Psychotic disorders are associated with multiple social disabilities in work, study, independent living, interpersonal relations and self-care, Reference Wiersma, Wanderling, Dragomirecka, Ganev, Harrison and van der Heiden1–Reference Juckel, Schaub, Fuchs, Naumann, Uhl and Witthaus4 and serious disability in functioning is one of the core features of the DSM–IV diagnosis of schizophrenia. Reference Burns and Patrick2 Disability refers to the limitations in an individual's ability to perform the expected socially and culturally defined roles and tasks. Reference Chwastiak and Von Korff5 In schizophrenia, disability is thought to be mainly associated with negative symptoms and neurocognitive and social cognitive deficits. Reference Gureje, Herrman, Harvey, Morgan and Jablensky6,Reference Cornblatt, Auther, Niendam, Smith, Zinberg and Bearden7
Functional decline has been found to occur before the first psychotic episode. In retrospective studies, Häfner et al Reference Häfner, Nowotny and Löffler8 and Larsen et al Reference Larsen, McGlashan, Johannessen and Vibe-Hansen9 found that deficits were already noticeable in the prodromal and premorbid phase of schizophrenia, respectively. Assessment scales frequently employed in prospective studies and developed for detecting individuals at an ultra-high risk of psychosis include the criterion of impaired functioning connected with genetic risk, as defined by either family history of psychosis or schizotypical personality disorder in the index person. Reference Yung, McGorry, McFarlane, Jackson, Patton and Rakkar10,Reference Miller, McGlashan, Rosen, Somjee, Markovich and Stein11 Preliminary findings from recent studies indicate that decline in functioning may be a predictor of psychosis transition. However, these studies mainly focused on social contacts. Reference Cornblatt, Auther, Niendam, Smith, Zinberg and Bearden7,Reference Häfner, Nowotny and Löffler8,Reference Yung, Buckby, Cotton, Cosgrave, Killackey and Stanford12–Reference Velthorst, Nieman, Becker, van der Fliert, Dingemans and Klaassen14
In the present 18-month follow-up study of a large European population of individuals clinically at high risk of psychosis, disability was investigated in more detail by examining the following six domains: understanding of and interaction with the world; moving and getting around; self-care; getting along with people; life activities; and participation in society. In line with the major focus of this multicentred, prospective naturalistic field study to identify risk factors for the transition from prodrome to a first psychotic episode, Reference Ruhrmann, Schultze-Lutter, Salokangas, Heinimaa, Linszen and Dingemans15 the aim of the present analyses is to investigate: first, differences in disability at baseline between those who do and those who do not make the transition to psychosis in a group at high clinical risk. And second, whether disability is a risk factor for transition to a first psychosis. For exploratory purposes, we further sought to investigate the association between social disability and clinical symptomatology. We hypothesised that the transition group would display significantly greater disability at baseline than the non-transition group.
Method
Recruitment
Between August 2002 and April 2006, data were collected from 245 help-seeking individuals (age 16-35) who met ultra-high-risk and/or ‘cognitive disturbances’ criteria and agreed to participate in the European Prediction of Psychosis Study (EPOS) Reference Ruhrmann, Schultze-Lutter, Salokangas, Heinimaa, Linszen and Dingemans15,Reference Klosterkötter, Ruhrmann, Schultze-Lutter, Salokangas, Linszen and Birchwood16 (Fig. 1). The European Prediction of Psychosis Study is a European collaboration of six centres in four countries: Germany, Finland, The Netherlands and the UK. Referral to the early detection services came from psychiatrists, psychologists, general practitioners, outreach clinics, counselling services, teachers or was self-initiated, among others.
