Cognitive impairment poses a major global public health challenge due to increasing prevalence in line with population ageing( Reference Peracino and Pecorelli 1 ). The transition from mild cognitive impairment (MCI) through to the various forms of dementia, such as Alzheimer’s disease (AD), is one of the costliest burdens on health service delivery( Reference Deary, Corley and Gow 2 ). The National Institute for Aging-Alzheimer’s Association (NIA-AA) developed core clinical criteria to inform the diagnosis of MCI( Reference Albert, DeKosky and Dickson 3 ). This identifies that a person with MCI should display a change in cognition, expressed through personal concern or identification from a physician. In addition, individuals should display a lower performance in at least one cognitive domain than that expected for their age and education, over a period of time. Such domains are memory, executive function, attention, visuospatial skills and language. Finally, individuals with MCI may have slight problems with complex daily tasks, however, generally live an independent lifestyle with minimal assistance( Reference Albert, DeKosky and Dickson 3 ). MCI is described as a transitional stage between the expected cognitive decline of normal ageing and that of dementia( Reference Roberts and Knopman 4 ). Furthermore, it has been estimated that 46 % of MCI patients develop dementia within 3 years from diagnosis( Reference Tschanz, Welsh-Bohmer and Lyketsos 5 ). Therefore, it is critical to identify effective interventions that can protect against cognitive decline in this vulnerable high risk group( Reference Martin, Clare and Altgassen 6 ).
Despite pharmacological advances, there are no effective treatments to delay or reverse cognitive impairment. The inflammatory mechanisms and oxidative stress involved in the aetiology of cognitive decline and dementia( Reference Cherbuin, Anstey and Baune 7 ), indicates a potential role for nutrition in its prevention( Reference Alles, Samieri and Feart 8 ). Furthermore, processes such as neurogenesis and neuronal connectivity involved in the function of the brain are influenced by dietary components( Reference Dauncey 9 , Reference Gomez-Pinilla and Tyagi 10 ). The role of nutrition in cognitive health outcomes has been examined in terms of a range of nutrients/dietary patterns, investigating the role that single nutrients, such as n-3 PUFA( Reference Cherbuin, Anstey and Baune 7 ), as well as whole foods/diet interventions, such as the Dietary Approaches to Stop Hypertension (DASH) diet( Reference Smith, Blumenthal and Babyak 11 ), a ketogenic diet( Reference Krikorian, Shidler and Dangelo 12 ) or the Mediterranean diet( Reference Valls-Pedret, Sala-Vila and Serra-Mir 13 ) may have, particularly in relation to their effect on reducing inflammation and oxidative stress( Reference Frisardi, Panza and Seripa 14 – Reference Knight, Bryan and Murphy 16 ). It has been suggested that, although investigations into single nutrients have importance from a mechanistic point of view, studies which provide whole-diet analysis acknowledge that, in everyday situations, foods are consumed in complex combinations and may be a more representative approach to measure the effect of diet on cognition( Reference Milte and McNaughton 17 ). Furthermore, ensuring older adults with MCI stay physically active could have beneficial effects on cognition( Reference Nagamatsu, Chan and Davis 18 , Reference Lautenschlager, Cox and Flicker 19 ), alongside engaging in cognitive training strategies to boost cognitive function. This involves a variety of either computerised or hand-written techniques to enhance memory, language and attention( Reference Li, Li and Li 20 ). However, the available research in this area is variable, with a lack of specific studies in MCI( Reference Martin, Clare and Altgassen 6 ).
Ultimately, there is a need for this systematic review to examine what is known to date about the role of diet on cognitive health, either independently or in conjunction with other lifestyle modifications, specifically in a MCI population. To our knowledge, the effect of dietary interventions on cognitive health outcomes, particularly in high risk populations, like MCI has not been previously systematically reviewed and therefore this has the potential to establish the evidence base for possible management strategies and also define the scope for future research, if required. Thus, the aim of this systematic review was to examine the effect of diet, either alone or in combination with lifestyle and/or cognitive strategies, on cognitive health outcomes in patients with MCI.
Methods
The methods for this systematic review were based on the Centre for Reviews and Dissemination guidance for undertaking systematic reviews in health care( 21 ) and the review protocol was registered with PROSPERO (PROSPERO 2017:CRD42017067267). To be included in this review, the article had to be a randomised controlled trial (RCT) design, conducted in patients with MCI and with diet as the main focus of the intervention. Pilot studies were excluded when a paper clearly stated that the research was a ‘pilot study’. Interventions could focus on diet alone (a dietary pattern or dietary supplements) or in combination with lifestyle and/or cognitive strategies. An overview of the inclusion and exclusion criteria is provided in Table 1. Incident dementia or AD was the primary outcome measure. Secondary outcomes included overall cognitive function or specific cognitive domains such as memory, executive function, language, attention or visuospatial skills measured using validated neuropsychological tests, for example, Mini Mental State Examination (MMSE), Cambridge Cognition Examination or Repeatable Battery for the Assessment of Neuropsychological Status.
Table 1 An overview of the inclusion and exclusion criteria for this systematic review
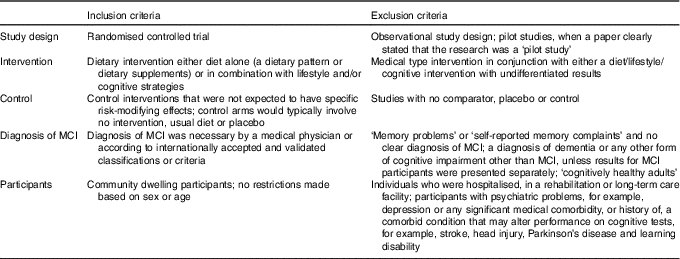
MCI, mild cognitive impairment.
Study identification
A comprehensive literature search was undertaken in June 2016 using Ovid MEDLINE, EMBASE, PsycINFO, Web of Science and Scopus. A suitable search strategy was devised considering key terms used in associated reviews relating to ‘diet’, ‘lifestyle’, ‘cognitive strategies’, ‘cognition’ and ‘behaviour change’. Studies were restricted to English Language and similar search terms were used in each database. This detailed search strategy was developed in Ovid MEDLINE (online Supplementary Table S1) and this strategy was tailored for the other databases. The literature search was repeated in November 2016 and March 2018 to identify new publications. The reference lists of articles and other relevant systematic reviews were screened for potential trials not identified by the electronic search.
