wang15,Reference Verma, Subramaniam, Liew and Poon17,Reference Ryan, Collins and Thakore18,Reference Misiak, Laczmanski, Sloka, Szmida, Piotrowski and Loska20,Reference Dasgupta, Singh, Rout, Saha and Mandal40,Reference Basoglu, Oner, Gunes, Semiz, Ates and Algul42–Reference Spelman, Walsh, Sharifi, Collins and Thakore44,Reference Sengupta, Parrilla-Escobar, Klink, Fathalli, Ying Kin and Stip46,Reference Wu, Huang, Wu, Zhong, Wei and Wang48–Reference Srihari, Phutane, Ozkan, Chwastiak, Ratliff and Woods51 Concentration was significantly decreased in patients compared with controls: g = −0.22, 95% CI −0.35 to −0.09; P = 0.001 (Fig. 3). This corresponds to an absolute decrease in LDL cholesterol of 0.15 mmol/L. There was no statistically significant between-sample heterogeneity (I 2 = 29%; Q = 16.8, P = 0.16). Findings of Egger's test (P = 0.13) suggested that publication bias was not significant, and visual inspection of the funnel plot suggested symmetry (online Fig. DS4). Restricting analyses to ten BMI-matched studies demonstrated that LDL cholesterol remained significantly decreased in patients (n = 532) compared with controls (n = 481): g = −0.18, 95% CI −0.31 to −0.04; P = 0.012. Reference Chen, Du, Yin, Yang, Nie and Wang15,Reference Ryan, Collins and Thakore18,Reference Misiak, Laczmanski, Sloka, Szmida, Piotrowski and Loska20,Reference Dasgupta, Singh, Rout, Saha and Mandal40,Reference Basoglu, Oner, Gunes, Semiz, Ates and Algul42,Reference Kirkpatrick, Garcia-Rizo, Tang, Fernandez-Egea and Bernardo43,Reference Sengupta, Parrilla-Escobar, Klink, Fathalli, Ying Kin and Stip46,Reference Wu, Huang, Wu, Zhong, Wei and Wang48–Reference Sarandol, Sarandol, Acikgoz, Eker, Akkaya and Dirican50 Meta-regression of effect size for LDL cholesterol on absolute difference in BMI between patient and control groups (scatter plot shown in online Fig. DS9) revealed that BMI difference between the two cohorts was not a significant moderator of the LDL cholesterol effect size (β = −0.15, 95% CI −0.49 to 0.18; P = 0.38). There were insufficient studies to perform a sensitivity analysis where groups were matched for dietary intake. After restricting analyses to seven studies matched for ethnicity, Reference Chen, Du, Yin, Yang, Nie and Wang15,Reference Verma, Subramaniam, Liew and Poon17,Reference Ryan, Collins and Thakore18,Reference Dasgupta, Singh, Rout, Saha and Mandal40,Reference Spelman, Walsh, Sharifi, Collins and Thakore44,Reference Sengupta, Parrilla-Escobar, Klink, Fathalli, Ying Kin and Stip46,Reference Srihari, Phutane, Ozkan, Chwastiak, Ratliff and Woods51 we found LDL cholesterol remained significantly decreased in patients (n = 540) compared with controls (n = 512): g = −0.29, 95% CI −0.46 to −0.11; P < 0.01. After exclusion of two studies in which participants were not specifically documented as being physically healthy, Reference Verma, Subramaniam, Liew and Poon17,Reference Srihari, Phutane, Ozkan, Chwastiak, Ratliff and Woods51 LDL cholesterol remained significantly decreased in patients (n = 570) compared with controls (n = 519): g = −0.15, 95% CI −0.28 to −0.02; P = 0.03. All studies involved in the LDL cholesterol analysis were matched for age and gender, so these sensitivity analyses were not required.
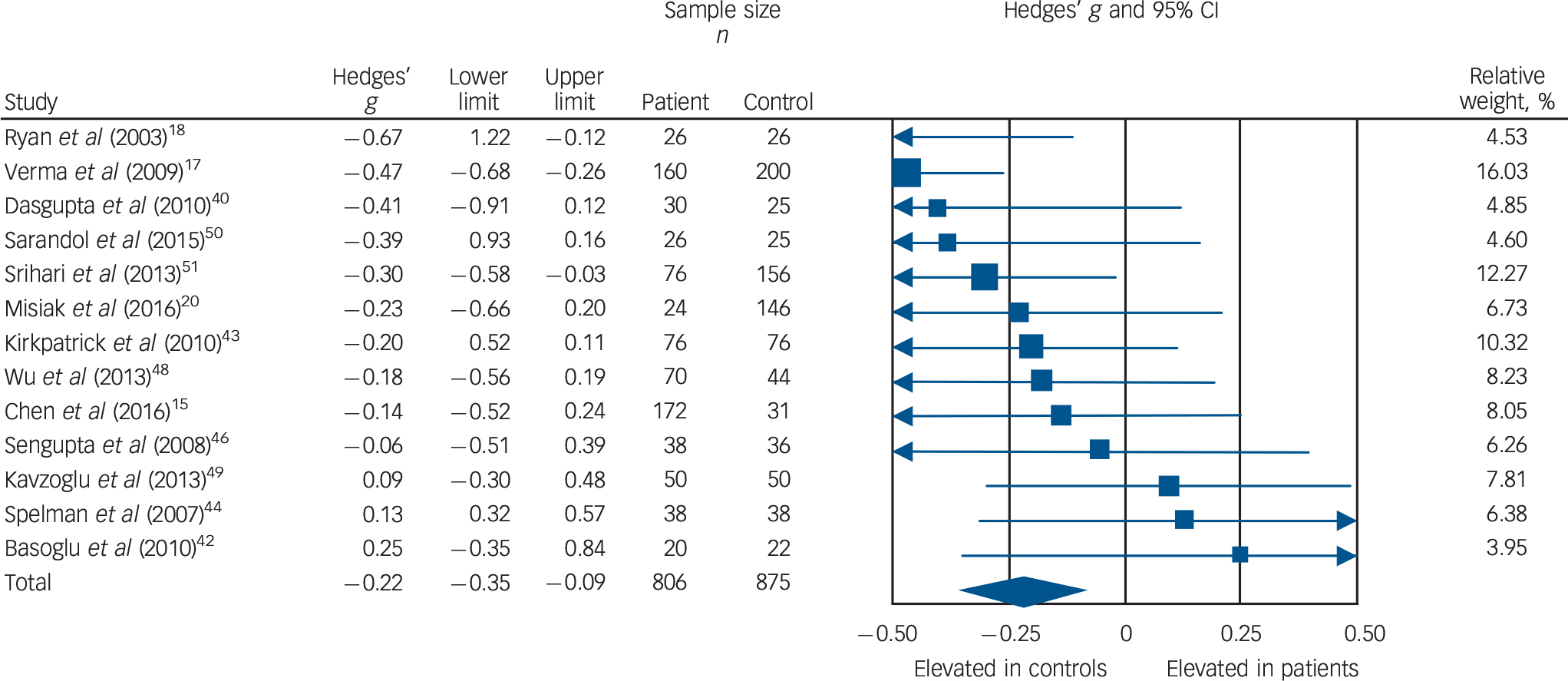
Fig. 3 Forest plot showing low-density lipoprotein (LDL) cholesterol concentration in patients with first-episode psychosis and controls. There was a significant reduction in LDL cholesterol concentration in patients (Hedges' g = −0.22, 95% CI −0.35 to −0.09; P = 0.001).
Triglycerides
