According to the FAO, the number of chronically undernourished people in the world stood at 805 million between 2012 and 2014( 1 ). Undernourishment occurs as a result of an energy intake less than the amount needed for light activity and to maintain an appropriate weight in proportion to height( 2 ). Due to significant nutrient deficiencies, undernourished individuals, particularly children, are considerably more susceptible in acquiring infectious diseases, including HIV. Sub-Saharan Africa has the highest proportion of undernourished people in the world, with close to one in four of its population considered chronically undernourished( 1 ). The region was also home to approximately 24·7 million people living with HIV in 2013, accounting for about 70 % of the global total of new HIV infections( 3 ). Given the increased availability of antiretroviral treatment (ART) in low-to-middle-income countries, the high prevalence of undernutrition can pose a significant hurdle to AIDS recovery; as food insecurity has been identified as a primary obstacle to ART adherence( Reference Au, Kayitenkore and Shutes 4 ). To date, ART coverage in Sub-Saharan Africa is estimated at 37 %, and roughly three out of every four people currently on ART live in Sub-Saharan Africa( 3 ).
According to the WHO guidelines, nutritional support should be incorporated into HIV care alongside ART. Recommendations include an increase in energy intake of up to 50 % compared with an HIV-negative individual, but a normal intake of micronutrients and protein( 5 ). Micronutrients play important functional roles in the human immune response; however, a number of micronutrient deficiencies have been reported in HIV-positive individuals. This has been linked to poor nutrient absorption due to intestinal cellular damage caused by oxidative stress.
Nutrients, immunity and HIV infection
Micronutrients in particular antioxidant minerals such as selenium, zinc and vitamins A, C and E play a key functional role in an active immune response( Reference Taylor 6 ). For example, the antioxidant role of vitamin A and in particular its role in maintaining mucosal immunity makes it an important dietary component( Reference Edge, McGarvey and Truscott 7 ). A function for vitamin A in enhancing T helper type 2 cytokine production and secretory IgA response to viral infections especially at high doses has also been reported( Reference Cui, Moldoveanu and Stephensen 8 ). In addition, carotenoids and other vitamin A compounds have been shown to inhibit reverse transcriptase in HIV-1 and -2( Reference Kaio, RONDÓ and Souza 9 ). Similarly, vitamin C has been shown to inhibit the HIV virus in vitro ( Reference Harakeh, Niedzwiecki and Jariwalla 10 ).
The combination of vitamin A deficiency and wasting have been identified as predictors of mortality in HIV disease( Reference Semba, Caiaffa and Graham 11 ). Low zinc and iron levels have also been associated with immune abnormalities, increased susceptibility to infections and gastrointestinal malfunction with diarrhoea( Reference Mocchegiani and Muzzioli 12 ). Such data support the argument for micronutrient supplementation in HIV patients; however consideration must be taken as high doses of iron can induce oxidative stress and stimulate microbial growth, whereas excess zinc can cause HIV gene expression, multi-mineralisation and integration( Reference Baum, Shor-Posner and Campa 13 , Reference Crist, Melekhin and Bian 14 ).
Antioxidant defence systems are essential in limiting various reactive species (RS) from causing intestinal cellular damage( Reference Sharma 15 , Reference Isaac, Alex and Knox 16 ). Immune activation can contribute considerably to the overproduction of RS at the site of infection such as the intestinal area. In HIV-infected individuals who face the double-burden of also being undernourished, the level of antioxidant protection may not be present to thwart this overproduction, which inevitably leads to oxidative stress( Reference Nkengfack, Torimiro and Englert 17 ). With the onset of oxidative stress, intestinal cells can become further degraded as RS oxidise the PUFA component of membranes, disrupting cellular structure and function in the process( Reference Sharma 15 ). This causes nutrient malabsorption, which is linked to wasting in HIV patients( Reference Isaac, Alex and Knox 16 ). In addition, oxidative stress-related tissue damage caused during HIV infection triggers the activation of NF-κB, which in turn can further enhance HIV replication( Reference Chan and Greene 18 ).
