Death rates are increased in people with severe mental illness (SMI) relative to the general population, and this is reflected in the 10- to 20-year reduced life expectancy observed. Reference Laursen1–Reference Chang, Hayes, Perera, Broadbent, Fernandes and Lee3 This has been found in a number of longitudinal studies. Reference Hoang, Stewart and Goldacre4–Reference Saha, Chant and McGrath7 However, it has been reported that the mortality gap has narrowed or plateaued since the mid-1990s in some countries. Reference Bushe, Taylor and Haukka8,Reference Wahlbeck, Westman, Nordentoft, Gissler and Laursen9 Since the turn of the millennium, a number of strategies aimed at reducing the mortality gap between people with SMI and the general population have been implemented in the UK. Reference Martin Roland10–Reference Swinson, Ashim, Windfuhr, Kapur, Appleby and Shaw17 Similar strategies have been employed in other countries, including the USA, Reference Druss, Rohrbaugh, Levinson and Rosenheck18,Reference Koyanagi and Carty19 Canada, Reference Callaghan and Khizar20,21 Australia Reference Galletly, Foley, Waterreus, Watts, Castle and McGrath22–Reference Pirkis, Harris, Hall and Ftanou24 and throughout Europe. Reference Fleischhacker, Cetkovich-Bakmas, De Hert, Hennekens, Lambert and Leucht25,Reference De Hert, Dekker, Wood, Kahl, Holt and Möller26 Few studies cover this period. It is therefore timely to review all-cause and cause-specific mortality rates in individuals with bipolar disorder and schizophrenia relative to the general population. We used cardiovascular (CVD) mortality and suicide as exemplars of natural and unnatural causes of death that have been targets of mental health policy. Evidence suggests CVD is the leading cause of death in individuals with SMI. Reference Laursen, Wahlbeck, Hallgren, Westman, Osby and Alinaghizadeh2,Reference Roshanaei-Moghaddam and Katon27,Reference Weiner, Warren and Fiedorowicz28 However, large representative longitudinal studies remain limited in number Reference Osborn, Levy, Nazareth, Petersen, Islam and King29 In the UK a number of initiatives have been targeted at reducing CVD morbidity; to encourage earlier diagnosis, Reference Martin Roland10 reduce risk factors Reference McCreadie30,Reference Cormac31 and ensure effective treatment implementation. Reference Miller32 Suicide is the cause of death that is most elevated in individuals with bipolar disorder and schizophrenia compared with the general population. Reference Hayes, Miles, Walters, King and Osborn33,Reference Brown34 Up-to-date estimates of suicide rates are important markers of the success of psychiatric care. Reference Swinson, Ashim, Windfuhr, Kapur, Appleby and Shaw17 Self-harm is the major risk factor for suicide, with a large number of those dying by suicide having a history of self-harm. Reference Owens, Horrocks and House35 Self-harm is also a marker of quality of life and emotional distress in individuals with SMI. Reference Singhal, Ross, Seminog, Hawton and Goldacre36 This study compares all-cause mortality, CVD death and CVD diagnosis, suicide and self-harm rates in people with bipolar disorder, and schizophrenia and a general population comparator group from 2000 to 2014.
Method
Study design and setting
We completed a cohort study in pseudonymised primary care electronic health records (EHR) provided by The Health Improvement Network (THIN) from 1 January 2000 until 31 December 2014. The database contains longitudinal records of over 11 million people and currently covers almost 6% of the UK population. Almost 99% of the UK population are registered with a primary care practice, and the majority will be so from birth to death. Reference Lis and Mann37 Individuals included in THIN are representative of the UK general population in terms of age, gender, medical conditions and death rates, and the behaviour of physicians entering data is representative in terms of consultation and prescribing statistics. Reference Blak, Thompson, Dattani and Bourke38 Primary care practices were only included in the study after the date at which they met acceptable mortality and computer usage reporting. Reference Maguire, Blak and Thompson39,Reference Horsfall, Walters and Petersen40 The reliability of THIN for research purposes has been validated against experimental and other observational evidence. Reference Langley, Szatkowski, Gibson, Huang, McNeill and Coleman41,Reference Lewis, Schinnar, Bilker, Wang and Strom42 THIN records are based on Read codes, a hierarchical coding system that includes diagnoses (which map onto ICD-10 codes), medication prescriptions, symptoms, examination findings, referrals, test results and hospital attendance information Reference Chisholm43 The study was approved by the Cegedim Strategic Data Medical Research UK Scientific Review Committee in March 2015. The scheme for THIN to provide anonymised patient data to researchers was approved in 2003 by the National Health Service South-East Multicentre Research Ethics Committee.