Inclusion criteria comprised ultra-high-risk criteria as assessed by the Structured Interview for Prodromal Syndromes (SIPS 3.0) Reference McGlashan, Miller, Woods, Rosen, Hoffman and Davidson17 and cognitive disturbances as assessed by the Bonn Scale for the Assessment of Basic Symptoms – Prediction List (BSABS–P), an abbreviated version of the Schizophrenia Proneness Instrument, Adult version (SPI–A). Reference Schultze-Lutter, Addington, Ruhrmann and Klosterkötter18
The ultra-high-risk approach consists of three alternative criteria:
-
(a) attenuated psychotic symptoms defined by at least one of the following symptoms with SIPS score ‘moderate’ to ‘severe but not psychotic’ (3–5), appearing several times per week for at least 1 week within the past 3 months: unusual thought content/delusional ideas, suspiciousness/persecutory ideas, grandiosity, perceptual abnormalities/hallucinations, disorganised communication and odd behaviour/appearance;
-
(b) brief limited intermittent psychotic symptoms (BLIPS) defined by hallucinations, delusions or formal thought disorders occurring within the past 3 months and resolving spontaneously within 1 week scoring ‘severe and psychotic’ (6) on the SIPS and achieving at least a ‘moderate’ score on the respective item of the Positive and Negative Syndrome Scale for Schizophrenia; Reference Kay, Fiszbein and Opler19 and
-
(c) genetic risk and functional deterioration defined by a 30% or greater reduction on the Global Assessment of Functioning Scale, modified version (GAF–M), Reference McGlashan, Miller, Woods, Rosen, Hoffman and Davidson17,Reference Hall20,Reference Hall and Parks21 compared with the highest level of previous functioning for at least 1 month within the previous year in combination with a first- or second-degree relative with a history of any DSM–IV psychotic disorder 22 or a DSM–IV schizotypal personality disorder of the index person.
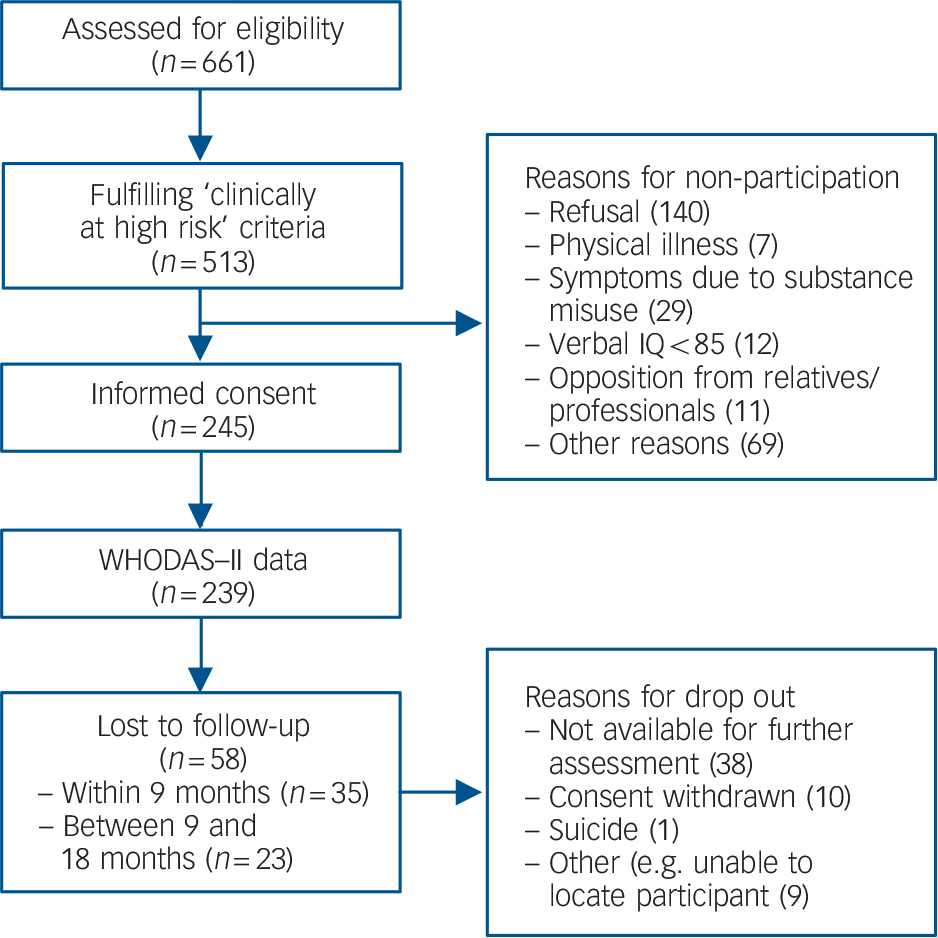
Fig. 1 Flow diagram of participants referred to the European Prediction of Psychosis Study.
The ‘cognitive disturbances’ criterion requires the presence of at least two of nine cognitive basic symptoms of at least ‘moderate’ severity (≥3) during the last 3 months and, independent of severity, first occurrence at least 1 year before intake. The nine basic symptoms were: inability to divide attention, thought interference, pressure, and blockage, disturbances of receptive and of expressive speech, disturbance of abstract thinking, unstable ideas of reference, captivation of attention by details of the visual field.