Data extraction
Titles and abstracts of potentially eligible studies were screened by the first author (A. M. M.). Any articles not meeting the inclusion criteria were excluded at this stage. Full text articles were obtained for the remaining studies and the study methodology was further assessed for eligibility (A. M. M.). Any queries with regards to inclusion of articles were discussed among the research team (C. T. M., J. V. W., B. M. and M. C. M.). A data extraction form was generated to summarise the key characteristics of the included articles, extracting information on participant, intervention, and methodological characteristics and cognitive outcome results. Data was extracted for the primary and secondary outcomes as stated previously. Information on quality of life and number of participants experiencing one or more serious adverse events was also extracted when provided in papers in addition to the primary and secondary outcomes mentioned. Where studies included validated biomarkers (e.g. structural MRI or amyloid imaging) secondary to cognitive outcome measures, these data were also extracted. The extraction was undertaken by the first author (A. M. M.) and this was independently checked by the second author (C. T. M.) and both reviewers discussed any discrepancies as required.
Quality assessment
The methodological quality of the included studies was assessed using the Jadad scale( Reference Jadad, Moore and Carroll 22 ). This scale has been widely used to assess the quality of RCT included in systematic reviews with regards to randomisation procedures, double blinding and participant withdrawals. A score of 1 was allocated for each ‘yes’ answer to the following three questions:
(1) Was the study described as randomised?
(2) Was the study described as double blind?
(3) Was there a description of withdrawals and drop outs?
An additional score of 1 was awarded if;
(4) The randomisation process was described and appropriate
(5) The method of double blinding was described and appropriate.
The maximum possible score was 5( Reference Jadad, Moore and Carroll 22 ).
The risk of bias was assessed using the Cochrane classification( Reference Higgins and Green 23 ). Each study was assessed for the following (where appropriate): (1) selection bias; (2) performance bias; (3) detection bias; (4) attrition bias and (5) reporting bias. Individual studies were assessed as either low, high or uncertain risk for the adequacy of the stated variables.
Data analysis
The data collected were expected to display a high degree of heterogeneity, therefore quantitative synthesis was unsuitable. The results were summarised using narrative synthesis and presented in tables.
Results
The systematic search in June 2016 generated a total of 2130 articles (2108 through database searches and twenty-two through searches of reference lists). Following the removal of 650 duplicates, 1480 articles were screened for eligibility by examining their titles and abstracts. This process excluded 1447 studies and the full texts of thirty-three papers were obtained; twenty-two articles were excluded for the reasons outlined in Fig. 1. Following a second (November 2016) and third (March 2018) literature search, five further studies were identified that met the inclusion criteria and so sixteen studies were included. As per the review protocol, the results have been displayed according to the primary (incident dementia or AD) and secondary (cognitive function) outcomes. For cognitive function, as per the NIA-AA criteria for the diagnosis of MCI( Reference Albert, DeKosky and Dickson 3 ), the results were grouped according to the following cognitive domains: (1) memory; (2) executive function; (3) attention; (4) language and (5) visuospatial skills, with an additional section reporting global cognitive function. When papers did not specify the cognitive domain measured, the results were grouped under ‘additional cognitive function measures’ (online Supplementary Table S2). A descriptive list of the most frequently reported cognitive function tests used in the studies is provided in the online Supplementary material.

Fig. 1 Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) flow diagram. MCI, mild cognitive impairment.
Study characteristics
An overview of the study characteristics is shown in Table 2. Of the sixteen studies included in analysis, thirteen studies used dietary supplements or single foods as their diet intervention, including folic acid( Reference Ma, Wu and Zhao 24 ), vitamin B combination (folic acid, vitamin B12 and vitamin B6)( Reference de Jager, Oulhaj and Jacoby 25 ), Gingko biloba ( Reference DeKosky, Williamson and Fitzpatrick 26 ), n-3 fatty acids (DHA+EPA( Reference Lee, Shahar and Chin 27 – Reference Phillips, Childs and Calder 29 ) and DHA( Reference Zhang, Miao and LI 30 )), vitamin E( Reference Petersen, Thomas and Grundman 31 ), Cr supplementation( Reference Krikorian, Eliassen and Boespflug 32 ), the medical food, Souvenaid containing the specific nutrition combination Fortasyn Connect( Reference Soininen, Solomon and Visser 33 ), cocoa flavanols( Reference Desideri, Kwik-Uribe and Grassi 34 ), concord grape juice( Reference Krikorian, Nash and Shidler 35 ) and wild blueberry juice( Reference Krikorian, Shidler and Nash 36 ). The three remaining studies focused their interventions on nutritional counselling in combination with healthy eating advice and energy restriction( Reference Horie, Serrao and Simon 37 ), high-saturated fat/high-glycaemic index (GI) diet v. a low-saturated fat/low-GI diet( Reference Bayer-Carter, Green and Montine 38 ) and a high carbohydrate v. a very low carbohydrate diet( Reference Krikorian, Shidler and Dangelo 12 ). A figure detailing the included studies and their dietary exposure linked to the cognitive outcome measures assessed is provided in the online Supplementary material (Fig. 1). One study( Reference Horie, Serrao and Simon 37 ) encouraged both intervention and control participants to partake in physical activity (150 min/week) as per World Health Organization( 39 ) recommendations. There were no studies which included cognitive strategies as part of their intervention. Furthermore, two studies stated that participants had amnesic mild cognitive impairment (aMCI)( Reference Bayer-Carter, Green and Montine 38 ) or prodromal AD( Reference Soininen, Solomon and Visser 33 ) while all other studies reported a diagnosis of MCI.