Triglyceride concentration was analysed using data from 17 studies, comprising 923 patients and 902 controls. Reference Chen, Du, Yin, Yang, Nie and Wang15,Reference Chen, Broqueres-You, Yang, Wang, Li and Yang16,Reference Ryan, Collins and Thakore18–Reference Misiak, Laczmanski, Sloka, Szmida, Piotrowski and Loska20,Reference Petrikis, Tigas, Tzallas, Papadopoulos, Skapinakis and Mavreas38–Reference Dasgupta, Singh, Rout, Saha and Mandal40,Reference Basoglu, Oner, Gunes, Semiz, Ates and Algul42–Reference Spelman, Walsh, Sharifi, Collins and Thakore44,Reference Sengupta, Parrilla-Escobar, Klink, Fathalli, Ying Kin and Stip46–Reference Srihari, Phutane, Ozkan, Chwastiak, Ratliff and Woods51 Concentration was significantly elevated in patients compared with controls: g = 0.14, 95% CI 0.00–0.28; P < 0.05 (Fig. 4). This corresponds to an absolute increase in triglyceride levels of 0.08 mmol/L. There was significant between-sample heterogeneity (I 2 = 48%; Q = 30.6, P = 0.02). Findings of Egger's test (P = 0.28) suggested that publication bias was not significant, and visual inspection of the funnel plot suggested symmetry. After restricting analyses to 14 BMI-matched studies there was no longer a significant difference in triglyceride concentrations between patients (n = 710) and controls (n = 657): g = 0.13, 95% CI −0.02 to 0.27; P = 0.09. Reference Chen, Du, Yin, Yang, Nie and Wang15,Reference Chen, Broqueres-You, Yang, Wang, Li and Yang16,Reference Ryan, Collins and Thakore18,Reference Misiak, Laczmanski, Sloka, Szmida, Piotrowski and Loska20,Reference Petrikis, Tigas, Tzallas, Papadopoulos, Skapinakis and Mavreas38–Reference Dasgupta, Singh, Rout, Saha and Mandal40,Reference Basoglu, Oner, Gunes, Semiz, Ates and Algul42,Reference Kirkpatrick, Garcia-Rizo, Tang, Fernandez-Egea and Bernardo43,Reference Sengupta, Parrilla-Escobar, Klink, Fathalli, Ying Kin and Stip46–Reference Sarandol, Sarandol, Acikgoz, Eker, Akkaya and Dirican50 Meta-regression of effect size for triglycerides on absolute difference in BMI between patient and control groups (online Fig. DS10) revealed that BMI difference between the two cohorts was a significant moderator of the triglyceride effect size (β = 0.29, 95% CI 0.04–0.53; P = 0.024). After restricting analyses to three studies matched for dietary intake, Reference Ryan, Collins and Thakore18,Reference Saddichha, Manjunatha, Ameen and Akhtar19,Reference Dasgupta, Singh, Rout, Saha and Mandal40 triglyceride concentration remained significantly elevated in patients (n = 155) compared with controls (n = 102): g = 0.34, 95% CI 0.09–0.59; P < 0.01. Exclusion of one study that failed to match for gender revealed that triglyceride concentration remained significantly elevated in patients (n = 883) compared with controls (n = 832): g = 0.15, 95% CI 0.00 to 0.30; P < 0.05. Reference Enez Darcin, Yalcin Cavus, Dilbaz, Kaya and Dogan39 After restricting analyses to six studies matched for ethnicity, Reference Chen, Du, Yin, Yang, Nie and Wang15,Reference Ryan, Collins and Thakore18,Reference Dasgupta, Singh, Rout, Saha and Mandal40,Reference Spelman, Walsh, Sharifi, Collins and Thakore44,Reference Sengupta, Parrilla-Escobar, Klink, Fathalli, Ying Kin and Stip46,Reference Srihari, Phutane, Ozkan, Chwastiak, Ratliff and Woods51 there was no longer a significant difference in total cholesterol concentration between patients (n = 380) and controls (n = 312): g = 0.06, 95% CI −0.12 to 0.22; P = 0.49. After exclusion of one study in which participants were not specifically documented as being physically healthy, Reference Srihari, Phutane, Ozkan, Chwastiak, Ratliff and Woods51 triglyceride concentration remained elevated in patients (n = 847) compared with controls (n = 746): g = 0.17, 95% CI 0.03 to 0.31; P = 0.02. All studies involved in the triglyceride analysis were matched for age so this sensitivity analysis was not required.