The extent of oxidative damage can be determined by measuring biochemical parameters such as glutathione (GSH), glutathione peroxidase, malondialdehyde or total antioxidant status( Reference Coaccioli, Crapa and Fantera 19 – Reference Posse and Baltussen 21 ). The GSH has been shown to be of importance in immune function and restoration of CD4T lymphocyte levels in HIV infection( Reference Herzenberg, De Rosa and Dubs 22 – Reference Rivas, Vera and Guaiquil 24 ). During an HIV infection, not only does GSH decrease, but there is also an increase in oxidised GSH and protein-bound GSH( Reference Wu, Fang and Yang 25 ). The accompanying change in GSH : oxidised GSH ratio indicates a change in redox status and increased oxidative stress( Reference Wu, Fang and Yang 25 ). Such oxidative stress can be exacerbated by folate deficiency, which is associated with increased production of homocysteine, a potential RS( Reference Wu, Fang and Yang 25 ).
Sulphur-containing amino acids are important in metabolism and immune function, which can become depleted in the onset of protein deficiency( Reference Wu, Fang and Yang 25 ). Furthermore, should protein also be deficient there would be insufficient building blocks for GSH synthesis (cysteine) and the methylation cycle (methionine)( Reference Wu, Fang and Yang 25 ). Cysteine and methionine are both important in the methylation cycle (crucial to folate, B12 and homocysteine metabolism) and the GSH pathway( Reference Wu, Fang and Yang 25 ). Protein deficiency would also lead to biochemical (metabolic) adaptations, which would mean various critical enzyme systems and immune globulin synthesis may become compromised( Reference Wu, Fang and Yang 25 ). The loss of lean tissue, which is a characteristic of HIV wasting, would ultimately lead to patient death.
HIV, food insecurity and undernutrition
The synergism of HIV and undernutrition is unfortunate as one has a debilitating effect on the other, often in spite of access to ART (Fig. 1). An HIV-positive person in a food insecure household would not consume an adequate diet consisting of nutrients required for maintaining a functional immune system to prevent opportunistic infections( Reference Ivers, Cullen and Freedberg 26 ). This situation usually leads to a rapid onset of AIDS, which causes a decrease in productivity due to illness and death. HIV-affected households are thus more likely to experience income losses, which in turn exacerbates food insecurity and low ART adherence, as difficult situations may arise such as choices between purchasing either food or treatment( 27 ).
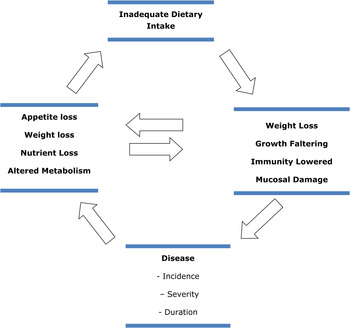
Fig. 1. (Colour online) The infection–malnutrition cycle.
As a general rule, drug adherence is difficult to achieve in an undernourished patient. ART has been shown to stimulate appetite in HIV patients, which helps with body mass recovery and improves immune function( Reference McDermott, Shevitz and Must 28 , Reference Raiten, Grinspoon and Arpadi 29 ). However, in a food-insecure household this may not be desirable as there would be insufficient food to satisfy a stimulated appetite. Current evidence suggests that food insecurity is a significant contributor to non-compliance with ART, and may offset the benefits of treatment. For example, HIV patients in Kenya who were offered free ART from Medecins Sans Frontiers, cited fears of the potential side effects of taking ART on an empty stomach as a key reason for declining treatment( Reference Unge, Johansson and Zachariah 30 ). Similarly, in Rwanda, patients interviewed listed the concern of having too much appetite without the food to fulfil it as an obstacle in maintaining adherence( Reference Au, Kayitenkore and Shutes 4 ). Considerable adverse effects have been observed in food-insecure patients on ART such as hepatic toxicity( Reference Sanne, Mommeja-Marin and Hinkle 31 ). In a Ugandan study, it was observed that food insecurity was heavily associated with non-adherence to ART, incomplete viral suppression and having a CD4 count <350 cells/mm3( Reference Weiser, Palar and Frongillo 32 ). This phenomenon has presented the argument for nutrition intervention to be formally introduced into HIV palliative care. Given that many HIV-infected persons in low-income countries also suffer from the complications of acute to severe malnutrition, the benefits of using ready-to-use-therapeutic foods (RUTF) is being investigated among this population. Research has indicated that RUTF use is generally accepted by HIV-infected persons as a means to counteract the adverse effects of ART; and has been described as ‘food with medicinal qualities’, making it less likely to face any cultural perceptions and practices associated with food, including sharing and fasting( Reference Olsen, Tesfaye and Kaestel 33 ).