Exclusion criteria were: a low verbal IQ (IQ<85); past or present psychotic episode lasting longer than 1 week (i.e. fulfilling DSM–IV 22 criteria of a brief psychotic episode for at least 7 days, assessed by the Structured Clinical Interview for DSM–IV Reference First, Spitzer, Gibbon and Williams23 ); and symptoms relevant for inclusion arising from a known general medical disorder or drugs or alcohol dependency. On account of the naturalistic design of the present study, (prior) use of antipsychotics was not considered an exclusion criterion.
The investigation was carried out in accordance with the latest version of the Declaration of Helsinki. The study design was approved by the medical ethics committees of all participating centres. Informed written consent from participants was obtained after the procedure had been fully explained.
Instruments
‘Clinically at high risk’ symptomatology
The SIPS 3.0, Reference McGlashan, Miller, Woods, Rosen, Hoffman and Davidson17 including GAF–M, was employed to determine the presence, severity and type of ultra-high-risk criteria. The Scale of Prodromal Symptoms (SOPS), the rating scale of the SIPS, has four subscales that include five positive symptom items, six negative symptom items, four disorganisation symptoms items and four general symptom items. All symptoms are rated on a seven-point rating scale anchored from 0 (never, absent) to 6 (severe/extreme – and psychotic for the positive items). Symptomatic criteria for prodromal state are exclusively based on positive symptom items and the disorganisation symptom odd behaviour/appearance.
The EPOS investigators received extensive training from Dr Tandy Miller, one of the SIPS authors, including a reliability check after approximately 6 months. The pair-wise interrater concordance of the SIPS was 77% and determined acceptable by the training team.
The Bonn Scale for the Assessment of Basic Symptoms – Prediction list (BSABS–P) Reference Schultze-Lutter, Addington, Ruhrmann and Klosterkötter18 was compiled to assess cognitive disturbances. To the item collection of the BSABS–P, three theoretical subscales were defined totalling 33 cognitive, perceptual and motor disturbances assessed on a seven-point severity scale (0–6) with maximum frequency of occurrence during the preceding 3 months as the guiding criterion. The investigators received repeated training by the scale's first author (F. Schultze-Lutter). Concordance rate with expert rating (F.S.L.) was 87.9%.
Disability
Disability was assessed with the World Health Organization's Disability Assessment Schedule (WHODAS–II). Reference Chopra, Herrman and Kennedy3,Reference Chopra, Couper and Herrman24 The WHO-DAS–II assesses disability in individuals irrespective of diagnosis. The nature of disability is rated directly from individuals' responses. Reference Chopra, Couper and Herrman24 The questionnaire has been conceptually linked to the International Classification of Functioning, Disability and Health, a model of functioning and disability that systematically organises the consequences of disease into three dimensions: body functions and structure, activities, and participation. Reference Chwastiak and Von Korff5 The 36-item interviewer-administered WHODAS–II assesses functioning and disability during the past 30 days and covers the following activity domains:
-
(a) domain 1: understanding and interacting with the world (six items);
-
(b) domain 2: moving and getting around (five items);
-
(c) domain 3: self-care (four items);
-
(d) domain 4: getting along with people (five items);
-
(e) domain 5: life activities (eight items):
-
(i) household activities (four items)
-
(ii) work activities (four items);
-
-
(f) domain 6: participation in society (eight items).
The domain life activities is divided in household activities (four items) and work activities (four items) and because of the small percentage of participants to whom this latter subdomain applied we looked at the subdomain household activities separately. In both subdomains, the amount of time spent on the activities was additionally recorded. The scale's general items were rated on a five-point severity scale, i.e. from 1 (no dysfunction) to 5 (serious to maximum dysfunction).