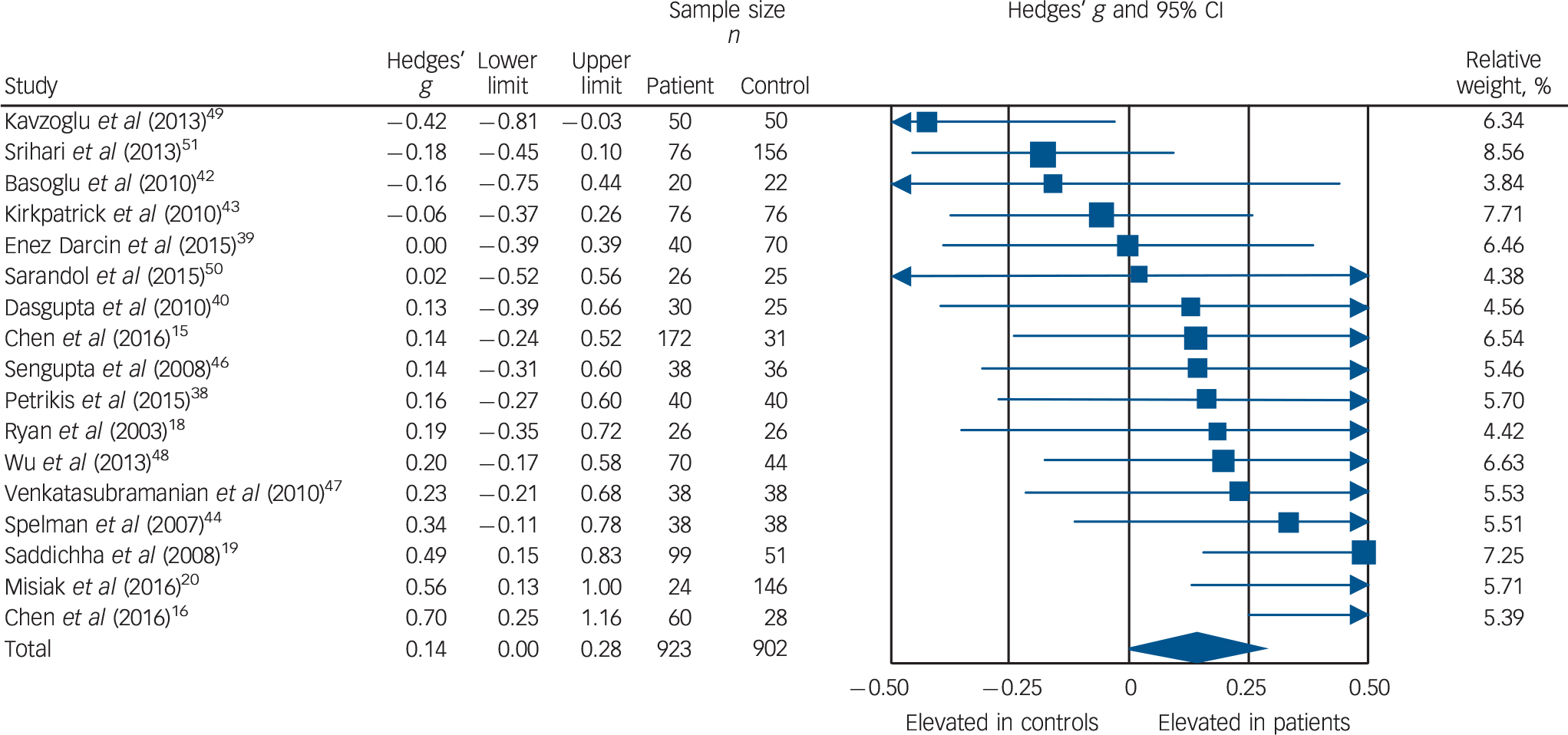
Fig. 4 Forest plot showing triglyceride concentration in patients with first-episode psychosis and controls. There was an elevation in triglyceride concentration in patients (Hedges' g = 0.14, 95% CI 0.00–0.28; P < 0.05).