Participants
All individuals aged 16 or over, ever receiving a diagnosis of bipolar disorder or schizophrenia were included. If individuals had multiple diagnoses, they were classified by the diagnosis most recently assigned. Patients with schizoaffective disorders and unipolar depression were excluded. The validity of bipolar disorder and schizophrenia diagnoses in primary care records has been established and the incidence of bipolar disorder and schizophrenia in THIN is similar to other European cohorts. Reference Nazareth, King, Haines, Rangel and Myers44,Reference Hardoon, Hayes, Blackburn, Petersen, Walters and Nazareth45 Individuals with bipolar disorder and schizophrenia were frequency matched with up to six individuals without these diagnoses to create a comparator group. The comparator group was matched on age (in 5-year age bands) and gender, from within the same primary care practice.
Outcomes
CVD was defined as any entry of myocardial infarction, ischemic heart disease or cerebrovascular event in the EHR. CVD diagnoses have previously been validated in THIN with positive predictive values (PPVs) greater than 90%. Reference Ruigómez, Martín-Merino and Rodríguez46,Reference Hammad, McAdams, Feight, lyasu and Dal Pan47 In line with other studies, CVD mortality was defined as a death code with a CVD cause, or a CVD code, followed by a death code in the following 60 days and a final date of any activity in the EHR within 6 months. Reference Ogdie, Yu, Haynes, Love, Maliha and Jiang48
The definition for self-harm events included Read codes for intentional poisoning, intentional self-injurious behaviour and self-harm acts of uncertain intent. This unitary definition of self-harm, where there is no distinction made between non-suicidal self-harm and self-harm with suicidal intent is consistent with UK research norms. Reference Haw, Casey, Holmes and Hawton49 The PPV of this outcome in THIN has been shown to be 97%. Reference Arana, Wentworth, Ayuso-Mateos and Arellano50 Suicide was defined as a death code identified as suicide or a self-harm code followed by a death code in the following 30 days and a final date of any activity in the EHR within 6 months, in line with previous research. Reference Arana, Wentworth, Ayuso-Mateos and Arellano50,Reference Hayes, Pitman, Marston, Walters, Geddes and King51
Statistical analyses
Rates of all-cause, cause-specific mortality (CVD deaths, suicide) and morbidity (CVD, self-harm) were calculated. Annual rates were calculated for all-cause mortality. In order to assess trends in rates over time, a segmented regression analysis using joinpoint models was performed. Reference Wagner, Soumerai, Zhang and Ross-Degnan52,Reference Kim, Fay, Feuer and Midthune53 To complete this analysis the Surveillance Research Program of the United States National Cancer Institute Joinpoint software was used (Version 4.3.1.0). This analysis identifies time points where there is a change in the linear slope of the trend. The optimum number of linear slopes and pinpoints is assessed using modified Bayesian Information Criteria. Reference Zhang and Siegmund54 The models incorporate estimated variation for each data point using the standard error of the rate. After identification of a change in trend, segmented regression can be fitted and annual percentage change in rate (with 95% confidence intervals) can be calculated.
Cox proportional hazards regression analyses were conducted comparing all-cause mortality rates in individuals with bipolar disorder, schizophrenia and the matched comparison group. The assumption of proportional hazards was tested by analysis of Schoenfeld residuals. Reference Schoenfeld55 A number of multivariable models were tested. First, the association between diagnosis and all-cause mortality was assessed, adjusting for age, gender and calendar year and clustering within primary care practices, then additionally adjusting for area-level deprivation (defined as quintiles of Townsend score, Reference Townsend56 based on the patient's lower super output area – an average of 650 households), and ethnicity (categorised as White British, White other, Black, Asian, mixed and other). This fully adjusted model was stratified by gender, and age (16–50 years, over 50 years old). As with the rate, annual adjusted hazard ratios (HRs) were calculated for all-cause mortality and trends in HRs were assessed using joinpoint regression. In this instance, a change in HR per year (with 95% CIs) was calculated.