Food supplementation
Food intervention strategies in food-insecure countries have traditionally involved imported food rations, which are not the most financially sustainable. In Mozambique, the cost of providing food assistance to HIV-infected persons was estimated at US$288 over a 3-month period, and highlighted that a significant portion of costs was related to overheads such as transport( Reference Posse and Baltussen 21 ). Thus the local production of RUTF in food-insecure regions is being investigated, albeit on a low level. Preliminary technical guidelines are available for small- and medium-scale therapeutic food production in resource-limited settings( Reference Manary 34 ). Local therapeutic food production programmes have been successfully implemented in a number of countries such as Niger, the Congo and Malawi. The majority of therapeutic foods have been designed as treatment for acute to severe malnutrition; however, research into how therapeutic foods can complement ART as part of HIV care is also ongoing( Reference Manary and Sandige 35 ). Despite some being locally sourced, most ingredients of RUTF are either imported or donated by the World Food Programme (WFP)( Reference Krumbein, Scherbaum and Biesalski 36 ). Thus operations remain largely unsustainable despite local production. In Malawi, the cost of local therapeutic food production was estimated at US$2·60/kg, with approximately 54 % of this price covering the cost of imported ingredients( Reference Manary and Sandige 35 ). In Kenya, a nutrient-dense therapeutic food formula using all locally sourced ingredients was developed, piloted for taste testing and found to be well adhered to by participants( Reference Hong, Hendricks and Wanke 37 ).
Aim of review
To date the biochemical aspect of HIV infection and its relationship with antioxidant nutrients has not often featured in nutritional intervention studies. Nevertheless, the use of food supplementation, in particular micronutrient-fortified, RUTF as part of nutrition intervention strategies has gained momentum. The aim of this review is to highlight the most recent studies related to food supplementation in HIV-infected adults in Sub-Saharan Africa; analysing where available, measurable outcomes such as weight gain and ART adherence with a view to provide evidence for debate on this important topic in Africa.
Selected studies
Relevant studies were identified by a search conducted in the PUBMED database. Keywords used were ‘food’ ‘HIV’ ‘supplementation’ and ‘intervention’. The search was confined to a 10-year period (2004–2014) and studies were chosen based on pre-defined inclusion/exclusion criteria. Studies were considered relevant if they measured at least two of the following outcomes, including weight/BMI, ART adherence and CD4 count. Studies were excluded if they were conducted outside of Sub-Saharan Africa, involved a non-food intervention and/or if there was either no article full-text available or the full text was not accessible free of charge.
A total of ten primary studies were identified and seven met the criteria to be included in this review. Two studies were conducted outside of Sub-Saharan Africa (Haiti and India) and one study involved a non-food intervention. All seven studies (one in Zambia, two in Uganda and four in Malawi) were conducted among adults aged 18 years and older (Table 1). Data extracted from each study included type of study, description of food intervention, location, duration, WHO clinical stage, sample size and measurable outcomes. In three studies, participants were described as food-insecure and in two studies participants were treatment naive. In all eight studies, participants were malnourished and/or wasted and presented with WHO clinical stage III or IV of HIV infection.
Table 1. Data from seven primary studies included in the present review

CSB, corn–soya blend; RUFS, ready-to-use fortified spread; RUTF, ready-to-use-therapeutic food; WFP, World Food Programme; MUAC, mid-upper-arm circumference; N/A, not applicable.