HDL cholesterol
High-density lipoprotein concentration was analysed using data from 16 studies, comprising 985 patients and 1036 controls. Reference Chen, Du, Yin, Yang, Nie and Wang15,Reference Verma, Subramaniam, Liew and Poon17–Reference Misiak, Laczmanski, Sloka, Szmida, Piotrowski and Loska20,Reference Petrikis, Tigas, Tzallas, Papadopoulos, Skapinakis and Mavreas38–Reference Dasgupta, Singh, Rout, Saha and Mandal40,Reference Basoglu, Oner, Gunes, Semiz, Ates and Algul42–Reference Spelman, Walsh, Sharifi, Collins and Thakore44,Reference Sengupta, Parrilla-Escobar, Klink, Fathalli, Ying Kin and Stip46,Reference Wu, Huang, Wu, Zhong, Wei and Wang48–Reference Srihari, Phutane, Ozkan, Chwastiak, Ratliff and Woods51 Concentrations were not altered in patients compared with controls: g = −0.19, 95% CI −0.39 to 0.02; P = 0.07 (online Fig. DS1). There was statistically significant between-sample heterogeneity (I 2 = 77%; Q = 66.2, P < 0.01). Findings of Egger's test (P = 0.67) suggested that publication bias was not significant, and visual inspection of the funnel plot suggested symmetry – although we noted a single outlying study with a large standard error, Reference Basoglu, Oner, Gunes, Semiz, Ates and Algul42 probably the consequence of a small sample size. After analyses were restricted to 12 BMI-matched studies, Reference Chen, Du, Yin, Yang, Nie and Wang15,Reference Ryan, Collins and Thakore18,Reference Misiak, Laczmanski, Sloka, Szmida, Piotrowski and Loska20,Reference Petrikis, Tigas, Tzallas, Papadopoulos, Skapinakis and Mavreas38–Reference Dasgupta, Singh, Rout, Saha and Mandal40,Reference Basoglu, Oner, Gunes, Semiz, Ates and Algul42,Reference Kirkpatrick, Garcia-Rizo, Tang, Fernandez-Egea and Bernardo43,Reference Sengupta, Parrilla-Escobar, Klink, Fathalli, Ying Kin and Stip46,Reference Wu, Huang, Wu, Zhong, Wei and Wang48–Reference Sarandol, Sarandol, Acikgoz, Eker, Akkaya and Dirican50 no significant difference remained in HDL cholesterol concentration between patients (n = 612) and controls (n = 591): g = −0.23, 95% CI −0.53 to 0.06; P = 0.12. Meta-regression of effect size for HDL cholesterol on difference in BMI between patient and control groups (scatter plot shown in online Fig. DS11) revealed that BMI difference between the two cohorts was not a significant moderator of the HDL cholesterol effect size (β = −0.10, 95% CI −0.85 to 0.65; P = 0.79). After restricting analyses to three studies matched for dietary intake, Reference Ryan, Collins and Thakore18,Reference Saddichha, Manjunatha, Ameen and Akhtar19,Reference Dasgupta, Singh, Rout, Saha and Mandal40 no significant difference remained in HDL cholesterol concentration between patients (n = 155) and controls (n = 102): g = −0.04, 95% CI −0.28 to 0.782; P = 0.78. Exclusion of one study that failed to match for gender revealed that HDL cholesterol remained unaltered between patients (n = 945) and controls (n = 966): g = −0.16, 95% CI −0.37 to 0.05; P = 0.12. Reference Enez Darcin, Yalcin Cavus, Dilbaz, Kaya and Dogan39 After restricting analyses to seven studies matched for ethnicity, Reference Chen, Du, Yin, Yang, Nie and Wang15,Reference Verma, Subramaniam, Liew and Poon17,Reference Ryan, Collins and Thakore18,Reference Dasgupta, Singh, Rout, Saha and Mandal40,Reference Spelman, Walsh, Sharifi, Collins and Thakore44,Reference Sengupta, Parrilla-Escobar, Klink, Fathalli, Ying Kin and Stip46,Reference Srihari, Phutane, Ozkan, Chwastiak, Ratliff and Woods51 no significant difference remained in HDL concentration between patients (n = 540) and controls (n = 512): g = −0.09, 95% CI −0.22 to 0.04; P = 0.16. After exclusion of two studies in which participants were not specifically documented as being physically healthy, Reference Verma, Subramaniam, Liew and Poon17,Reference Srihari, Phutane, Ozkan, Chwastiak, Ratliff and Woods51 HDL cholesterol levels remained unaltered between patients (n = 749) and controls (n = 680): g = −0.21, 95% CI −0.46 to 0.04; P = 0.10. All studies involved in HDL cholesterol analysis were matched for age so this sensitivity analysis was not performed.
Leptin
Leptin concentration in patients and controls was analysed using data from five studies, comprising 162 patients and 164 controls. Reference Arranz, Rosel, Ramirez, Duenas, Fernandez and Sanchez41,Reference Basoglu, Oner, Gunes, Semiz, Ates and Algul42,Reference Spelman, Walsh, Sharifi, Collins and Thakore44,Reference Wang, Yang, Chen, Lee, Yeh and Lu45,Reference Venkatasubramanian, Chittiprol, Neelakantachar, Shetty and Gangadhar47 Leptin concentration was not altered in patients compared with controls: g = 0.05, 95% CI −0.31 to 0.42; P = 0.78 (online Fig. DS2). Between-sample heterogeneity was medium (I 2 = 59%; Q = 12.3, P = 0.03). Visual inspection of the funnel plot suggested asymmetry, although this interpretation should be taken with caution in the context of the small number of studies, and the single study driving asymmetry exhibiting a large standard error, Reference Wang, Yang, Chen, Lee, Yeh and Lu45 probably the consequence of small sample size (n = 7). Egger's test of the intercept was not possible owing to the small number of studies. After restricting analyses to BMI-matched studies, Reference Arranz, Rosel, Ramirez, Duenas, Fernandez and Sanchez41,Reference Basoglu, Oner, Gunes, Semiz, Ates and Algul42,Reference Wang, Yang, Chen, Lee, Yeh and Lu45,Reference Venkatasubramanian, Chittiprol, Neelakantachar, Shetty and Gangadhar47 there remained no significant difference in leptin concentration between patients (n = 124) and controls (n = 126): g = −0.07, 95% CI −0.32 to 0.17; P = 0.56. All studies involved in leptin analysis matched for age so this sensitivity analysis was not required. Exclusion of one study that failed to match for age revealed that leptin concentration remained unaltered between patients (n = 112) and controls (n = 114): g = 0.14, 95% CI −0.35 to 0.63; P = 0.57. Reference Arranz, Rosel, Ramirez, Duenas, Fernandez and Sanchez41 There were insufficient studies to perform sensitivity analyses where groups were matched for dietary intake or ethnicity.