The associations between diagnosis and CVD death, CVD diagnosis, suicide and self-harm were assessed using the same multivariable approach, additionally adjusting for average number of visits to the physician per year of follow-up (to account for the likelihood of having a diagnostic code recorded in the EHR). For each outcome these models were stratified by gender, age (16–50 years, over 50 years old) and calendar period (start of 2000 until end of 2004, start of 2005 until end of 2009, start of 2010 until end of 2014) to examine potential effect modification.
A further model also including smoking status (worst ever of: never, ex-, current smoker), body mass index (BMI) (worst ever of: healthy weight, overweight, obese), and diagnoses of hypercholesterolemia (defined as total cholesterol ⩾5.2mmol/L), Reference Ford, Mokdad, Giles and Mensah57 hypertension (defined as code for hypertension or two sequential records of systolic blood pressure > 140 mmHg) 58 and type 2 diabetes mellitus during follow-up was tested to see if these covariates explained elevated rates of CVD death and diagnoses. All analyses were completed using statistical software (Stata, version 14).
Results
Clinical and demographic features
A total of 17 314 people had a diagnosis of bipolar disorder and 22 497 had a diagnosis of schizophrenia with active records between the start of 2000 and the end of 2014. These were matched with 219 387 individuals who never received bipolar disorder or schizophrenia diagnoses. There were 1266 deaths in total in the group with bipolar disorder, 2061 in those with schizophrenia and 6279 in the comparison group (Table 1).
Table 1 Cohort charactersitics
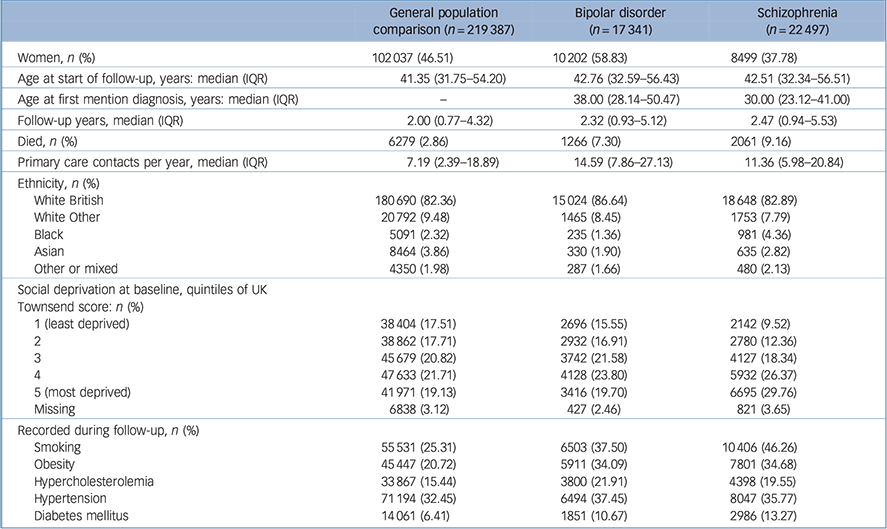
| General population comparison (n = 219 387) |
Bipolar disorder (n = 17 341) |
Schizophrenia (n = 22 497) |
|
|---|---|---|---|
| Women, n (%) | 102 037 (46.51) | 102 02 (58.83) | 8499 (37.78) |
| Age at start of follow-up, years: median (IQR) | 41.35 (31.75–54.20) | 42.76 (32.59–56.43) | 42.51 (32.34–56.51) |
| Age at first mention diagnosis, years: median (IQR) | – | 38.00 (28.14–50.47) | 30.00 (23.12–41.00) |