Table 2 Overview of study characteristics

MCI, mild cognitive impairment; aMCI, amnesic mild cognitive impairment; GI, glycaemic index; AD, Alzheimer’s disease; RAVLT, Rey Auditory Verbal Learning Test; CDR, clinical dementia rating; V-PAL, verbal paired associates learning; MMSE, Mini Mental State Examination; HVLT-R, Hopkins Verbal Learning Test – revised; CERAD, Consortium to Establish a Registry for Alzheimer’s Disease; CLOX, executive clock drawing task; WAIS-RC, Wechsler Adult Intelligence Scale Revised; ADAS-Cog, Alzheimer’s Disease Assessment Scale cognitive assessment; CDR-SB, clinical dementia rating-sum of boxes; CVLT, California Verbal Learning Test; LDST, letter digit substitution test; NTB, neuropsychological test battery.
Primary outcome measure – incident dementia and Alzheimer’s disease
In all, three of the included studies had an outcome measure of incident dementia and/or AD( Reference DeKosky, Williamson and Fitzpatrick 26 , Reference Petersen, Thomas and Grundman 31 , Reference Soininen, Solomon and Visser 33 ). Vitamin E supplementation over 3 years showed no significant difference in the diagnostic rate of AD in participants with MCI taking vitamin E (2000 IU) v. placebo (hazard ratio (HR) 1·02, 95 % CI 0·57, 1·13)( Reference Petersen, Thomas and Grundman 31 ). In the vitamin E group, 33/257 (13 %) and 38/259 (15 %) participants in the placebo group progressed to possible or probable AD in the first 12 months (relative risk (RR) 1·02, 95 % CI 0·96, 1·10). At 36 months, 76/257 (30 %) in the vitamin E group and 73/259 (28 %) in the placebo had progressed to AD (RR 1·03, 95 % CI 0·79, 1·35)( Reference Petersen, Thomas and Grundman 31 ). Likewise, a USA based study with intervention follow up over 6·1 years and found no significant difference between G. biloba v. placebo for the outcomes of all dementia (9·82/100 person-years v. 8·68/100 person-years, HR 1·13, 95 % CI 0·85, 1·50), AD without vascular dementia (VaD) (7·02/100 person-years v. 6·09/100 person-years, HR 1·15, 95 % CI 0·83, 1·61), AD with VaD (2·10/100 person-years v. 2·20/100 person years, HR 0·96, 95 % CI 0·54, 1·71), total AD (9·12/100 person-years v. 8·28/100 person-years, HR 1·10, 95 % CI 0·83, 1·47) and VaD without AD (0·18/100 person-years v. 0·30/100 person-years, HR 0·59, 95 % CI 0·10, 3·51)( Reference DeKosky, Williamson and Fitzpatrick 26 ). Finally, supplementation with Souvenaid (125 ml/d of the specific nutrition combination Fortasyn Connect) v. control, showed no statistically significant difference in diagnosis of dementia at 24 months between groups (59/158 (37 %) (control) v. 62/153 (41 %) (intervention))( Reference Soininen, Solomon and Visser 33 ).
Secondary outcome measure-cognitive function
Memory
As shown in Table 3, there were twenty-five cognitive tests used to measure the domain of memory, and it was assessed in fifteen out of the sixteen studies (94 %) and hence was the most tested cognitive domain. Overall, nine out of the fifteen studies (53 %) (B vitamin( Reference de Jager, Oulhaj and Jacoby 25 ), DHA+EPA( Reference Lee, Shahar and Chin 27 – Reference Phillips, Childs and Calder 29 ), DHA( Reference Zhang, Miao and LI 30 ), vitamin E( Reference Petersen, Thomas and Grundman 31 ), cocoa flavonols( Reference Desideri, Kwik-Uribe and Grassi 34 ), concord grape juice( Reference Krikorian, Nash and Shidler 35 ) and wild blueberry juice( Reference Krikorian, Shidler and Nash 36 )) showed a significant difference between groups at study completion in at least one cognitive function test measuring memory. Fish oil supplementation (3×430 mg DHA+150 mg EPA daily for 12 months), produced significant improvements in visual reproduction I and Rey Auditory Verbal Learning Test delayed recall v. placebo group (all P<0·05)( Reference Lee, Shahar and Chin 27 ). In addition, there was a significant improvement in memory performance (cognitive Z score) in the fish oil v. placebo group (P=0·001)( Reference Lee, Shahar and Chin 27 ). In a second study investigating n-3 PUFA supplementation (480 mg DHA+720 mg EPA daily for 6 months v. placebo)( Reference Bo, Zhang and Wang 28 ), borderline statistical significance (P=0·047) was reported between intervention and control for working memory. However, a third study investigating 625 mg EPA+600 mg DHA v. placebo showed no significant improvements in memory( Reference Phillips, Childs and Calder 29 ). A fourth study who investigated DHA supplementation only (2 mg/d v. placebo)( Reference Zhang, Miao and LI 30 ), found significant improvements for short-term memory (P≤0·0001) and long-term memory (P≤0·0001) in comparison to the placebo group. In a trial investigating the effect of cocoa flavanols (high flavonols (HF) 990 mg v. intermediate flavonols (IF) 520 mg v. low flavonols (LF) 45 mg of flavanols daily for 8 weeks)( Reference Desideri, Kwik-Uribe and Grassi 34 ), verbal fluency test scores significantly improved (P=0·0001), with a significantly greater score in HF participants in comparison with the LF group (P≤0·05).
Table 3 Summary table of cognitive function results grouped as per National Institute for Aging-Alzheimer’s Association (NIA-AA)(3) criteria

RAVLT, Rey Auditory Verbal Learning Test; HVLT-R, Hopkins Verbal Learning Test – Revised; tHcy, total homocysteine; GI, glycaemic index; aMCI, amnesic mild cognitive impairment; V-PAL, Verbal Paired Associates Learning; CVLT, California Verbal Learning Test; CDT, clock drawing test; NTB, neuropsychological test battery; CLOX, executive clock drawing task. * Statistically significant difference (P≤0·05) between intervention and control groups at study completion.
† Statistically significant difference P≤0·05 within group.
‡ No statistically significant difference between intervention and control at study completion.
§ Statistically significant difference between intervention and control at stated time-point.
