Published online by Cambridge University Press: 21 July 2021
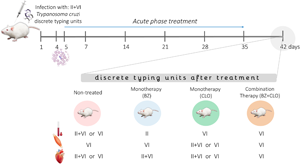
The aim of the present work was to evaluate the distribution of the different clones of the parasite prevailing after treatment with benznidazole (BZ) and clomipramine (CLO), in mice infected with Trypanosoma cruzi, Casibla isolate which consists of a mixture of two discrete typing units (DTUs). Albino Swiss mice were infected and treated with high and low concentrations of BZ (100 or 6.25 mg/kg), CLO (5 or 1.25 mg/kg), or the combination of both low doses (BZ6.25 + CLO1.25), during the acute phase of experimental infection. Treatment efficacy was evaluated by comparing parasitaemia, survival and tissular parasite presence. For DTUs genotyping, blood, skeletal and cardiac muscle samples were analysed by multiplex quantitative polymerase chain reaction. The combined treatment had similar outcomes to BZ6.25; BZ100 was the most effective treatment, but it failed to reach parasite clearance and produced greater histological alterations. Non-treated mice and the ones treated with monotherapies showed both DTUs while BZ6.25 + CLO1.25 treated mice showed only TcVI parasites in all the tissues studied. These findings suggest that the treatment may modify the distribution of infecting DTUs in host tissues. Coinfection with T. cruzi clones belonging to different DTUs reveals a complex scenario for the treatment of Chagas disease and search for new therapies.