| Follow-up years, median (IQR) | 2.00 (0.77–4.32) | 2.32 (0.93–5.12) | 2.47 (0.94–5.53) |
| Died, n (%) | 6279 (2.86) | 1266 (7.30) | 2061 (9.16) |
| Primary care contacts per year, median (IQR) | 7.19 (2.39–18.89) | 14.59 (7.86–27.13) | 11.36 (5.98–20.84) |
| Ethnicity, n (%) | |||
| White British | 180 690 (82.36) | 15 024 (86.64) | 18 648 (82.89) |
| White Other | 20 792 (9.48) | 1465 (8.45) | 1753 (7.79) |
| Black | 5091 (2.32) | 235 (1.36) | 981 (4.36) |
| Asian | 8464 (3.86) | 330 (1.90) | 635 (2.82) |
| Other or mixed | 4350(1.98) | 287 (1.66) | 480 (2.13) |
| Social deprivation at baseline, quintiles of UK Townsend score: n (%) |
|||
| 1 (least deprived) | 38 404 (17.51) | 2696 (15.55) | 2142 (9.52) |
| 2 | 38 862 (17.71) | 2932 (16.91) | 2780 (12.36) |
| 3 | 45 679 (20.82) | 3742 (21.58) | 4127 (18.34) |
| 4 | 47 633 (21.71) | 4128 (23.80) | 5932 (26.37) |
| 5 (most deprived) | 41 971 (19.13) | 3416 (19.70) | 6695 (29.76) |
| Missing | 6838 (3.12) | 427 (2.46) | 821 (3.65) |
| Recorded during follow-up, n (%) | |||
| Smoking | 55 531 (25.31) | 6503 (37.50) | 10 406 (46.26) |
| Obesity | 45 447 (20.72) | 5911 (34.09) | 7801 (34.68) |
| Hypercholesterolemia | 33 867 (15.44) | 3800 (21.91) | 4398 (19.55) |
| Hypertension | 71 194 (32.45) | 6494 (37.45) | 8047 (35.77) |
| Diabetes mellitus | 14 061 (6.41) | 1851 (10.67) | 2986 (13.27) |
All-cause mortality
The rate of all-cause mortality in individuals with bipolar disorder was 210.34 per 10 000 person-years at-risk (PYAR, 95% CI 199.07–222.25). Trends in bipolar disorder mortality rate suggested a reduction over follow-up time. Joinpoint regression fitted a linear model with a significant annual percentage change (APC) in rate of −4.1% (95% CI −5.1 to −3.1) (Fig. 1). In individuals with schizophrenia the mortality rate was 248.57 per 10 000 PYAR (95% CI 238.06–259.53). Similarly, the rate of mortality in schizophrenia reduced between 2000 and 2014, with joinpoint regression fitting a model with no joinpoints, and an APC of −2.0% (95% CI −3.0 to −0.9). In the comparison population the mortality rate was relatively stable between 2000 and 2003 (APC 4.2, 95% CI −7.6 to 17.6) and then decreased until the end of the study period (APC −8.1, 95% CI −9.6 to −6.5).