*** Statistically significant difference P≤0·001 within group.
B vitamin supplementation( Reference de Jager, Oulhaj and Jacoby 25 ) (0·8 mg folic acid, 0·5 mg vitamin B12, 20 mg vitamin B6 daily for 2 years), demonstrated improvement in verbal memory but only in those participants with low baseline B vitamin/folic acid status. The odds of correctly remembering a word in the Hopkins Verbal Learning Test were 69 % greater for a person in the high total homocysteine (tHcy) group if they were taking B vitamins, than if they were taking placebo (OR=1·69, P=0·001)( Reference de Jager, Oulhaj and Jacoby 25 ). For category fluency (Consortium to Establish a Registry for Alzheimer’s Disease), in the high tHcy group, the average number of words was 9·4 % greater at follow up in those on B vitamin treatment compared with the placebo (P=0·04). However, in the low tHcy group (indicating higher B vitamin/folic acid status) there was no significant difference between the treatment group and placebo( Reference de Jager, Oulhaj and Jacoby 25 ). In another B vitamin study, investigating folic acid alone (400 μg daily for 6 months) v. conventional treatment( Reference Ma, Wu and Zhao 24 ) results showed for short term memory that the intervention group had a significant increase in score from baseline to 6 months in comparison to the control (P≤0·001). Results also indicated that elevated homocysteine levels at baseline were associated with significantly poorer cognitive performance at intervention completion for the intervention group in comparison to the control( Reference Ma, Wu and Zhao 24 ).
Vitamin E supplementation (2000 IU daily for 2 years)( Reference Petersen, Thomas and Grundman 31 ), the medical food, Souvenaid containing the specific nutrition combination Fortasyn Connect (125 ml daily)( Reference Soininen, Solomon and Visser 33 ) and chromium picolinate (CrPic) supplementation (1000 μg daily for 12 weeks)( Reference Krikorian, Eliassen and Boespflug 32 ) had no significant improvement in comparison to placebo for memory. Supplementation with CrPic showed significantly reduced intrusion errors, with the intervention group making significantly fewer errors on California Verbal Learning Test (CVLT) for learning (P=0·01) than the placebo group, however there was no significant reduction for recall and recognition memory( Reference Krikorian, Eliassen and Boespflug 32 ). In an investigation of the effects of a high carbohydrate diet (50 % of total energy content) v. a very low carbohydrate (5–10 % of total energy content) diet in participants with MCI( Reference Krikorian, Shidler and Dangelo 12 ), pre-intervention carbohydrate levels were recorded as 207 g for those in the ‘high’ carbohydrate group and 190 g in the ‘low’ carbohydrate group. Post-intervention carbohydrate levels measured 197 g for the ‘high’ carbohydrate group and 34 g for the ‘low’ carbohydrate group. These figures indicate that those in the ‘low’ group had a major dietary change whereas the ‘high’ group could be regarded as a control. Results showed no significant effect of the intervention for memory performance (brief visuospatial memory test, story recall and word list) between intervention and control groups( Reference Krikorian, Shidler and Dangelo 12 ). Concord grape juice( Reference Krikorian, Nash and Shidler 35 ) (daily consumption between 6 and 9 ml/kg for 12 weeks) significantly improved verbal learning compared with the placebo (P=0·04). However, there were no significant differences between those consuming the grape juice and placebo for delayed verbal recall and spatial memory( Reference Krikorian, Nash and Shidler 35 ). Furthermore, wild blueberry juice( Reference Krikorian, Shidler and Nash 36 ) (daily consumption between 6 and 9 ml/kg for 12 weeks) had a significant improvement from baseline score to 12 weeks for verbal paired associates learning (V-PAL) cumulative learning (P=0·009). In addition, mean scores for CVLT word list recall improved significantly within the intervention group from baseline to 12 weeks (P=0·04). There was a significant difference in V-PAL score between intervention and control groups (P=0·03), however no significant difference was observed for CVLT performance between groups( Reference Krikorian, Shidler and Nash 36 ).
Executive function
The domain of executive function was measured by twelve tests (Table 3). For this cognitive domain, measured within nine studies (56 %), two RCT showed a statistically significant improvement between groups at study completion( Reference de Jager, Oulhaj and Jacoby 25 , Reference Desideri, Kwik-Uribe and Grassi 34 ). At 24 months follow-up, the odds of a correctly drawn item from CLOX1 (an executive clock drawing task), after controlling for confounders (CLOX2 at follow-up, CLOX1 at baseline, age, education, ApoE ε4 status and sex), was 30 % greater in those receiving B-vitamins v. placebo (P=0·02)( Reference de Jager, Oulhaj and Jacoby 25 ). For cocoa flavonol supplementation( Reference Desideri, Kwik-Uribe and Grassi 34 ), better scores for trail making test, part B (P≤0·05) were reported among participants who received HF and IF treatments v. the LF group. In addition, the time required to complete the trail making task, B significantly changed during the duration of the study (P≤0·0001). However, DHA+EPA supplementation( Reference Lee, Shahar and Chin 27 , Reference Phillips, Childs and Calder 29 ), nutritional counselling with energy restriction( Reference Horie, Serrao and Simon 37 ), high fat/high GI v. low fat/low GI diet( Reference Bayer-Carter, Green and Montine 38 ), high carbohydrate v. low carbohydrate diet( Reference Krikorian, Shidler and Dangelo 12 ), supplementation with Fortasyn Connect (Souvenaid)( Reference Soininen, Solomon and Visser 33 ) and vitamin E( Reference Petersen, Thomas and Grundman 31 ) showed no significant difference in cognitive function tests between groups at study completion. There was a significant improvement in comparison with placebo at 6 months for those consuming vitamin E supplements (P<0·05)( Reference Petersen, Thomas and Grundman 31 ). However, thereafter, this significant difference was not maintained beyond this time point.