Discussion
The main findings of this meta-analysis are that first-episode psychosis is associated with decreased total and LDL cholesterol levels but increased triglyceride levels compared with healthy control groups, with no difference in HDL cholesterol or leptin levels. The absolute difference in lipid parameters demonstrated in FEP compared with controls was a decrease in total cholesterol of 0.26 mmol/L (upper limit for total cholesterol levels in adults 5.00 mmol/L), Reference Perk, De Backer, Gohlke, Graham, Reiner and Verschuren52 a decrease in LDL cholesterol of 0.15 mmol/L (upper limit for LDL cholesterol levels in adults 3.00 mmol/L), Reference Perk, De Backer, Gohlke, Graham, Reiner and Verschuren52 and an increase in triglycerides of 0.08 mmol/L (upper limit for triglyceride levels in adults 1.70 mmol/L). Reference Perk, De Backer, Gohlke, Graham, Reiner and Verschuren52 Data from studies focusing on the efficacy of statin therapy in individuals at low risk of cardiovascular disease demonstrate that an absolute reduction in LDL cholesterol of 1 mmol/L is associated with a relative risk reduction of major vascular events of 21%, Reference Mihaylova, Emberson, Blackwell, Keech and Simes53 so a reduction in LDL cholesterol of 0.15 mmol/L seen in FEP may be associated with an approximate 3% risk reduction in cardiovascular disease. Total and LDL cholesterol findings remained significant in the sensitivity analyses, except for the one for ethnicity, where total cholesterol difference was no longer significant. Specifically, total and LDL cholesterol findings remained significant in BMI-matched analyses, and meta-regression revealed that BMI difference between the two cohorts was not a significant moderator for either finding. The triglyceride findings were no longer significant in sensitivity analyses that matched for BMI and ethnicity, and meta-regression revealed that BMI difference between the two cohorts was a significant moderator. Leptin concentrations were not altered in patients relative to controls, although this result should be interpreted with caution owing to the small sample size used in this analysis.
Our findings extend the meta-analysis by Perry et al, Reference Perry, Mcintosh, Weich, Singh and Rees21 by using a larger sample (2351 participants compared with 1137 in Perry's paper) and including additional lipidomic measures and sensitivity analyses. Our finding of reduced total cholesterol is consistent with their finding, but in addition we show a significant reduction in LDL cholesterol and increase in triglyceride levels, which Perry et al did not find, potentially because of lack of power in their study. Our findings contrast with evidence of elevated total and LDL cholesterol in chronic schizophrenia. Reference Vancampfort, Wampers, Mitchell, Correll, De Herdt and Probst14 Our findings also contrast with recent evidence of broader metabolic dysfunction in FEP, specifically glucose dysregulation. Reference Perry, Mcintosh, Weich, Singh and Rees21,Reference Pillinger, Beck, Gobjila, Donocik, Jauhar and Howes25,Reference Greenhalgh, Gonzalez-Bianco, Garcia-Rizo, Fernandez-Egea, Miller and Arroyo54 However, our demonstration of raised triglyceride concentrations in FEP is compatible with the notion of FEP being associated with impairments in glucose homeostasis, with hypertriglyceridaemia recognised as accompanying development of type 2 diabetes mellitus. Reference Lin, Berlin, Younge, Jin, Sibley and Schreiner55 Indeed, in apparently healthy men aged 26–45 years the hazard ratio for developing type 2 diabetes in those with high triglyceride levels (1.85–3.38 mmol/L) compared with low triglyceride levels (0.34–0.75 mmol/L) is 2.11. Reference Tirosh, Shai, Bitzur, Kochba, Tekes-Manova and Israeli56
Strengths and limitations
Strengths of this analysis include the focus on patients with FEP and minimal or no antipsychotic exposure, thus limiting the duration of secondary illness-related factors that may result in dyslipidaemia. Nevertheless, population studies and meta-analyses have previously demonstrated that individuals with FEP already have poorer dietary habits, decreased physical activity and an increased likelihood of smoking compared with age-matched controls. Reference Stubbs, Firth, Berry, Schuch, Rosenbaum and Gaughran57–Reference Myles, Newall, Compton, Curtis, Nielssen and Large60 In line with clinical practice, Reference Breitborde, Srihari and Woods29 our definition of FEP included patients with several years of illness, and duration of untreated psychosis was not documented in all studies; thus there were insufficient studies to perform meta-regression analyses examining the impact of illness duration. Therefore, some patients might already have been exposed to lifestyle risk factors for some time. However, prolonged duration of poor lifestyle habits would not be expected to result in the total and LDL cholesterol reductions we observed. Similarly, the limitation that a third of the analysed patient cohort had been prescribed up to 14 days of antipsychotic medication might be expected to be associated with increased total and LDL cholesterol levels. This suggests that the reductions in cholesterol levels observed might be larger when the impact of poor lifestyle habits and medication are controlled for. Matching for diet was performed in only three studies, Reference Ryan, Collins and Thakore18,Reference Saddichha, Manjunatha, Ameen and Akhtar19,Reference Dasgupta, Singh, Rout, Saha and Mandal40 and a sensitivity analysis examining dietary impact could be performed only for