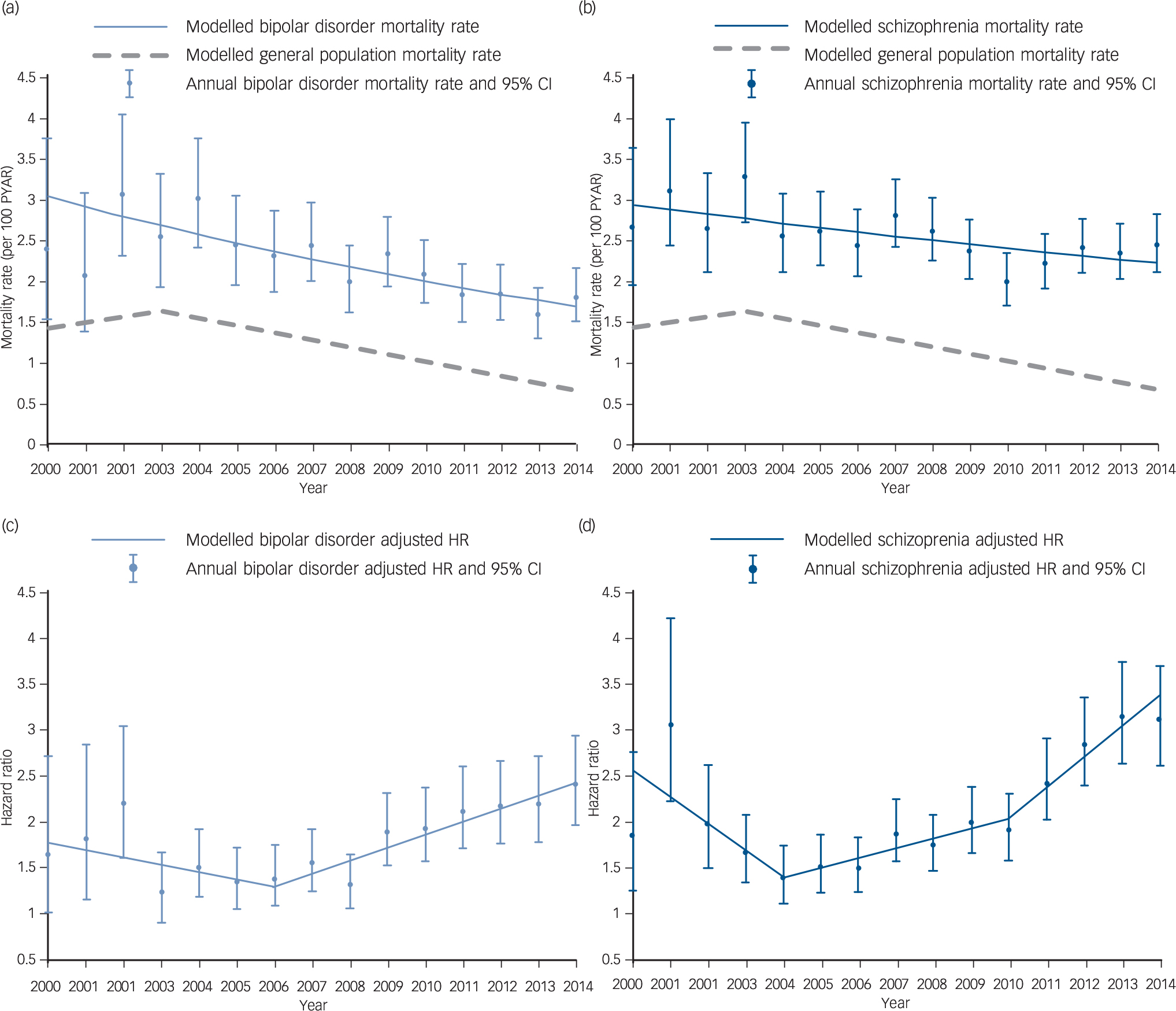
Fig. 1 All-cause mortality rate and adjusted hazard ratios compared with the general population. In bipolar disorder and schizophrenia (2000–2014).
Mortality rates, per 100 person-years at-risk (PYAR) in (a) bipolar disorder and (b) schizophrenia groups. Adjusted hazard ratios (HRs) in (c) bipolar disorder and (d) schizophrenia groups.
Mortality was elevated in those with bipolar disorder (HR = 1.77, 95% CI 1.67–1.88) and schizophrenia (HR = 2.08, 95% CI 1.98–2.19) relative to the general population comparison group, after adjustment for age gender, calendar year, area-level deprivation and ethnicity (Table 2). Stratification by gender suggested that men and women with bipolar disorder had similarly elevated mortality rates (P = 0.297), but in those with schizophrenia men's morality rate (HR = 2.50, 95% CI 2.32–2.69) was more elevated than women's (HR = 1.78, 95% CI 1.66–1.90, test for interaction P < 0.0001). Mortality rates in those aged 16–50 were more elevated, relative to the comparison group, than those over 50 (test for interaction in both groups P < 0.0001). Individuals with bipolar disorder aged 16–50 had an adjusted HR of 3.22 (95% CI 2.77–3.75), individuals with schizophrenia had an adjusted HR of 4.69 (95% CI 4.16–5.29) in comparison with those aged 16–50 in the general population. There was evidence of a change in HRs over the study period for groups with both bipolar disorder and schizophrenia diagnoses (Fig. 1). Joinpoint analysis fitted a model with one joinpoint at 2006 for bipolar disorder. This model suggested that the HR decreased by 0.08 (95% CI −0.18–0.02) per year between 2000 and 2006, and significantly increased by 0.14 (95% CI 0.10–0.19) per year from 2006 to 2014 (Fig. 1). For schizophrenia, joinpoint regression fitted three linear models (two joinpoints); from 2000 to 2004 the HR reduced significantly by 0.29 (95% CI −0.48 to −0.10) per year, from 2004 to 2010 the HR increased by 0.11 (95% CI 0.04 to 0.17) per year, and from 2010 to 2014 increased by 0.34 (95% CI 0.18 to 0.49) (Fig. 1).