In addition to the scores on each single item and in congruence with Chopra et al, Reference Chopra, Herrman and Kennedy3 we determined an average score for each domain, thereby allowing a comparison between groups in the WHODAS–II profiles. Hence, the average score for each participant could range from 1 (indicating no reported difficulty) to 5 (indicating severe difficulty) in each domain. Reference Chopra, Herrman and Kennedy3
Until now, no study has been conducted on the specific psychometric properties of the WHODAS–II when used with people at clinically high risk for psychosis. However, the validity of the instrument has been tested in numerous other patient populations, suggesting acceptable internal consistency, test–retest reliability and convergent validity. Reference Chwastiak and Von Korff5,Reference Chopra, Couper and Herrman24–Reference Federici, Meloni, Mancini, Lauriola and Belardinelli26 In a validation study of 904 participants across ten different conditions (for example depression), the WHODAS–II has been shown to have good reliability (Cronbach's α ranged from 0.70 to 0.97 for the different subscales). Reference Pösl, Cieza and Stucki25 Further, Chopra et al Reference Chopra, Couper and Herrman24 and McKibbin et al Reference McKibbin, Patterson and Jeste27 have found the WHODAS–II to have fair test–retest reliability in people with long-term psychotic disorders despite deficiencies in reality testing.
In spite of this demonstrated satisfying reliability, the two items ‘getting dressed’ and ‘problems because of barriers or hindrances in the world around you’ were thought to induce equivocal interpretation within our young sample. ‘Hindrances in the world around you’, for example, may have been misinterpreted as hindrances caused by other people. Only those who interpreted hindrances as physical (for example, hindrances caused by a wheelchair) understood the question correctly. Because of the questionable reliability of these two items in our group, they were not entered in the analyses and not considered further.
Follow-up
Follow-up assessments with the SIPS and BSABS–P took place at 9 and 18 months. Transition to psychosis was operationalised as a continuation of BLIPS, i.e. any single item on the positive subscale of SIPS (SIPS–Positive) with a score of 6 for more than 7 days. Reference Cannon, Cadenhead, Cornblatt, Woods, Addington and Walker13,Reference Yung, Phillips, Yuen and McGorry28,Reference Yung, Nelson, Stanford, Simmons, Cosgrave and Killackey29 Following identification of full-blown psychotic symptoms in the SIPS interview, the diagnostic category on transition was determined by applying DSM–IV criteria 22 of psychotic disorders and affective disorders with psychotic features. Thereby, the different time threshold of Criterion B of ‘brief psychotic disorder’ was adapted to the BLIPS definition. Past and present psychosis as parts of the exclusion criteria as well as psychotic diagnosis on transition were assessed with the SCID–I. Reference First, Spitzer, Gibbon and Williams23
Statistical analysis
Statistical analyses were performed using SPSS (version 16.0.2) for Windows. Comparisons between the transition group and non-transition group were made with Pearson's chi-squared tests and an independent sample t-test for the GAF score.
To examine differences in the categorical domains as well as in the separate categorical items of the WHODAS–II, Mann–Whitney U-tests were used. Kaplan–Meier survival analysis was used to calculate survival time, i.e. time to transition.
The effect of different covariates on time to transition was estimated with the Cox proportional hazard model. In this model, transition to psychosis was entered as the status variable, time to onset of psychosis or to follow-up as the time variable and the weighted sum scores of the WHODAS–II domains (with the exception of work activities) as covariates. Weighted sum scores of each domain were computed by dividing the sum score of all items (except for the items ‘getting dressed’ and ‘problems because of barriers or hindrances in the world around you’) with the number of items. Subsequently, these scores were entered into the analysis as ordinal data using the backward Wald method. Treatment was entered as an additional covariate in the model using the blockwise method. The recruiting centre was entered as a stratum variable in the regression model. P<0.05 was considered statistically significant. A survival curve was obtained using Kaplan–Meier survival analysis.
Bivariate correlations between social disability and SIPS symptoms were evaluated using Spearman's rank correlation. We adjusted the type I error, i.e. alpha, for multiple testing by dividing the overall alpha level of 0.05 by the number of items. After this correction, P≤0.0013 was considered statistically significant.
Results
Participant characteristics
At 18-month follow-up, 37 participants had made a transition to psychosis. The transition group did not differ significantly from the non-transition group in terms of age, gender and drug use. In congruence with results of previous studies, Reference Cannon, Cadenhead, Cornblatt, Woods, Addington and Walker13,Reference Ruhrmann, Schultze-Lutter, Salokangas, Heinimaa, Linszen and Dingemans15 we found a difference in the level of global functioning (GAF score) at baseline (P<0.005), with a significantly lower GAF score in the transition group (42.4, s.d. = 1.6) compared with the non-transition group (52.7, s.d. = 0.8).