triglycerides (which maintained significance for raised triglyceride levels in patients compared with controls). Similarly, only two studies matched for the amount of regular exercise taken by participants. Reference Ryan, Collins and Thakore18,Reference Saddichha, Manjunatha, Ameen and Akhtar19 High fat intake and obesity are the most common causes of dyslipidaemia. Reference Durrington36 Exercise is associated with reduced lipid levels, potentially through enhancing skeletal muscle lipid metabolism preferentially over glycogen. Reference Earnest, Artero, Sui, Lee, Church and Blair61 We cannot therefore exclude dietary or exercise differences as contributing to our findings, although poor lifestyle factors would be expected to oppose the effects we observed, not inflate them. Moreover, overmatching of patients and controls for lifestyle factors could result in samples poorly representative of populations. Data from large-scale, population-based studies and registries have established that plasma lipids and lipoproteins change modestly 1–6 h following habitual food intake (compared with fasting levels, non-fasting triglyceride levels increase by up to 0.3 mmol/L and non-fasting total and LDL cholesterol levels decrease by up to 0.2 mmol/L, probably owing to haemodilution due to fluid intake with a meal). Reference Langsted, Freiberg and Nordestgaard62 In view of this we extracted study information regarding duration of fasting (online Table DS10). Sensitivity analyses excluding studies with unclear fasting duration showed that the findings of reduced total and LDL cholesterol levels in FEP compared with controls remained significant, Reference Chen, Broqueres-You, Yang, Wang, Li and Yang16,Reference Saddichha, Manjunatha, Ameen and Akhtar19,Reference Arranz, Rosel, Ramirez, Duenas, Fernandez and Sanchez41,Reference Kirkpatrick, Garcia-Rizo, Tang, Fernandez-Egea and Bernardo43,Reference Wang, Yang, Chen, Lee, Yeh and Lu45,Reference Sarandol, Sarandol, Acikgoz, Eker, Akkaya and Dirican50 and also confirmed the findings of no significant difference in HDL cholesterol or leptin levels between patients and controls. However, triglyceride levels were no longer elevated in patients compared with controls (results in online supplement DS1). Notwithstanding these analyses, we cannot exclude the possibility that the patients were less compliant with the fast. This warrants further investigation in future studies.
Another potential limitation of the meta-analysis is the inclusion of participants who might not have been representative of FEP and control populations in general. However, all studies included in the meta-analysis used DSM-IV criteria for selection of patients, and all studies were deemed to be adequate for case selection as part of the Newcastle-Ottawa Scale quality assessment (online Table DS10). We can therefore be confident that the sampled patient group was adequately representative of the patient population. However, we note that eight studies used hospital and university staff or relatives of staff as controls; Reference Verma, Subramaniam, Liew and Poon17,Reference Ryan, Collins and Thakore18,Reference Petrikis, Tigas, Tzallas, Papadopoulos, Skapinakis and Mavreas38,Reference Enez Darcin, Yalcin Cavus, Dilbaz, Kaya and Dogan39,Reference Arranz, Rosel, Ramirez, Duenas, Fernandez and Sanchez41,Reference Sengupta, Parrilla-Escobar, Klink, Fathalli, Ying Kin and Stip46,Reference Wu, Huang, Wu, Zhong, Wei and Wang48,Reference Sarandol, Sarandol, Acikgoz, Eker, Akkaya and Dirican50 these individuals may be poorly representative of the socioeconomic background of population controls. However, higher socioeconomic status is associated with reduced cardiovascular risk, for example total cholesterol is inversely associated with educational level. Reference Benetou, Chloptsios, Zavitsanos, Karalis, Naska and Trichopoulou63 This would therefore not explain our findings of reduced total and LDL cholesterol in FEP. Four studies failed to detail how controls were recruited; Reference Chen, Broqueres-You, Yang, Wang, Li and Yang16,Reference Misiak, Laczmanski, Sloka, Szmida, Piotrowski and Loska20,Reference Venkatasubramanian, Chittiprol, Neelakantachar, Shetty and Gangadhar47,Reference Kavzoglu and Hariri49 however, sensitivity analyses excluding these four studies did not alter results (online supplement DS1).
Sensitivity analyses that matched for ethnicity resulted in a loss of significance for both total cholesterol and triglyceride findings, which may point to ethnic differences between patients and controls contributing to results. Moreover, a BMI-matched sensitivity analysis for triglycerides resulted in loss of significance for the finding of raised triglycerides in FEP, and meta-regression revealed that BMI difference between patient and control groups was a significant moderator of the triglyceride effect size. Obesity is a major cause of dyslipidaemia and lipid levels are directly related to BMI. Reference Durrington36 Thus, group differences in BMI could have contributed to observed triglyceride differences, although overall patient groups had lower BMI levels compared with controls (average difference 0.39 kg/m2). However, BMI-matched sensitivity analyses for total and LDL cholesterol remained significant, and meta-regression demonstrated that BMI difference between the two groups did not moderate observed effect sizes. Moreover, poor lifestyle habits in the patient cohort would be expected to increase total and LDL cholesterol levels, thus are unlikely to explain the reduction in cholesterol parameters we observed.
