No CrossRef data available.
Published online by Cambridge University Press: 20 September 2019
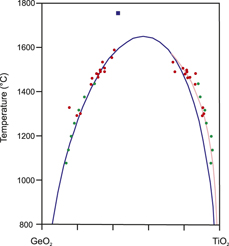
The system GeO2–TiO2 was studied experimentally at high pressure and temperature to measure the miscibility of the two components and to test its applicability as a temperature sensor in high-pressure experiments. Significant solubility between the two end-members was found, with two coexisting solid solutions at high pressure exhibiting mutual solubility that increases with temperature along a solvus. The two solid solution compositions at the solvus can be distinguished readily by X-ray diffraction. At higher temperatures, a complete solid solution exists between the two end-members. The complete solution occurs above a critical line in P–T space (a critical point at each pressure). The critical point is located near 1630 °C and mole fraction  ${X_{{\rm{Ti}}{{\rm{O}}_{\rm{2}}}}} = 0.57$ at 6.6 GPa and changes by 60 ± 5° per GPa in the region from 4 to 7 GPa. A model for the shape of the solvus is developed using X-ray diffraction data points from a series of quench experiments and an in situ experiment, and the model is used to estimate the thermal gradients in a Kawai-type multianvil assembly.
${X_{{\rm{Ti}}{{\rm{O}}_{\rm{2}}}}} = 0.57$ at 6.6 GPa and changes by 60 ± 5° per GPa in the region from 4 to 7 GPa. A model for the shape of the solvus is developed using X-ray diffraction data points from a series of quench experiments and an in situ experiment, and the model is used to estimate the thermal gradients in a Kawai-type multianvil assembly.