Only four participants met all three ultra-high-risk criteria, three of whom also met cognitive disturbances criteria. In total 143 participants displayed both ultra-high-risk and cognitive disturbances symptoms, 72 met the ultra-high-risk criteria, and 24 reported only cognitive disturbances symptoms. These three cohorts did not significantly differ in transition rates. The mean time to transition from baseline examination was 496.8 days (s.e. = 8.5, 95% CI 480.2–513.6).
A total of 239 participants completed the WHODAS–II at baseline (131 males, mean age 22.5, s.d. = 5.3). Of these, 35 were lost to follow-up at 9 months and altogether 58 at 18 months. Participants did not differ significantly from those lost to follow-up in terms of age, gender, severity of negative symptoms, severity of disability, global functioning and familial risk. However, differences were found on two positive symptom items of the SIPS; those lost to follow-up had a lower score on suspiciousness (t =–2.07, P = 0.040) and a higher score on hallucinations at baseline (t = 2.15, P = 0.030). As regards medication, antidepressants were prescribed to 45 (18.8%) of the 239 participants, antipsychotics to 29 (12.1%) and both antidepressants and antipsychotics to 22 (9.2%); for 30 (12.6%) participants no reliable information on their medication was available.
Disability
Online Table DS1 displays the overall scores of the domains as assessed by the WHODAS–II. Within the entire group, the highest disability scores were found in domain 6: participation in society (mean 2.38, s.d. = 0.96, median 2.29). Mann–Whitney U-tests indicate that the baseline ratings of the transition group were significantly higher than those of the non-transition group only in domain 4, getting along with people (z = –3.03, P = 0.002). This difference was mainly caused by the items making new friends (z = –3.40, P = 0.001), maintaining a friendship (z =–3.00, P = 0.003) and dealing with people you do not know (z =–2.28, P = 0.023).
Although no significant difference was found for average scores in domain 6, participation in society, a difference was found within its component item, joining community activities (z =–2.0, P = 0.05), with the transition group reporting more difficulties in this area.
Questions related to work activities could not be answered by 33% of the participants (37% of the transition group and 31% of the non-transition group), mostly because of participants not having been employed or otherwise working (i.e. self-employed, studying, in school or training or doing housework etc.) in the past 30 days before the interview. For the remaining participants, the difference between the two groups in number of hours spent on work or study is approaching significance (t =–1.93, P = 0.056), with the transition group working less total hours than the non-transition group. The amount of missing data because of the absence of work/study in the subdomain work activities rendered further interpretation of this section impracticable.
Cox regression analysis
The backward stepwise Cox proportional hazard model was used to identify the predictive value of the weighted sum scores of the six domains, except for the subdomain work activities. In this analysis, domain 4, getting along with people, was retained in the model as a predictor of a first psychotic episode (β = 0.672, s.e. = 0.187, Wald = 12.964, P<0.001, hazard ratio (HR) = 1.958, 95% CI 1.358–2.822). Thus the relative risk of developing a psychosis nearly doubled with increasing difficulties in contact with other people. None of the other covariates further contributed independently to the prediction of a first psychotic episode.
Adding treatment to the model, the use of antipsychotics (β =–1.682, s.e. = 0.534, P = 0.002) and antidepressants (β =–2.125, s.e. = 0.743, P = 0.004) were kept in the equation. Domain 4, getting along with people, continued to contribute significantly to the equation, and the change in hazard ratio was only minor (β = 0.569, s.e. = 0.184, Wald = 9.548, P = 0.002, HR = 1.767, 95% CI 1.238–2.550).
The survival curves of the two disability groups for the domain getting along with people are presented in Fig. 2; the rate of transition differed significantly between the group with moderate to severe disability (n = 72) and the group with no to mild disability (n = 167) in this domain (ω2 = 11.91, P = 0.001).
Spearman's rank correlation
Table 1 displays the correlations between symptoms as assessed with the SIPS and social disability before (P<0.05) and after Bonferroni adjustment (P adjusted<0.0013). We found disability in the fourth domain, getting along with people, and the sixth domain, participation in society, to be related to both negative and general symptoms of the SIPS. Although not surviving Bonferroni adjustment, there was an additional association between social disability and the SIPS positive item suspiciousness. The highest correlations, however, were found between getting along with people and the negative symptom social anhedonia and withdrawal as well as between participation in society and the general symptom dysphoric mood.