Although participants in the meta-analysis were described as physically healthy with no illness that would affect lipid parameters, only seven studies referred to the use of over-the-counter medication as a specific exclusion criterion, Reference Ryan, Collins and Thakore18,Reference Petrikis, Tigas, Tzallas, Papadopoulos, Skapinakis and Mavreas38–Reference Arranz, Rosel, Ramirez, Duenas, Fernandez and Sanchez41,Reference Kirkpatrick, Garcia-Rizo, Tang, Fernandez-Egea and Bernardo43,Reference Spelman, Walsh, Sharifi, Collins and Thakore44 and only six studies defined psychotropic use other than antipsychotics as an exclusion criterion; Reference Chen, Broqueres-You, Yang, Wang, Li and Yang16,Reference Dasgupta, Singh, Rout, Saha and Mandal40–Reference Kirkpatrick, Garcia-Rizo, Tang, Fernandez-Egea and Bernardo43,Reference Venkatasubramanian, Chittiprol, Neelakantachar, Shetty and Gangadhar47 see online Table DS9. Thus, the potential use of psychiatric medication other than antipsychotics that might increase lipid parameters, such as mood stabilisers, Reference Stubbs, Firth, Berry, Schuch, Rosenbaum and Gaughran57 or lipid-lowering medication is a potential confounding factor for some of the studies that we were unable to account for fully. However, evidence in chronic schizophrenia indicates that patients are less likely to receive lipid-lowering drugs than the general population, Reference Nasrallah, Meyer, Goff, McEvoy, Davis and Stroup64 so this is unlikely to account for the reductions we see. Although no study included in our analysis specifically excluded participants based on a historical diagnosis of dyslipidaemia, all but two studies included statements that described participants as physically healthy (Table DS9). Reference Verma, Subramaniam, Liew and Poon17,Reference Phutane, Tek, Chwastiak, Ratliff, Ozyuksel and Woods65 Like the process of BMI-matching patients and controls, this preferential selection of physically healthy participants may have excluded participants truly representative of the patient (and control) cohort, with overmatching of groups.
Our database search strategy was complemented by hand-searching, which identified additional studies that were included in the final analysis. This could indicate that there are other studies missed by the database search; however, as we hand-searched the reference lists of the studies included and additional review articles, any missing articles have eluded the field in general.
We observed relatively high heterogeneity in some of our analyses, notably of triglycerides. The use of plasma or serum sampling for lipid parameters, which has been reported to result in a 3–5% variation in cholesterol measures, 66,Reference Cloey, Bachorik, Becker, Finney, Lowry and Sigmund67 and differences in sample inclusion criteria may account for some of this heterogeneity. In addition, six studies failed to document the model or brand of analyser used (online Table DS10), thereby precluding confirmation of interassay reliability. Reference Saddichha, Manjunatha, Ameen and Akhtar19,Reference Enez Darcin, Yalcin Cavus, Dilbaz, Kaya and Dogan39,Reference Kirkpatrick, Garcia-Rizo, Tang, Fernandez-Egea and Bernardo43,Reference Spelman, Walsh, Sharifi, Collins and Thakore44,Reference Sengupta, Parrilla-Escobar, Klink, Fathalli, Ying Kin and Stip46,Reference Srihari, Phutane, Ozkan, Chwastiak, Ratliff and Woods51 Nevertheless, the random effects model we used is robust to heterogeneity and subgroup analyses explored potential sources of heterogeneity. It is important to note that because most studies in our analysis selected patients who were physically healthy, and matched for a number of risk factors for dyslipidaemia such as BMI, our findings should not be extended to patients with these risk factors. By the same token, our meta-analysis should not be extended to draw conclusions on rates of categorical diagnoses of dyslipidaemia in FEP. The data in our analyses are cross-sectional and future longitudinal research is required to disentangle the relationships we observed and the potential role of modifiable factors.
Future directions
Although the effect sizes observed in our meta-analysis are modest, they are striking in the context of the opposite findings in patients with chronic psychotic disorders. Our findings indicate that psychotic disorders are not associated with an intrinsic elevation in total cholesterol, LDL cholesterol or reduction in HDL cholesterol, and indicate that patients have favourable profiles of these lipids early in their illness. Our results contrast with the findings of meta-analyses in patients with chronic disorder, which, in comparison with the general population, show increased risk of the metabolic syndrome and hypercholesterolaemia. Reference Vancampfort, Wampers, Mitchell, Correll, De Herdt and Probst14 Taken together with these, our findings suggest that the increased risk of hypercholesterolaemia in psychotic illness is a consequence of potentially modifiable factors associated with chronic psychotic illness. Whether lower cholesterol in FEP is indicative of inherent cardiometabolic differences between FEP and controls, as suggested by recent meta-analytic evidence of glucose dysregulation in early psychosis, Reference Perry, Mcintosh, Weich, Singh and Rees21,Reference Greenhalgh, Gonzalez-Bianco, Garcia-Rizo, Fernandez-Egea, Miller and Arroyo54,Reference Pillinger, Beck, Gobjila, Donocik, Jauhar and Howes68 remains to be determined. Recent Genome Wide Association Study evidence has demonstrated pleiotropic enrichment between genetic polymorphisms associated with both schizophrenia and cardiovascular disease, and lipid disorder Reference Andreassen, Djurovic, Thompson, Schork, Kendler and O'Donovan69 This could suggest there are common molecular pathways underlying both psychosis and metabolic dysregulation. Reference Liu, Li, Zhang, Deng, Yi and Shi70 Adipocytokine disturbances are one possibility: there is robust evidence of immune dysregulation in antipsychotic-naïve FEP, with elevated peripheral cytokines that include the adipocytokines interleukin-6 (IL-6) and tumour necrosis factor alpha (TNF-α). Reference Upthegrove, Manzanares-Teson and Barnes71 Interleukin-6 has been demonstrated to reduce lipid levels in animal models. Reference Katsume, Saito, Yamada, Yorozu, Ueda and Akamatsu72 Active, untreated rheumatoid arthritis, which is associated with elevated IL-6 and TNF-α levels, is associated with decreased total and LDL cholesterol levels, Reference Myasoedova, Crowson, Kremers, Fitz-Gibbon, Therneau and Gabriel73 and these increase with IL-6 and TNF-α monoclonal blockade. Reference Nishimoto, Yoshizaki, Miyasaka, Yamamoto, Kawai and Takeuchi74 Thus, by extension, inflammation could be a mechanism underlying the lipid alterations we found. There were insufficient data for us to test the hypothesis of broader adipocytokine dysregulation in FEP further, and our leptin analysis demonstrated no difference between patients and controls. Further preclinical and clinical studies are required to probe this potential association.