In Zambia, approximately 54·6 % of the 1·1 million people living with HIV have access to ART as of 29 September 2014( 44 ). In 2013, there were approximately 27 000 AIDS-related deaths( 44 ). A non-randomised study by Cantrell et al. investigated the effect of food supplementation on ART adherence in HIV adults in Zambia( Reference Cantrell, Sinkala and Megazinni 38 ). Food supplementation consisted of micronutrient fortified corn–soya blend (CSB) containing 6·2 kg protein; and 620 ml vegetable oil per month for each individual ration. The programme ran from May 2004 to March 2005 (11 months) and involved 636 HIV-positive adults with a mean CD4 cell count of 131 cells/mm3. One hundred and thirty-four participants (22 %) had a CD4 count of <50 cells/mm3. Three hundred and forty seven (79 %) were categorised as WHO clinical stage III or IV, whereas nine-two (21 %) were categorised in clinical stage I or II.
The 411 women had a mean baseline weight of 52·9 kg, whereas the 218 men had a mean baseline weight of 56·1 kg. All participants were described as food-insecure. Food distribution was based on whether the patient was a primary income earner, in which case they would receive an individual ration along with additional rations sufficient for six household members. Participants classified as the food group began food rations in September 2004, whereas the control group were assigned to receive rations at a later date. ART adherence was measured within the first 12 months, according to the medication possession ratio, where the number of days late for pharmacy refills was expressed as a percentage of the total days of therapy, and then subtracted from 100 %. It was reported that adherence was higher in the food group compared with the control group, 70 % (258/366) patients in the food group achieved an adherence of 95 %, compared with 48 % (79/166) in the control group. Differences in weight gain between the food and control groups were 5·4 v. 5·1 kg respectively after 6 months, and 6·3 v. 5·4 kg after 12 months, which although NS, was described as a ‘trend towards modest benefit’. There was no reported increase in CD4 count in the food group compared with the control group. All food rations used in the study were donated by the WFP( Reference Cantrell, Sinkala and Megazinni 38 ).
Approximately 46 % of Malawi's HIV population of one million people were receiving ART by 2014; of which approximately 90 % were adults( 44 ). A randomised, investigator blinded, controlled trial in Malawi reported on the effectiveness of ready-to-use fortified spread (RUFS) v. CSB in achieving weight gain in HIV adults after 3·5 months( Reference Cantrell, Sinkala and Megazinni 38 ). The study consisted of 491 HIV adults receiving ART. All participants had a BMI of <18·5 kg/m2, a CD4 count of <250 cells/mm3, and were categorised as WHO clinical stage III or IV. Primary outcomes included BMI and fat-free mass, whereas secondary outcomes included ART adherence, survival, CD4 count, HIV viral load and quality of life. After 14 weeks, mortality rates were similar in both groups (27 % in RUFS v. 26 % in CSB).
There was no significant difference in ART adherence, which was high in both groups; neither was there a difference in CD4 cell count, viral load or quality of life. Participants in the RUFS group reported a 13 % weight increase compared with a 10 % increase in the CSB group. Fat-free mass contributed to 2·9 kg (51·8 %) of the 5·6 kg weight gain in the RUFS group and 2·2 kg (51·2 %) of the 4·3 kg weight gain in the CSB group. BMI increased by 2·2 in the RUFS group, compared with 1·7 in the CSB group. In the present study, the CSB was priced at US$5·40/patient per month, whereas the RUFS cost US$16·00/patient per month, which was approximately US$1 more expensive than first line ART( Reference Ndekha, van Oosterhout and Zijlstra 39 ).
A prospective descriptive study highlighted the effects of a locally produced RUTF in sixty HIV-positive adults in Malawi( Reference Bahwere, Sadler and Collins 40 ). A total of forty-five out of sixty participants (75 %) were at WHO clinical stage IV and fifteen (25 %) were at WHO clinical stage III. Participant eligibility included a BMI of <17 kg/m2 and a middle-upper-arm circumference (MUAC) of <210 mm.