Attention
As shown in Table 3, five of the sixteen (31 %) included studies measured the domain of attention. Nutritional counselling v. standard care showed no significant change in attention between groups after 12 months( Reference Horie, Serrao and Simon 37 ). Whereas, cocoa flavonol supplementation( Reference Desideri, Kwik-Uribe and Grassi 34 ), significantly better scores for trail making test, part A (P≤0·05) were reported among participants who received HF and IF treatments in comparison to the LF group. In addition, the time required to complete the trail making task, part A significantly changed during the duration of the study (P≤0·0001)( Reference Desideri, Kwik-Uribe and Grassi 34 ). DHA+EPA supplementation( Reference Lee, Shahar and Chin 27 ) (one study) showed a significant improvement in digit span score from baseline to 12 months in the fish oil group v. placebo (P≤0·0001)( Reference Lee, Shahar and Chin 27 ). However, there was no significant treatment effect reported between the fish oil and placebo groups for any of the other measures of attention( Reference Lee, Shahar and Chin 27 ). Supplementation with DHA only( Reference Zhang, Miao and LI 30 ) showed significant improvements in digit span score in comparison to the placebo (P≤0·0001). However, a third study with DHA+EPA supplementation( Reference Phillips, Childs and Calder 29 ) found no significant differences between groups for attention.
Language
In all, two of the sixteen (13 %) studies measured the cognitive domain of language (Table 3). There were no significant differences between groups for nutritional counselling with energy restriction( Reference Horie, Serrao and Simon 37 ). For vitamin E supplementation( Reference Petersen, Thomas and Grundman 31 ), there was a significant difference in score from the baseline value between groups at 6 months (P≤0·05), 12 months (P≤0·05) and 18 months (P≤0·05), however, thereafter this significant difference was not maintained until intervention completion (36 months)( Reference Petersen, Thomas and Grundman 31 ).
Visuospatial skills
In all, four studies (25 %) measured the cognitive domain of visuospatial skills (Table 3). Supplementation with folic acid was the only study to show a significant interaction effect between groups for visuospatial skills (P=0·03)( Reference Ma, Wu and Zhao 24 ). In addition, higher baseline homocysteine levels were associated with poorer cognitive performance on the block design test at the end of the intervention in comparison with the placebo (estimate value=−0·079, P≤0·001)( Reference Ma, Wu and Zhao 24 ). Fish oil supplementation with concentrated DHA+EPA( Reference Lee, Shahar and Chin 27 ), DHA( Reference Zhang, Miao and LI 30 ) or vitamin E supplementation( Reference Petersen, Thomas and Grundman 31 ) did not show any significant differences between groups.
Global cognitive function
For cocoa flavonol supplementation( Reference Desideri, Kwik-Uribe and Grassi 34 ) (online Supplementary Table S2), there was no significant change in MMSE score between the HF, IF or LF treatment groups over the duration of the study (P=0·13). However, results also showed that the composite cognitive Z score significantly changed during the study (P≤0·0001). The cognitive Z score at the end of the study follow-up was significantly (P≤0·05) better in the HF group in comparison to the LF group( Reference Desideri, Kwik-Uribe and Grassi 34 ). Vitamin B supplementation( Reference de Jager, Oulhaj and Jacoby 25 ) indicated no significant effect of treatment (P=0·57) on global cognition as measured by MMSE. However, analysis did show that those who had high baseline concentrations of homocysteine and were treated with B vitamins, were 1·58 more likely to provide a correct answer on the MMSE test than the placebo group (P<0·001). However, there was no significant difference for those with low baseline homocysteine, between the B vitamin or placebo groups. Similarly, fish oil supplementation( Reference Lee, Shahar and Chin 27 ) (one study) showed no statistically significant differences between groups for cognitive function as measured by the MMSE. Furthermore, vitamin E supplementation( Reference Petersen, Thomas and Grundman 31 ) at 6 months intervention showed a significant difference in comparison with placebo for overall cognitive function calculated by a composite Z score (P≤0·01). However, at 36 months this significant difference between groups was not maintained.
Assessment of methodological quality and risk of bias
The quality( Reference Jadad, Moore and Carroll 22 ) of the sixteen included studies varied, with eight studies achieving the maximum total score of 5( Reference de Jager, Oulhaj and Jacoby 25 – Reference Bo, Zhang and Wang 28 , Reference Zhang, Miao and LI 30 , Reference Petersen, Thomas and Grundman 31 , Reference Soininen, Solomon and Visser 33 , Reference Desideri, Kwik-Uribe and Grassi 34 ) (online Supplementary Table S3). Thus, it was deemed that these studies stated appropriate randomisation processes, were clearly indicated as double blinded and the authors accounted for any participant withdrawals during the study. In all, two studies( Reference Krikorian, Shidler and Dangelo 12 , Reference Bayer-Carter, Green and Montine 38 ) scored one on the Jadad scale( Reference Jadad, Moore and Carroll 22 ) and stated that participants were randomised however did not specify the randomisation process, if double-blinding took place and if any participant withdrawals occurred. Low risk of bias scores( Reference Higgins and Green 23 ) were allocated for selection bias (n 9)( Reference Ma, Wu and Zhao 24 – Reference Bo, Zhang and Wang 28 , Reference Zhang, Miao and LI 30 , Reference Petersen, Thomas and Grundman 31 , Reference Soininen, Solomon and Visser 33 , Reference Desideri, Kwik-Uribe and Grassi 34 ), performance bias (n 7)( Reference de Jager, Oulhaj and Jacoby 25 , Reference DeKosky, Williamson and Fitzpatrick 26 , Reference Bo, Zhang and Wang 28 – Reference Zhang, Miao and LI 30 , Reference Soininen, Solomon and Visser 33 , Reference Desideri, Kwik-Uribe and Grassi 34 ), attrition (n 9)( Reference Ma, Wu and Zhao 24 , Reference de Jager, Oulhaj and Jacoby 25 , Reference Lee, Shahar and Chin 27 – Reference Zhang, Miao and LI 30 , Reference Soininen, Solomon and Visser 33 , Reference Desideri, Kwik-Uribe and Grassi 34 , Reference Horie, Serrao and Simon 37 ) and detection bias (n 6)( Reference Ma, Wu and Zhao 24 , Reference DeKosky, Williamson and Fitzpatrick 26 , Reference Zhang, Miao and LI 30 , Reference Soininen, Solomon and Visser 33 , Reference Desideri, Kwik-Uribe and Grassi 34 , Reference Horie, Serrao and Simon 37 ) (online Supplementary Table S4). A high risk score was documented for detection bias (n 3)( Reference Krikorian, Shidler and Dangelo 12 , Reference Bayer-Carter, Green and Montine 38 ) and performance bias (n 2)( Reference Krikorian, Shidler and Dangelo 12 ) as there were no details provided of any double blinding method used.
