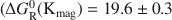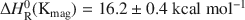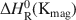Extreme silver enrichment at the surface of the complex sulphide, tennantite (ideal formula: Cu12As4S13), occurs following exposure to alkaline solutions, and involves the development of an Ag-rich sulphide surface species. The tennantite has a low bulk Ag content of 0.3 at.%, and a percentage surface enrichment of Ag is thirty-six times that of the bulk. The techniques of X-ray photoelectron spectroscopy (XPS) and reflection extended X-ray absorption fine structure spectroscopy show the new phase to be a Ag sulphide species compositionally similar to cupriferous proustite ((Cu,Ag)3AsS3). Solution experiments and XPS depth profiling show that the surface is most depleted in Cu and Zn, and enriched in Ag compared to the bulk tennantite. Selective dissolution and reprecipitation at the tennantite surface cannot explain the enrichment of Ag relative to the bulk. Migration must have occurred and could have been driven by the leaching out of Cu which produces a metal-depleted surface, coupled to the relative incompatibility of Ag in the tennantite lattice. To account for the extreme enrichment at the surface, Ag must have diffused from depths of up to 9 nm, probably via structural weaknesses and vacancies in the tennantite lattice.