Participants were given 500 g daily of the RUTF, which consisted of chick peas, sesame seeds, vegetable oil, sugar, maize and a micronutrient complex. This provided a daily amount of 11217·3 kJ (2681 kcal) energy, 61·5 g protein, 24 mg niacin, 2·5 mg vitamin B6, 8 μg vitamin B12, 241·5 mg vitamin C and 62 mg zinc, among other micronutrients. Only eight participants (13·3 %) were already on triple ART including stavudine, lamivudine and nevirapine. A daily dose of cotrimoxazole (960 mg) was also given to participants for the 3-month duration of the study. Adherence to the food supplement was measured by the number of finished pots and the measurable outcomes included MUAC, BMI, weight gain and physical activity. Out of sixty participants, forty four (73·3 %) reported increases in weight, MUAC and BMI (2·5 kg, 15 mm and 0·8 kg/m2, respectively). A lower BMI, MUAC and weight was observed in participants already on ART at the start of the study compared with ART naive participants. The number of participants able to walk to the clinic increased from twenty five to forty seven (41·7 to 78·3 %). The cost of a 500 g/d ration of RUTF was valued at US$45·90/person per month, which was more cost effective than other nutrition interventions including the standard food aid supplement (CSB and oil) valued at US$53·25 per month( Reference Bahwere, Sadler and Collins 40 ).
According to the Joint United Nations Programme on HIV/AIDS, Kenya has the fourth largest ART programme among low- and middle-income countries, although only 41 % of the estimated 1·6 million HIV population currently receives treatment( 44 ). Following close behind at fifth is neighbouring country Uganda, which had 38 % of its estimated 1·6 million HIV population on ART in 2014. A retrospective cohort analysis involved the use of RUTF originally developed for treatment of severe malnutrition such as Plumpy'Nut® and Nutriset, aimed to investigate nutritional outcome in HIV adults and to highlight the factors behind treatment failure( Reference Unge, Johansson and Zachariah 30 ). Over a 2-year period (March 2006–August 2008) a total of 1106 malnourished adults from a Kenyan and a Ugandan HIV programme participated in the study. Participants had a median CD4 count of 114 cells/mm3 and a BMI <17 kg/m2. A total of 617 participants were severely malnourished (55·8 %), whereas 489 were moderately malnourished (44·2 %). The amount of participants categorised in either WHO clinical stage III or IV was 705 (63·7 %). Out of the 790 adults eligible for ART, 133 (16·8 %) were already on treatment, whereas 470 (59·5 %) began treatment either at the start or during the programme. A total of 187 (23·7 %) participants did not receive any treatment for the duration of the study. The RUTF consisted of a micronutrient fortified, energy-dense spread of peanut, milk powder, oil and sugar. Participants achieving BMI ≥18 kg/m2 with no oedema for a minimum of 2 weeks were defined as cured, based on the programme guidelines and permitted to exit the study. A total of 524 participants (47·4 %) achieved this target and were discharged after a median period of 3·7 months, with a median weight gain of 8 kg (1·6 g/kg per d). Forty-nine participants (4·4 %) were discharged as uncured after a median of 7·1 months with a median CD4 count of 292 cells/mm3 In total, 531 participants (48·0 %) failed to complete the programme, which included 250 (22·6 %) who defaulted and 132 (11·9 %) who died; both of which were severely malnourished, received nutritional treatment for <3 months, had low CD4 cell counts (96 and 36 cells/mm3, respectively) and/or received no ART despite being eligible( Reference Ahoua, Umutoni and Huerga 41 ).
A retrospective study conducted in Malawi involving 593 HIV wasted adults investigated the effects of food supplementation v. no nutritional intervention( Reference Van Oosterhout, Ndekha and Moore 45 ). Participants were previously ART naive, either WHO clinical stage III or IV, a BMI <18·5 kg/m2 and CD4 count <250 cells/mm3. All initiated ART upon enrolment, and were compared with a historical control group that received no supplementation but was given a dosage of cotrimoxazole prophylaxis. Outcomes were measured at the end of supplementation at 14 weeks and again at 26 weeks. Approximately half (245) of participants in the supplement group received CSB, whereas the remainder (244) received an RUFS; both of which were fortified with micronutrients. Higher increases of BMI were reported in the supplemented group compared with the control group. Although eight (4 %) more people receiving RUFS died, it was more effective at increasing BMI and weight than the CSB, as increases of 0·5 kg/m2 and 1·2 kg, respectively, were observed. However, only 9 % of participants receiving the RUFS supplement had a BMI that was <18 kg/m2. In comparison with the control group, treatment adherence improved among the supplemented group with only four people stopping ART (one on RUFS v. three on CSB). There was no significant difference in CD4 count. The CSB supplement was priced at a monthly cost of US$5, whereas the RUFS was priced at US$16/month, which at the time of the study was US$1 more expensive than first-line ART treatment in Malawi( Reference Van Oosterhout, Ndekha and Moore 45 ).