Table 2 All-cause mortality rates
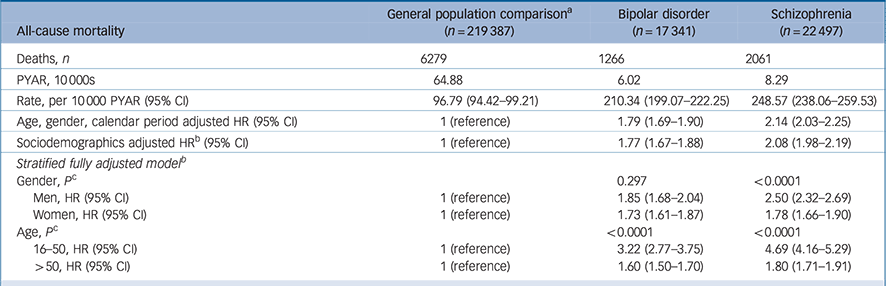
| All-cause mortality | General population comparison
a
(n = 219 387) |
Bipolar disorder (n = 17 341) |
Schizophrenia (n = 22 497) |
|---|---|---|---|
| Deaths, n | 6279 | 1266 | 2061 |
| PYAR, 10000s | 64.88 | 6.02 | 8.29 |
| Rate, per 10000 pyar (95% CI) | 96.79 (94.42–99.21) | 210.34 (199.07–222.25) | 248.57 (238.06–259.53) |
| Age, gender, calendar period adjusted HR (95% CI) | 1 (reference) | 1.79 (1.69–1.90) | 2.14 (2.03–2.25) |
| Sociodemographics adjusted HR b (95% CI) | 1 (reference) | 1.77 (1.67–1.88) | 2.08 (1.98–2.19) |
| Stratified fully adjusted model b | |||
| Gender, P c | 0.297 | <0.0001 | |
| Men, HR (95% CI) | 1 (reference) | 1.85 (1.68–2.04) | 2.50 (2.32–2.69) |
| Women, HR (95% CI) | 1 (reference) | 1.73 (1.61–1.87) | 1.78 (1.66–1.90) |
| Age, P c | <0.0001 | <0.0001 | |
| 16–50, HR (95% CI) | 1 (reference) | 3.22 (2.77–3.75) | 4.69 (4.16–5.29) |
| >50, HR (95% CI) | 1 (reference) | 1.60 (1.50–1.70) | 1.80 (1.71–1.91) |
PYAR, person-years at-risk; HR, hazard ratio.
a. General population comparison group of up to six individuals without bipolar disorder or schizophrenia matched for gender, age group and primary care practice.
b. Age, gender, calendar period, area-level deprivation, ethnicity.
c. Test for heterogeneity.
CVD mortality and CVD diagnoses
There was an elevated HR for CVD deaths in schizophrenia (HR = 1.39, 95% CI 1.12–1.73) after accounting for age, gender, calendar year, area-level deprivation, ethnicity and average number of visits to the physician during follow-up (Table 3). Following additional adjustment for smoking, hypercholesterolemia, hypertension, BMI and diabetes CVD deaths were no longer elevated in people with schizophrenia relative to the general population (HR = 1.22, 95% CI 0.98–1.52). The HR for bipolar disorder was not elevated, relative to the comparison group and there was no evidence that this differed by gender or age (Table 3). However, stratification by 5-year periods suggested that CVD deaths were elevated in people with bipolar disorder, relative to the general population after 2010 (HR = 1.92, 95% CI 1.24–2.98), but not before this. Among those individuals with schizophrenia, gender was not an effect modifier (P = 0.068). The HR for CVD mortality in people with schizophrenia aged 16–50 was 3.20 (95% CI 1.62–6.31), whereas in those over 50 it was 1.29 (95% CI 1.03–1.63, test for interaction P = 0.013) (Table 3).