Discussion
The aim of the present systematic review was to examine the effect of diet, either alone or in combination with lifestyle and/or cognitive strategies, on cognitive health outcomes in patients with MCI. Together with the limited number of RCT conducted and the heterogeneity of the studies in this review, a narrative synthesis of the findings was implemented. Studies varied greatly in terms of the nature of dietary intervention and cognitive outcome measures used. Furthermore, there were no studies that measured the effectiveness of lifestyle and/or cognitive strategies in combination with their dietary intervention. Overall, it was evident that the findings were inconsistent across the studies and do not provide clear evidence to support the effect of any specific diet or dietary component on cognition in MCI patients.
Diet has been suggested to have a significant association with cognitive decline and progression to dementia, particularly showing a protective role against the harmful effects of neuro-inflammation and oxidative stress( Reference Tang, Harrison and Albanese 40 ). Although the pathways related to their role are complex and variable throughout the literature( Reference Frisardi, Panza and Seripa 14 – Reference Knight, Bryan and Murphy 16 , Reference Smith and Blumenthal 41 ) it is thought that antioxidants in foods such as fruit and vegetables help to reduce oxidative stress levels in the brain and n-3 PUFA in foods such as oily fish, are additionally linked to reduced inflammation( Reference Alles, Samieri and Feart 8 ). There are plausible suggestions to support these mechanisms by the results of this review. There were some improvements in cognitive function, particularly in the domain of memory, reported for polyphenol compounds (e.g. cocoa flavonols( Reference Desideri, Kwik-Uribe and Grassi 34 )), fish oil supplementation with concentrated DHA+EPA( Reference Lee, Shahar and Chin 27 , Reference Bo, Zhang and Wang 28 ) or DHA alone( Reference Zhang, Miao and LI 30 ) and beverages which are high in these bioactive, antioxidant properties e.g. concord grape juice( Reference Krikorian, Nash and Shidler 35 ) and wild blueberry juice( Reference Krikorian, Shidler and Nash 36 ). However, some of these studies either had small, potentially underpowered sample sizes, used a limited number of cognitive tests to measure outcomes or had shorter intervention durations therefore these results should be interpreted with caution.
Nutrient and food supplementation
As mentioned, antioxidant compounds such as vitamins A, C and E have a role in regulation of oxidative stress, a pathway linked with neurodegeneration and cognitive decline( Reference Morris 42 ). However in this review, diet supplementation with vitamin E( Reference Petersen, Thomas and Grundman 31 ) had no significant effect on progression from MCI to dementia and/or AD or on cognitive function at intervention completion. Furthermore, meta-analyses have reported no significant effect of vitamin E on cognitive function outcomes( Reference Forbes, Holroyd-Leduc and Poulin 43 , Reference Farina, Llewellyn and Isaac 44 ). The particular form of vitamin E used could have an influence on the impact of this nutritional component on cognitive decline, with research suggesting total tocopherol plasma concentrations rather than single tocopherols may be more valuable at predicting cognitive impairment, particularly AD( Reference Mangialasche, Kivipelto and Mecocci 45 ). Furthermore, as we consume foods in complex patterns, resulting in ingestion of combinations of various forms of vitamin E, it may be more beneficial to focus research efforts away from single forms and follow a more holistic investigation( Reference Caracciolo, Xu and Collins 15 ). In this review, supplementation with cocoa flavonols( Reference Desideri, Kwik-Uribe and Grassi 34 ) showed better cognitive performances for those who received higher flavonols concentrations compared with lower concentrations. There are suggestions in the literature that flavonoids may exert their neuroprotective properties in a similar mechanism to antioxidants in the body( Reference Best and Dye 46 ). However, further indications suggest that flavonoids may have a more prominent role in the regulation of neuronal signalling pathways( Reference Vazour, Vafeiadou and Rodriguez-Mateos 47 ) or neuro-inflammation( Reference Spencer 48 ). It is clear that further research is required to fully explore the mechanism of action of flavonoid compounds and investigate the potential role they may have in protecting against cognitive decline( Reference Bell, Lamport and Butler 49 ).
Low folate and B vitamin status is linked to cognitive dysfunction during the ageing process and better cognitive performances have been associated with higher intakes of B vitamins( Reference Porter, Hoey and Hughes 50 – Reference Tangney, Tang and Evans 52 ). Furthermore, increased levels of homocysteine have been linked to poorer cognition, particularly in memory and attention( Reference Faux, Ellis and Porter 53 – Reference Duthie, Whalley and Collins 55 ). This may be explained by the role that B vitamins have in one-carbon metabolic pathways in the body, acting as co-factors for the remethylation of homocysteine to methionine, producing the methyl-donor, S-adenosylmethionine. This methyl donor has a specific role in the methylation of phospholipids and neurotransmitters in the brain, thus indicating how a depletion in B vitamins status may influence cognitive function and ultimately, cognitive impairment( Reference Moore, O’Shea and Hughes 56 , Reference Smith and Refsum 57 ). In this review, supplementation with a B vitamin combination( Reference de Jager, Oulhaj and Jacoby 25 ) or with folic acid alone( Reference Ma, Wu and Zhao 24 ) had significant effects on executive function( Reference de Jager, Oulhaj and Jacoby 25 ) and furthermore, when baseline homocysteine levels were elevated, there were significant improvements in global cognition( Reference de Jager, Oulhaj and Jacoby 25 ), memory( Reference Ma, Wu and Zhao 24 , Reference de Jager, Oulhaj and Jacoby 25 ) and visuospatial skills( Reference Ma, Wu and Zhao 24 ). In support, not only have improvements been observed in performance based cognitive tests, B vitamin supplementation (folic acid, vitamin B6 and B12 combination) have resulted in reduced rates of brain atrophy in MCI( Reference Smith, Smith and de Jager 58 , Reference Douaud, Refsum and de Jager 59 ); a process which could result in progression to AD if allowed to advance. However, findings are mixed with meta-analyses of clinical trial data reporting no significant effect of B vitamins on cognitive function( Reference Forbes, Holroyd-Leduc and Poulin 43 , Reference Clarke, Bennett and Parish 60 ). Therefore, further trial research is warranted to confirm the role of B vitamins in reducing cognitive decline.