An observational study described the effect of food supplementation on food-insecure HIV adults needing home-based care in Malawi( Reference Van Oosterhout, Ndekha and Moore 45 ). Food was provided to households by the WFP, and contained monthly rations of 50 kg maize, 5 kg beans and 7·5 kg Likuni Phala (cereal–soya blend). Half of the households in the study also received a random allocation of 4 litres oil, which was not strictly adhered to.
The study consisted of 360 participants, and food distribution ran for 14 months from July 2003 to September 2004. The study was divided into three periods: January 2003–July 2003, August 2003–November 2003 and mid-November 2003–July 2004. Approximately ninety-seven enrolled patients were chronically ill; up to 70 % presented advanced stages of HIV infection (WHO clinical stage IV). Half of the participants were malnourished upon enrolment, with a BMI <18·5 kg/m2. Mortality rate was high, as 112 (one-third) of participants died within the first 4 months. By the end of the study, 199 participants (56 %) had died. Only twenty-two participants survived through all three periods. A slight increase in BMI was observed among these participants (0·49 kg/m2 per 100 d by the second survey and 0·46 kg/m2 by the third survey), but this was NS. Participants assigned to receive oil supplements showed no significant change in mean BMI, although survival was better in this group. At the time of the present study, ART was not available free in Malawi and all participants were treatment naive.
Changes in CD4 count were not reported in the present study. Although ART adherence was not specified, it was indicated that participants with increased physical activity were now able to walk to Voluntary Counselling and Testing centres and enrol in ART programmes( Reference Bowie, Kalilani and Marsh 42 ).
A prospective quasi-experimental study evaluated the impact of a WFP food basket (200 g maize meal, 40 g pulses, 10 g vitamin A-fortified vegetable oil, 5 g iodised salt, 50 g micronutrient fortified CSB/person per day) provided monthly over a 12-month period (August 2008–October 2009) to HIV adults in Uganda that were both food-insecure and ART naive( Reference Rawat, Faust and Maluccio 43 ).
A total of 904 adults participated in the initial study, based on their eligibility to receive the WFP food basket and having a CD4 count between 200 and 450 cells/m3; whereas 604 adults were part of the follow-up study. The food basket provided approximately 4602·4 kJ (1100 kcal)/person per day and resulted in a BMI increase of 0·6 kg/m2 and a MUAC increase of 6·7 mm. No significant association was observed between food assistance and CD4 count; however, in participants with a CD4 count >350 cells/m3, Hb concentrations increased by approximately 1·0 g/dl( Reference Rawat, Faust and Maluccio 43 ).
Types of food supplementation
In the studies highlighted in this review, food supplementation predominantly consisted of RUTF and CSB, both of which had a high nutrient density and were developed to treat severe malnutrition. Notable differences existed between these two supplements, namely preparation methods and cost. As the RUTF did not require cooking, it was considered less likely to be shared with other household members, compared with the CSB, which was considered to be more culturally accepted. The CSB was also significantly less expensive than the RUTF.
According to Ahoua et al. the currently available RUTF was designed to treat paediatric malnutrition; it may not be the most suitable formulation for the treatment of malnutrition in HIV-infected adults ( Reference Au, Kayitenkore and Shutes 4 1 ) . In two studies the effect of WFP food rations were examined( Reference Bowie, Kalilani and Marsh 42 , Reference Van Oosterhout, Ndekha and Moore 45 ). In these studies both study populations were ART naive, which may account for the small-to-modest increases in BMI observed (0·6 and 0·49 kg/m2, respectively). Although the food ration studies showed little to no improvement in anthropometric gains it highlighted the impact of HIV-related complications on nutritional recovery and survival. It also highlighted the ineffectiveness and lack of sustainability of food distribution programmes.