Table 3 Cardiovascular disease mortality and suicide rates
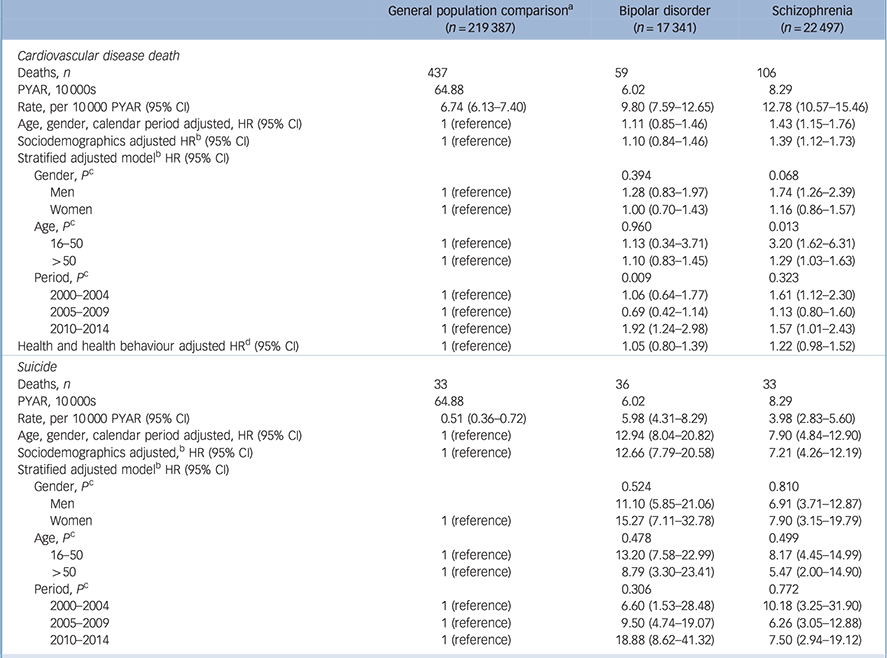
| General population comparison
a
(n = 219 387) |
Bipolar disorder (n = 17 341) |
Schizophrenia (n = 22 497) |
|
|---|---|---|---|
| Cardiovascular disease death | |||
| Deaths, n | 437 | 59 | 106 |
| PYAR, 10000S | 64.88 | 6.02 | 8.29 |
| Rate, per 10000 PYAR (95% CI) | 6.74 (6.13–7.40) | 9.80 (7.59–12.65) | 12.78 (10.57–15.46) |
| Age, gender, calendar period adjusted, HR (95% CI) | 1 (reference) | 1.11 (0.85–1.46) | 1.43 (1.15–1.76) |
| Sociodemographics adjusted HR b (95% CI) | 1 (reference) | 1.10 (0.84–1.46) | 1.39 (1.12–1.73) |
| Stratified adjusted model b HR (95% CI) | |||
| Gender, P c | 0.394 | 0.068 | |
| Men | 1 (reference) | 1.28 (0.83–1.97) | 1.74 (1.26–2.39) |
| Women | 1 (reference) | 1.00 (0.70–1.43) | 1.16 (0.86–1.57) |
| Age, P c | 0.960 | 0.013 | |
| 16–50 | 1 (reference) | 1.13 (0.34–3.71) | 3.20 (1.62–6.31) |
| >50 | 1 (reference) | 1.10 (0.83–1.45) | 1.29 (1.03–1.63) |
| Period, P c | 0.009 | 0.323 | |
| 2000–2004 | 1 (reference) | 1.06 (0.64–1.77) | 1.61 (1.12–2.30) |
| 2005–2009 | 1 (reference) | 0.69 (0.42–1.14) | 1.13 (0.80–1.60) |
| 2010–2014 | 1 (reference) | 1.92 (1.24–2.98) | 1.57 (1.01–2.43) |
| Health and health behaviour adjusted HRd (95% CI) | 1 (reference) | 1.05 (0.80–1.39) | 1.22 (0.98–1.52) |
| Suicide | |||
| Deaths, n | 33 | 36 | 33 |
| PYAR, 10 000s | 64.88 | 6.02 | 8.29 |
| Rate, per 10000 PYAR (95% CI) | 0.51 (0.36–0.72) | 5.98 (4.31–8.29) | 3.98 (2.83–5.60) |
| Age, gender, calendar period adjusted, HR (95% CI) | 1 (reference) | 12.94 (8.04–20.82) | 7.90 (4.84–12.90) |
| Sociodemographics adjusted, b HR (95% CI) | 1 (reference) | 12.66 (7.79–20.58) | 7.21 (4.26–12.19) |
| Stratified adjusted model b HR (95% CI) | |||
| Gender, P c | 0.524 | 0.810 | |
| Men | 11.10 (5.85–21.06) | 6.91 (3.71–12.87) | |
| Women | 1 (reference) | 15.27 (7.11–32.78) | 7.90 (3.15–19.79) |
| Age, P c | 0.478 | 0.499 | |
| 16–50 | 1 (reference) | 13.20 (7.58–22.99) | 8.17 (4.45–14.99) |
| >50 | 1 (reference) | 8.79 (3.30–23.41) | 5.47 (2.00–14.90) |
| Period, P c | 0.306 | 0.772 | |
| 2000–2004 | 1 (reference) | 6.60 (1.53–28.48) | 10.18 (3.25–31.90) |
| 2005–2009 | 1 (reference) | 9.50 (4.74–19.07) | 6.26 (3.05–12.88) |
| 2010–2014 | 1 (reference) | 18.88 (8.62–41.32) | 7.50 (2.94–19.12) |
PYAR, person-years at-risk.
a. General population comparison group of up to six individuals without bipolar disorder or schizophrenia matched for gender, age group, primary care practice.
b. Age, gender, calendar period, area-level deprivation, ethnicity, number of primary care contacts.
c. Test for heterogeneity.
d. Adjusted for age, gender, calendar period, area-level deprivation, ethnicity, smoking, high cholesterol, high blood pressure, body mass index, diabetes mellitus and primary care contacts.
