No CrossRef data available.
Published online by Cambridge University Press: 16 September 2019
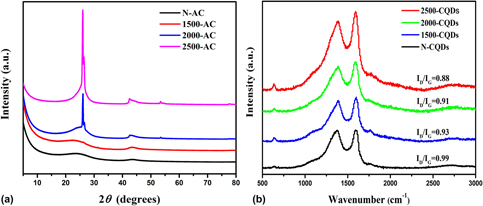
The microstructure of carbon quantum dots (CQDs) has a great influence on their fluorescence properties. Here, different microstructures of CQDs were synthesized by the selective oxidation of graphitized activated carbon using HNO3/HClO4 as the oxidant. We characterized the microstructure and surface chemistry of the CQDs, and the results show that the degree of graphitization of activated carbon has a significant effect on the structure and fluorescence properties of the obtained CQDs. The fluorescence of the CQD solution can be tuned from yellow to green by regulating the degree of graphitization of the activated carbon by heat treatment at high temperature (up to 2500 °C). Moreover, the increased degree of graphitization of the raw carbon precursor is beneficial for significantly reducing the fluorescence self-absorption quenching of the concentrated CQD solution. Importantly, the as-prepared CQDs have no cytotoxicity and can be used as bioimaging agents.