Table 1 Spearman's rank order correlations between social disability and clinical syndromes (n = 239)

| Getting along with peopleb | Participation in societyb | |||
|---|---|---|---|---|
| Measuresa | Rho | P | Rho | P |
| P1. Unusual thought content/delusional ideas | 0.11 | 0.11 | 0.24 | 0.71 |
| P2. Suspiciousness/persecutory ideas | 0.20 | 0.002* | 0.17 | 0.008* |
| P3. Grandiose ideas | –0.01 | 0.90 | –0.05 | 0.44 |
| P4. Perceptual abnormalities/hallucinations | –0.003 | 0.97 | 0.05 | 0.41 |
| P5. Disorganized communication | 0.09 | 0.15 | 0.14 | 0.04 |
| N1. Social anhedonia and withdrawal | 0.36 | <0.001** | 0.29 | <0.001** |
| N2. Avolition | 0.24 | <0.001** | 0.33 | <0.001** |
| N3. Decreased expression of emotion | 0.30 | <0.001** | 0.23 | <0.001** |
| N4. Decreased experience of emotions and self | 0.23 | <0.001** | 0.17 | 0.01* |
| N5. Decreased ideational richness | 0.09 | 0.16 | 0.08 | 0.20 |
| N6. Deterioration in role functioning | 0.24 | <0.001** | 0.29 | <0.001** |
| D1. Odd behaviour or appearance | 0.22 | 0.001** | 0.17 | 0.008* |
| D2. Bizarre thinking | 0.17 | 0.01* | 0.09 | 0.16 |
| D3. Trouble with focus and attention | 0.17 | 0.009* | 0.22 | 0.001** |
| D4. Personal hygiene/social attentiveness | 0.16 | 0.02 | 0.12 | 0.06 |
| G1. Sleep disturbance | 0.16 | 0.01* | 0.21 | 0.001** |
| G2. Dysphoric mood | 0.27 | <0.001** | 0.36 | <0.001** |
| G3. Motor disturbances | 0.18 | 0.006* | 0.27 | <0.001** |
| G4. Impaired tolerance to normal stress | 0.27 | <0.001** | 0.34 | <0.001** |
Discussion
In the present study on the role of disability in the transition to psychosis, we employed a validated and reliable questionnaire to measure a wide range of disability in a large sample of young people clinically at high risk for a psychosis. To our knowledge, our study is not only the first prospective study to use the WHODAS–II in a large clinical high-risk sample but also the first prospective study that in detail examined disability as a risk factor of a transition to psychosis. As expected, we found that within this help-seeking population, the transition group was experiencing significantly greater social disability at baseline compared with the non-transition group.
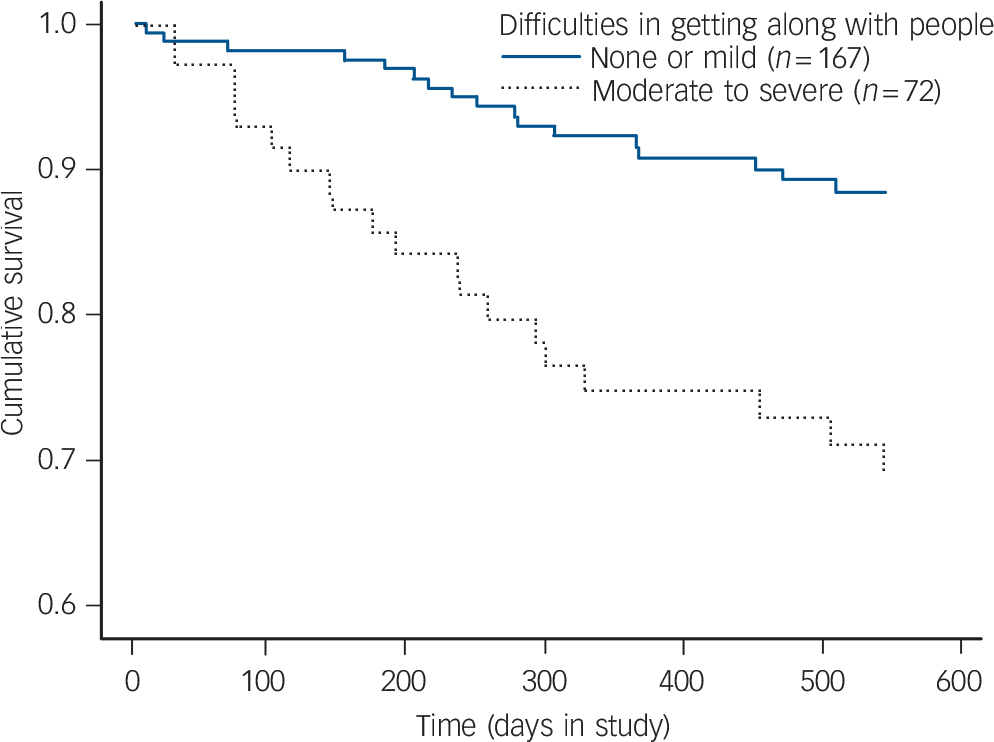
Fig. 2 Survival analysis for 18-month follow-up (n = 239).