A message for clinicians is that the secondary, potentially modifiable risk factors should be considered and addressed from onset of illness before dyslipidaemia has developed. Potential modifiable factors associated with schizophrenia include poor nutrition, Reference Dipasquale, Pariante, Dazzan, Aguglia, McGuire and Mondelli75 sedentary behaviour, Reference Stubbs, Gardner-Sood, Smith, Ismail, Greenwood and Farmer76 alcohol and substance misuse, Reference Koola, McMahon, Wehring, Liu, Mackowick and Warren77 poorer access to healthcare, Reference Kurdyak, Vigod, Calzavara and Wodchis78 and long-term antipsychotic treatment. Reference Malhotra, Graver, Chakrabarti and Kulhara11,Reference Galling, Roldan, Nielsen, Nielsen, Gerhard and Carbon79 Among these, antipsychotics – especially the newer antipsychotics – are well recognised as potentially modifiable risk factors for metabolic dysfunction. Reference De Hert, Detraux, van Winkel, Yu and Correll80–Reference Howes, Bhatnagar, Gaughran, Amiel, Murray and Pilowsky82 The mechanisms by which antipsychotics contribute to lipid disorder remain poorly understood, but may result from weight gain secondary to activity at 5-hydroxytryptamine (serotonin) 5-HT2 and 5-HT1 receptors, leading to increased dietary fat intake. Reference Reynolds, Zhang and Zhang83,Reference Kim, Huang, Snowman, Teuscher and Snyder84 It is recognised that individuals with FEP and a relatively lower BMI tend to gain weight rapidly when treated with an antipsychotic associated with a high weight gain potential. Reference Kinon, Kaiser, Ahmed, Rotelli and Kollack-Walker85 Longitudinal studies examining changes in lipids from the at-risk mental state and over the course of the first episode and first few years of illness would be useful to determine what underlies the transition from low cholesterol and LDL levels to elevated levels. Such research should consider the relative role of modifiable risk factors such as nutrition, physical activity and antipsychotic medication on lipid changes. We also demonstrated the paucity of studies examining adipocytokine parameters in early schizophrenia. This indicates that more research is required to examine the potential mechanisms underlying the lipid profile we have identified in this meta-analysis.
Our finding of increased triglyceride concentrations in FEP is consistent with meta-analytic evidence of raised triglyceride levels in patients with chronic illness compared with the general population. Reference Vancampfort, Wampers, Mitchell, Correll, De Herdt and Probst14 Elevated triglyceride levels are considered a marker of insulin resistance, and predict risk for impaired fasting glucose concentration. Reference Tirosh, Shai, Bitzur, Kochba, Tekes-Manova and Israeli56 As such, triglycerides are used in diabetes risk prediction models. Reference Stern, Fatehi, Williams and Haffner86–Reference Schmidt, Duncan, Bang, Pankow, Ballantyne and Golden88 The proposed mechanism sees insulin resistance at the level of the adipocyte associated with increased intracellular hydrolysis of triglycerides and release of fatty acids into the circulation. Reference Ginsberg89 This induces hepatic production and release of very low-density lipoproteins, which results in hypertriglyceridaemia. Reference Ginsberg89 Thus, raised triglyceride concentrations in FEP may reflect early glucose dysregulation and insulin resistance, consistent with recent meta-analyses indicating that patients with FEP show altered glucose homeostasis. Reference Perry, Mcintosh, Weich, Singh and Rees21,Reference Pillinger, Beck, Gobjila, Donocik, Jauhar and Howes25,Reference Greenhalgh, Gonzalez-Bianco, Garcia-Rizo, Fernandez-Egea, Miller and Arroyo54 Given that they are indicators of early glucoregulatory dysfunction, triglycerides could be a focus for early intervention to prevent the development of diabetes in schizophrenia.
Implications
Our data suggest that total cholesterol and low-density lipoprotein levels are reduced early in the course of schizophrenia, indicating that the hypercholesterolaemia seen in chronic disorder is secondary and potentially modifiable. In contrast, our finding of elevated triglyceride levels early in the course of schizophrenia is a metabolic indicator of an increased risk of diabetes. In terms of translation to the clinical domain, these findings suggest that prevention of dyslipidaemia and diabetes needs to be prioritised from onset of psychosis, with the adoption of early lifestyle interventions and careful antipsychotic prescribing which considers both benefits and risks. Reference Cooper, Reynolds, Barnes, England and Haddad90
Funding
The study was funded by the UK Medical Research Council, the Maudsley Charity, the Brain and Behavior Research Foundation and the Wellcome Trust. The funders had no role in study design, data collection, data analysis, data interpretation or writing of the report.
Acknowledgements
We thank Professor Venkatasubramanian of the National Institute of Mental Health and Neuroscience, Bengaluru, India, Dr Vivek Phutane of the Yale University School of Medicine and Connecticut Mental Health Center, USA, and Dr Blazej Misiak of the Wroclaw Medical University, Poland, for providing additional data.








eLetters
No eLetters have been published for this article.