All but one study used an intervention strategy based on imported RUTF and food rations. The locally produced RUTF examined by Bahwere et al. performed well in improving physical activity of participants; and was more affordable than imported food supplements( Reference Bahwere, Sadler and Collins 40 ). Studies were noted to run for relatively short durations, which was a significant limitation. The longest study was conducted over a 2-year period, whereas three studies ran between 11 and 14 months. A further three studies ran for 3–3·5 months; however, this was in order to limit metabolic side effects associated with ART regimens such as lipodystrophy due to the high-fat content of the RUTF supplement. Still, such a limited follow-up time was considered insufficient to fully observe the impact of the food intervention. It was also not possible to monitor the effect of access to and consumption of other food sources, which may have impacted on the results of weight gain.
Study outcomes
Despite these limitations a key observation made in all studies was that food supplementation improved weight gain and BMI. Most notably was the 6·3 kg weight increase after 12 months in participants from the study by Cantrell et al. ( Reference Cantrell, Sinkala and Megazinni 38 ). Also, in all studies where it was measured, ART adherence was significantly high among participants receiving food supplementation. None of the food intervention strategies however had any significant impact on CD4 cell count in the studies where it was measured. Where measured, mortality rates were relatively high; and were largely associated with participants who were severely malnourished and ART naive( Reference Ahoua, Umutoni and Huerga 41 , Reference Bowie, Kalilani and Marsh 42 ). These results are significant and provide justification for future study, as it highlights the important role food and nutrition security plays in improving overall quality of life for HIV patients.
A follow-up to the earlier Ndekha study by the same research group assessed the BMI of patients subsequent to food supplementation( Reference Ndekha, van Oosterhout and Saloojee 46 ). Participants were followed at 3- and 9-month intervals after the end of the intervention trial. The initial increase in BMI was not sustained without supplementation, neither was the improvement in ART adherence and quality of life( Reference Ndekha, van Oosterhout and Saloojee 46 ) The study concluded that food supplementation must be maintained throughout in order to achieve maximum nutritional gains. Clearly, this can only be achieved with a sustainable nutrition intervention programme.
Field studies involving nutrition intervention for HIV patients have generally relied on anthropometric data as primary indicators of supplementation efficacy; however, the inclusion of biochemical parameters such as total antioxidant content and lipid peroxidation can provide substantial information as to the occurrence and extent of nutrition recovery. Malondialdehyde and 4-hydroxynonenal are identified as lipid peroxidation end products and plasma-level measurements have been used to indicate oxidative stress occurring in the body. Oxidative stress caused by RS overproduction can be indicative of HIV infection and/or undernutrition( Reference Nkengfack, Torimiro and Englert 17 ); which is linked to nutrient malabsorption and wasting in HIV patients( Reference Isaac, Alex and Knox 16 ). A 1988 study showed malondialdehyde levels to be as much as 30 % higher in HIV-positive patients compared with their HIV-negative counterparts( Reference Ndekha, van Oosterhout and Saloojee 46 ); while a study almost a decade later made similar observations in children and new-born infants( Reference Sönnerborg, Carlin and Akerlund 47 ). None of the studies indexed in PUBMED and included in this review made an assessment of biochemical parameters.
The Food Multi-Mix concept
According to the Food Multi-Mix (FMM) concept described by Zotor et al., a food-based approach to address nutritional needs for therapeutic use can be employed as a cost-effective and sustainable means to improve nutrition in HIV patients( Reference Zotor and Amuna 48 ). The aim of the FMM concept is to achieve affordability, sustainability and acceptance of therapeutic food supplementation. The FMM concept has been previously described in its utilisation of locally produced and commonly consumed foods within a community setting for clinically based interventions( Reference Zotor and Amuna 48 ). It allows for nutritional support in HIV and wasting syndromes, which is aimed at replenishing energy and nutrient losses and improving weight gain. In selecting food ingredients, total energy requirements, including additional demands to compensate for altered metabolism and special roles of nutrients, including vitamins A, C, folate and minerals such as zinc, selenium were all taken into consideration in accordance with the specific requirements of an HIV patient.