Difficulties in the domain getting along with people appeared to contribute most to the prediction of the onset of a first psychotic episode within a study of people clinically at high risk. Within this category, developing or maintaining friendships appeared to be particularly problematic for those later developing psychosis, since those who made a transition reported significantly greater difficulties at baseline in both developing and maintaining friendships compared with those who did not make the transition. Although we only compared disability in cross-section, such difficulties may well be progressive. In a representative group of young people with early-onset psychosis, Boeing et al Reference Boeing, Murray, Pelosi, McCabe, Blackwood and Wrate30 found that up to 82% of the sample experienced a moderate to severe ‘lack’ of friendships during their first psychotic episode. This is a noteworthy finding, since strong social networks are related to both better health status and better quality of life. Reference Becker, Leese, McCrone, Clarkson, Szmukler and Thornicroft31
Furthermore, our correlation analyses suggest that social disability in individuals clinically at high risk may be mainly related to negative and general symptoms although a modest association was found between social disability and the positive SIPS item suspiciousness. Difficulties in getting along with people showed high correlations with negative symptoms, and disturbances in participating in society were mainly associated with general symptoms such as dysphoric mood. Since recent studies have found strong associations between negative symptoms and transition to psychosis in a sample clinically at high risk, Reference Cannon, Cadenhead, Cornblatt, Woods, Addington and Walker13,Reference Velthorst, Nieman, Becker, van der Fliert, Dingemans and Klaassen14 it is not surprising that difficulties in getting along with people was the best predictor of a first psychotic episode in our sample.
Our findings on social disability in our sample are also in line with previous studies of individuals clinically at high risk, in which transition was associated with more severe social anhedonia and withdrawal. Reference Cannon, Cadenhead, Cornblatt, Woods, Addington and Walker13,Reference Velthorst, Nieman, Becker, van der Fliert, Dingemans and Klaassen14,Reference Mason, Startup, Halpin, Schall, Conrad and Carr32,Reference Riecher-Rössler, Gschwandtner, Aston, Borgwardt, Drewe and Fuhr33 In a retrospective study, participants diagnosed with schizophrenia frequently increasingly lagged behind their healthy peers in social development from an early, pre-psychotic state on. Reference Häfner, Nowotny and Löffler8,Reference Rössler, Salize, van Os and Riecher-Rössler34 Using the Premorbid Adjustment Scale, Shapiro et al Reference Shapiro, Marenco, Spoor, Egan, Weinberger and Gold35 found social abnormalities in childhood even before the age of 11.
These studies suggest that social deficits may precede positive ultra-high-risk symptoms. Such deficits may be caused by the early onset of attenuated negative symptoms as suggested by the clinical high-risk model of Cornblatt et al. Reference Cornblatt, Lencz, Smith, Correll, Auther and Nakayama36 Negative and general affective symptoms may cause the adolescent to withdraw from increasingly demanding and complex social contacts when adult roles have to be taken up. Reference Häfner, Nowotny and Löffler8 In addition, the reported negative attitude of others when the individual is in the clinically at high risk phase Reference Salokangas, Heinimaa, Svirskis, Laine and Huttunen37 may also contribute to this social withdrawal. Irrespective of the possible nature of any interaction between negative and positive symptoms and social disability, poor premorbid social functioning has been related to elevated risk and poor outcomes in psychotic disorders, Reference Gureje, Herrman, Harvey, Morgan and Jablensky6 thus making it a significant target for an early intervention in the at-risk state. Improving social functioning, for example through cognitive–behavioural therapy in the clinically at high risk period, may protect the adolescent or young adult from social isolation, and maintaining reality testing with peers may reduce the risk of later delusional ideation.