PUFA have been associated with promoting cognitive function, primarily as a result of their anti-inflammatory properties( Reference Gillette-Guyonnet, Secher and Vellas 61 ). Furthermore, n-3 fatty acids, particularly DHA, are a key component of neuronal membranes in the brain, influencing neurogenesis and neuronal function( Reference Smith and Blumenthal 41 , Reference Sydenham, Dangour and Lim 62 ). In this review, supplementation with DHA+EPA( Reference Lee, Shahar and Chin 27 , Reference Bo, Zhang and Wang 28 ) reported significant improvements in the domain of memory, with DHA supplementation alone( Reference Zhang, Miao and LI 30 ) showing an additional improvement in attention, albeit by a single cognitive test. In contrast, evidence from meta-analyses have reported no significant effect of n-3 fatty acids on cognitive outcomes( Reference Forbes, Holroyd-Leduc and Poulin 43 , Reference Sydenham, Dangour and Lim 62 ). Furthermore, it has been suggested that fatty acid supplementation in individuals who are homozygous carriers of the ApoE ε4 allele, a risk factor for cognitive decline, could be resistant from the potential protective effects of fatty acids on cognitive health( Reference Chouinard-Watkins and Plourde 63 ). Thus, this is an important covariate to consider when designing trials to test effectiveness of fatty acid supplementation. However, some observational evidence does exist to support the role of n-3 fatty acids in promoting cognition with a study that followed non-demented participants for 4 years, finding higher plasma EPA concentrations to be associated with a lower incidence of dementia( Reference Samieri, Feart and Letenneur 64 ). In addition, an intervention study with older adults with subjective memory impairment investigated fatty acid supplementation (EPA+DHA) v. maize oil placebo( Reference Boespflug, McNamara and Eliassen 65 ). Results showed significantly improved cortical blood oxygen level-dependent activity during a working memory task in the fish oil group compared with placebo. In this review, one study investigating DHA+EPA supplementation( Reference Phillips, Childs and Calder 29 ) found no effect on cognitive function in comparison to control. A plausible explanation for this finding could be that the placebo used this study was olive oil, a component of the Mediterranean diet associated with improved cognitive function owing to its anti-inflammatory properties( Reference Martinez-Lapiscina, Clavero and Toledo 66 ). Therefore, further investigation of the role of fatty acids and cognitive decline is justified through well-designed, robust studies.
Whole-foods/dietary patterns
Only three of the sixteen studies included in this review( Reference Krikorian, Shidler and Dangelo 12 , Reference Horie, Serrao and Simon 37 , Reference Bayer-Carter, Green and Montine 38 ), focused their diet intervention on ‘whole-foods/dietary patterns’ rather than single-nutrient supplements or single food products. In everyday situations, individuals consume holistic dietary patterns which involve complex interactions between nutrients( Reference Zamroziewicz and Barbey 67 ). It therefore could be suggested that the more representative intervention design to measure the effects of diet on cognition could be that which involved a dietary pattern rather than focused on a single nutrient. In this review, however, these studies were heterogeneous in terms of the dietary intervention and reported mixed findings. Research evidence suggests that ketogenic diets( Reference Lange, Lange and Makulska-Gertruda 68 ) and energy restriction( Reference Gillette-Guyonnet and Vellas 69 ) may have a promising, yet under-investigated, role in AD prevention, suggesting links to brain glucose metabolism( Reference Lange, Lange and Makulska-Gertruda 68 ), reduction in oxidative stress( Reference Gillette-Guyonnet and Vellas 69 ) and anti-inflammatory mechanisms( Reference Gillette-Guyonnet and Vellas 69 ). There is also emerging evidence from observational studies to suggest a protective role for healthy dietary patterns such as the Mediterranean diet on MRI measured brain structures( Reference Luciano, Corley and Cox 70 – Reference Gardener, Scarmeas and Gu 72 ) and therefore further investigation of such dietary patterns is necessary, with the inclusion of more rigorous assessment measures, to help to provide insight into potential mechanisms of how diet can impact brain health.
Use of biomarkers and cognitive markers
CSF biomarkers may be a valuable asset in detecting pathological changes in neurological diseases, owing to the processes of extracellular amyloid-β deposition and accumulation of hyperphosphorylated tau proteins( Reference Forlenza, Diniz and Teixeira 73 ). One study( Reference Bayer-Carter, Green and Montine 38 ) in this review included biomarker analysis in addition to cognitive test measures. Increased concentrations of CSF Aβ42 were observed in those with a MCI consuming a low diet (low saturated fat/low GI) in comparison to healthy controls who observed a decrease in CSF Aβ42 levels (online Supplementary material). Thus, CSF biomarkers in this study changed in response to diet in aMCI patients in the absence of any discernible changes in cognitive function test scores, albeit in very small sample. These differences could provide insights into the mechanisms of action of β-amyloid in the body in cognitive impairment. In particular, biomarker analysis may be more sensitive to dietary changes and could be an important consideration for future dietary intervention studies as the use of biomarkers could be a more rigorous approach to assess cognitive performance in this patient group( Reference Barberger-Gateau 74 ). Furthermore, it has been suggested that the use of brain imaging as a cognitive marker such as MRI scanning is a more robust measure of cognition in comparison to questionnaire based tests( Reference Porter, Hoey and Hughes 50 , Reference de Jager and Kovatcheva 75 ). In all, three studies in this review reported on cognitive marker information, including MRI( Reference Zhang, Miao and LI 30 , Reference Soininen, Solomon and Visser 33 ) and functional MRI imaging( Reference Krikorian, Eliassen and Boespflug 32 ), as an additional outcome measure for cognitive function, depicting some significant interaction effects for the intervention group that were not entirely reflected by cognitive function tests (online Supplementary material). Brain imaging techniques have been used in nutrition and cognition research, with investigations into B vitamins utilising MRI scanning to detect changes in brain atrophy in MCI( Reference Smith, Smith and de Jager 58 , Reference Douaud, Refsum and de Jager 59 ), functional MRI scanning to explore fish oil supplementation in older adults with subjective memory impairment( Reference Boespflug, McNamara and Eliassen 65 ) as well as investigations of β-amyloid load using positron emission tomography (PET) and neuronal activity via PET imaging with 2-[18F]fluoro-2-deoxy-d-glucose( Reference Mosconi and McHugh 76 ). Therefore, the use of these higher quality methods could be implemented in future dietary intervention trials to comprehensively measure the potential effects of diet on cognition and explore mechanisms.