In various FMM, β-carotene (vitamin A equivalent) content was on average 30 % above recommended nutrient intake values per 300 g serving. Two servings would thus provide 60 % more than recommended nutrient intake values. Apart from eggs, the carotene sources of FMM are mostly plant-derived and are thus more affordable, thus reducing the cost of meal provision. HIV patients also require additional protein to ensure adequate immune system function. The consumption of 200 g of any of the FMM can provide at least two-thirds of daily protein requirements and help prevent protein malnutrition, rebuild lean body mass and assist with enzyme production. The high protein content of FMM can provide the equivalent of 143·4 % RNI per serving for a 70 kg man and 286·8 % of RNI in two servings per day. In a study conducted by Amuna et al. ( Reference Amuna, Zotor and Chinyanga 49 ), it was argued that a 70 kg adult HIV-infected male can meet at least 52·4 % of his daily energy requirement by consuming a 300 g serving of FMM. Regarding energy requirements, two servings per day of FMM can provide between 9·56 MJ (2293 kcal) and 10·06 MJ (2413 kcal), which is equivalent to daily requirements. This can further be enriched by adding vegetable oils (rich in n-3 and n-6 unsaturated fatty acids) and sugar where required. A typical high-energy FMM with energy density between 15·93 and 16·76 J/g (3·82 and 4·02 kcal/g) may thus offset the weight loss resulting from abnormally excessive cytokine production, e.g. TNF-α and IL-1( Reference Amuna, Zotor and Chinyanga 49 ).
The use of plant sources of oils, including nuts in FMM provide n-3 and n-6 unsaturated fatty acids. The inclusion of saturated fat sources is avoided as hypertriglyceridaemia has been observed in HIV patients, which can be exacerbated by ART( Reference Hellerstein, Wu and McGrath 50 ). Preliminary results obtained in the use of FMM concept is encouraging for its application among HIV-infected individuals and warrants further investigation. Furthermore, as most areas heavily affected by HIV are resource poor, it is anticipated that a food-based approach would acquire sustainability over time should locally based foods be utilised.
Conclusion
The nutritional and metabolic challenges of HIV/AIDS and other cachectic conditions are of increasing scientific interest. ART and other pharmacological approaches to management continue to improve prognosis and the quality of life. However, the present review has shown that these have limited success in the absence of adequate nutritional support. Nutritional status and weight loss have been a major prognostic index of survival particularly in developing countries( Reference Sicotte, Langlois and Aho 51 ). In countries where ART is both unavailable and inaccessible due to cost, emphasis on using foods with functional properties (nutraceuticals) as a management tool is warranted.
The present review demonstrates an overall consensus that food-based intervention can play a supportive role in overall weight gain and improving ART adherence. Although BMI is seen as an important indicator of nutritional recovery, measures such as body protein repletion, increase in muscle strength, clinical improvements in appetite and a reduced frequency of opportunistic infections can further strengthen the argument that nutritional intervention is integral to HIV palliative care. However, the biochemical processes that occur during HIV infection require a more thorough investigation. In the studies included in this review, biochemical data such as micronutrient status and lipid peroxidation were not included in either baseline or outcome measurements. In addition, as all but one intervention were based on imported food interventions, supplementation did not reflect the local diet of participants. Studies involving locally produced food supplements, a longer follow-up time and biochemical measurements may provide more substantial information on the efficacy of nutrition intervention. The inclusion of locally/community-grown crops in therapeutic food production may considerably reduce both ingredient and transport costs, as well as introduce a market for small-scale community farmers. Support from the government and non-governmental organisations can also assist in improving patient access to therapeutic foods. In addition, further investigation is needed into the clinical efficacy of therapeutic food formulas (including the FMM approach) given to HIV patients.
Acknowledgements
Thanks are due to the authors who contributed to this paper, as well as to all researchers working in the field of HIV and food security.
Financial Support
None.
Conflicts of Interest
None.
Authorship
K. A. conducted the study search and results analysis, as well as contributed background information on food insecurity, HIV, ART and therapeutic foods in the introduction. F. Z. and P. A. contributed information on the FMM concept and background information on the biochemical relationship between food and HIV. B. E. coordinated the review and contributed by editing and preparing the paper for submission.