In addition, to improve psychosis prediction, an assessment of disability in people clinically at high risk may be relevant in identifying important needs, for both the individual and their families. Reference Herrman, Hawthorne and Thomas38 Help in meeting these needs through appropriate early intervention is likely to also improve the quality of life of individuals clinically at high risk and, thereby, to diminish the likelihood of transition to psychosis.
Limitations
In spite of the outlined strengths of our study, some critical issues regarding the use of the WHODAS–II in a clinically at high risk sample need to be addressed. First, the WHODAS–II questionnaire does not take into account some of the unique social issues that occur in adolescence. In their study of individuals at ultra-high risk, Cornblatt et al Reference Cornblatt, Auther, Niendam, Smith, Zinberg and Bearden7 employed two new, broad measures (social and role scale) based on the Social and Occupational Functioning Assessment Scale (SOFAS) and GAF respectively to identify disability, incorporating specific issues such as peer acceptance and dating. An additional consideration of these scales in future research may help to decide whether these are superior to the WHODAS–II in younger samples and/or whether they further contribute to improvements in psychosis prediction.
Second, a methodological issue must be considered. As fitted models always perform in an ‘optimistic manner’ Reference Hosmer and Lemeshow39 in the model-development data, cross-validation in an independent sample is needed to control for tailor-made modelling. Although sample splitting in theory is an option for model validation in large samples, the limited number of transitions did not allow it for statistical reasons. Existing or future samples of comparable size and risk definition are required to validate our findings.
Another potential limitation concerns the 9-month gap in between assessments that may have introduced some inaccuracies in recall, particularly of transition. Indeed, some participants may have had some difficulties in distinguishing their attenuated symptoms from full-blown psychotic symptoms after several months, whereas others may have failed to recall a past psychotic episode at the time of the interview. This latter possibility, however, seems unlikely as researcher staff were trained to probe for transition at follow-up assessments. Additionally, if the participant had not remained in the care of the specialised centre but was back-referred, the referring practitioner as well as the participant and his/her family had been asked to re-establish contact as soon as they suspected psychosis or if the mental state of the individual deteriorated notably.
Finally, once participants made a transition to a psychosis, they were not included in follow-up assessments of the WHODAS–II. Thus it will not be possible to further examine the course and severity of disability after the onset of psychoses. Addressing this issue in future research is recommended.
In conclusion, our study gives further evidence that a decline in social functioning may be a risk factor for a first psychosis within samples clinically at high risk. Reference Cannon, Cadenhead, Cornblatt, Woods, Addington and Walker13,Reference Ruhrmann, Schultze-Lutter, Salokangas, Heinimaa, Linszen and Dingemans15 In combination with other predictors (neurobiological or psychopathological parameters), social disability measures may make a valuable contribution to a more accurate prediction of first psychotic episodes.
Acknowledgements
The International Advisory Board ‘European Prediction of Psychosis’ (EPOS) group comprises: Patrick D. McGorry, Australia; Thomas H. McGlashan, USA; Martin Knapp, UK.
The authors thank the following scientists for their work for EPOS: Reinaud van der Fliert, MSc, Rianne Klaassen, MD (Department of Psychiatry, AMC, University of Amsterdam, the Netherlands); Heinz Picker, PhD, Meike Neumann, Dipl-Psych, Anke Brockhaus-Dumke, MD, Ralf Pukrop, PhD (Department of Psychiatry & Psychotherapy/FETZ, University of Cologne, Cologne, FRG); Tanja Svirskis, MD, PhD, Jukka Huttunen, BM, Tiina Laine, MSc, Tuula Ilonen, PhD, Terja Ristkari, MA, Jarmo Hietala, MD, PhD, (Department of Psychiatry, University of Turku, Turku, Finland); Amanda Skeate, DClin (ED:IT, Early Intervention Service, Birmingham, UK); Yehonala Gudlowski, Dipl-Psych (Charité, Berlin, FRG); Seza Ozgürdal, Dipl-Psych, Henning Witthaus, MD (Charité, Berlin, and Ruhr-University, Bochum, FRG); Shôn Lewis, MD, PhD, Antony Morrisson, PhD (School of Psychological Sciences, University of Manchester, Manchester, UK).






eLetters
No eLetters have been published for this article.