The mixed evidence found on the effect of diet on cognition among MCI participants may be explained by the heterogeneity of studies included, owing to variation in cognitive outcome measures used, differences in the diet intervention type (supplements v. single food products v. dietary patterns), variations in sample size and duration of intervention. Furthermore, the small number of dietary intervention studies conducted among this patient group make it difficult to provide conclusive evidence to support the effect of diet on cognitive outcomes. Of the sixteen included studies, those with B vitamin and/or folic acid supplementation( Reference Ma, Wu and Zhao 24 , Reference de Jager, Oulhaj and Jacoby 25 ), DHA/EPA supplementation( Reference Lee, Shahar and Chin 27 , Reference Bo, Zhang and Wang 28 , Reference Zhang, Miao and LI 30 ) or cocoa flavonol rich drinks( Reference Desideri, Kwik-Uribe and Grassi 34 ) appeared to have the most consistent effects on cognitive outcomes. However, it is difficult to confirm that these dietary interventions are the most effective in terms of promoting cognitive function due to the low number of studies testing the same intervention. Nonetheless, the outcomes of the systematic review highlight the need for well-designed, robust RCT, with pretested and informed methodological characteristics to further explore the role of diet in cognitive decline.
Limitations
During the literature search for this review, a broad search strategy was used to ensure the search covered all related aspects to the reviews aims and objectives. However, search limitations were set to only include studies in English language and the grey literature was not included for this review, therefore this could have resulted in language and publication bias. As RCT were the study design of choice for inclusion, this may have caused selection bias. However, as RCT are considered the best design for assessing the effect of a dietary intervention with their ability to identify causality( Reference Misra 77 ), this therefore provides justification for the decision. Pilot studies were not included in this review, as these studies are likely to have an underpowered sample size. The number of studies included in this review were small, however, as there are few RCT completed in this area, this supports the need for further intervention studies to increase the evidence-base. Due to the heterogeneity of the included studies, the data were not meta-analysed. Instead, a rigorous narrative review was implemented. Study characteristics, such as short study durations, may have not provided sufficient time to view a change in cognitive outcomes. It has been suggested that long term, RCT are the best approach in the design of a nutritional intervention to measure cognitive performance, with estimations that the most effect preventative trials require up to 3–5 years duration and follow-up( Reference Ferry, Coley and Andrieu 78 ). Furthermore, ensuring a sufficient sample size though determination by a power calculation will provide a more stringent approach to the research design. Therefore, it is important when designing intervention studies that duration and sample size are pre-tested, though a feasibility study or by comparison to similar studies in the field.
In all, eight of the sixteen studies in this review achieved the maximum quality score as assessed by the Jadad scale( Reference Jadad, Moore and Carroll 22 ). Those studies who received the lowest scores failed to provide details on the randomisation and blinding processes which took place in the study. It is important to note however, as both studies involved a dietary pattern intervention rather than a supplement/placebo, it is impractical to ensure participants and researchers are blinded to the intervention group. Therefore, the decision that these studies are of ‘low quality’ is difficult to confirm. Furthermore, for risk of bias, a number of studies were allocated uncertain risk for selection, performance, attrition and detection bias due to inadequate information on randomisation, double blinding and/or withdrawals. Finally, a challenge within this review was the heterogeneity of cognitive outcome measures used to determine cognitive change. Some studies grouped results by domain, while others by the single cognitive tests used. This made it difficult when presenting the results of this review, as some study results did not exactly fit within the cognitive domains, as these were not specified in the original paper. In line with the NIA-AA criteria for the diagnosis of MCI( Reference Albert, DeKosky and Dickson 3 ), which state that for a diagnosis of MCI individuals must have deterioration in one or more cognitive domains, it would be beneficial for analysis purposes if future intervention studies could assess cognition based on these domains to allow better comparison of results. However, in saying that, even the tests used to measure cognition within domains vary greatly and there is a lack of standardisation. It is evident therefore, that there is a demand to determine a specialised cognitive test battery that can be used to measure change in cognition, particularly within an MCI population. Furthermore, change in cognition requires time, more rigorous examinations and evaluation by clinical specialist( Reference Katsiardanis, Diamantaras and Dessypris 79 ). These are all important considerations for future intervention trials going forward.
Conclusion
To date there is insufficient RCT evidence on the effect of whole diets or specific dietary components on cognitive outcomes in MCI patients. Existing studies are heterogeneous in terms of the dietary intervention, duration, sample size and cognitive outcome measures assessed, with the most consistent results for cognitive function shown by B vitamins, folic acid, DHA and/or EPA and cocoa flavonol supplementation. Further exploration of the potential beneficial effect of diet on cognitive outcomes in MCI is merited.
Acknowledgements
This research received no specific grant from any funding agency or from commercial or not-for-profit sectors.
The authors’ responsibilities were as follows: A. M. M. had the primary responsibility of writing the manuscript, A. M. M. conducted the literature search; A. M. M. and C. T. M. conducted and reviewed the data extraction; J. V. W., B. M., C. T. M. and M. C. M. reviewed and approved the manuscript. All authors were involved in editing and revisions of the article.
The authors declare no conflicts of interest arising from the conclusions of this work.
Supplementary material
For supplementary material/s referred to in this article, please visit https://doi.org/10.1017/S0007114518002945