CVD diagnosis rates were elevated in both bipolar disorder and schizophrenia groups compared with the comparison group after adjustment for age, gender, calendar year, area-level deprivation, ethnicity and average number of visits to the physician during follow-up (HR = 1.41, 95% CI 1.26–1.58, and HR = 1.36, 95% CI 1.24–1.50, respectively) (online Table DS1). Records of smoking, hypercholesterolemia, hypertension, BMI and diabetes mellitus in the patient notes did not fully explain the increased rates (HR = 1.26, 95% CI 1.12–1.41 in bipolar disorder, and HR = 1.22, 95% CI 1.11–1.35 in schizophrenia after adjustment for these confounders).
Men were significantly more likely to receive a CVD diagnosis than women in both bipolar disorder and schizophrenia groups (HR = 1.55, 95% CI 1.31–1.82 and HR = 1.37, 95% CI 1.20–1.57, respectively). Women with either SMI diagnosis did not have elevated rates relative to women in the general population (online Table DS1). Increased rates of CVD mortality in those aged 16–50 were not reflected in equally increased rates of CVD diagnosis (HR = 1.66, 95% CI 1.29–2.15).
Suicide and self-harm
After accounting for age, gender, calendar year, area-level deprivation, ethnicity and average number of visits to the physician per year of follow-up, the suicide rate in those with bipolar disorder was 12.66 (95% CI 7.79–20.58) times that of the comparison group (Table 3). The adjusted HR in the group with schizophrenia was 7.21 (95% CI 4.26–12.19). Increased suicide rates were observed in both bipolar disorder and schizophrenia irrespective of gender, age or calendar period. However, because of the low event rate, there was no evidence of differences by gender, age group or time period (Table 3).
Self-harm rates were elevated in both bipolar disorder (HR = 25.24, 95% CI 23.63–29.96) and schizophrenia (HR = 22.14, 95% CI 19.58–25.03) after adjusting for sociodemographic characteristics and physician visits (online Table DS1). Self-harm rates did not differ by gender in either bipolar disorder (P = 0.096) or schizophrenia (P = 0.735). Self-harm was dramatically elevated in those aged 16–50 with a diagnosis of bipolar disorder (HR = 55.74, 95% CI 45.35–68.52) and schizophrenia (HR = 52.07, 95% CI 42.43–63.92). Graphical inspection of the Schoenfeld residuals revealed no violations of the assumption of proportional hazards.
Discussion
Main findings
In this cohort of over 17 000 people with bipolar disorder and over 22 000 with schizophrenia, we found decreasing rates of all-cause mortality for both disorders since 2000. However, the HRs for mortality relative to a matched general population comparison group increased from the mid-2000s. This suggests that the improvement in health in the general population is increasing more rapidly than in those with SMI, and health inequalities are growing. This is despite a greater focus on this problem at a national and international level, in specialist, primary care and public health settings.
After accounting for sociodemographic characteristics, over the 15-year follow-up period, the rate of all-cause mortality in those with bipolar disorder was 1.77 times the general population (95% CI 1.67–1.88) and in individuals with schizophrenia was 2.08 times greater (1.98–2.19). These results are slightly lower than those from meta-analyses in the existing literature, Reference Saha, Chant and McGrath7,Reference Hayes, Miles, Walters, King and Osborn33 but are consistent with population-based samples and hide the underlying trend in increasing mortality relative to the general population over this period. Deaths in those aged 16–50 were markedly elevated for both bipolar disorder and schizophrenia. In individuals with schizophrenia, the risk of dying before 50 of CVD is strikingly elevated, and CVD is infrequently diagnosed in advance of the terminal event, especially in women. Increased CVD death rates in those with schizophrenia were explained by conventional CVD risk factors (smoking, BMI, hypercholesterolemia, hypertension and type 2 diabetes mellitus), but increased CVD morbidity rates were not. Deaths identified as suicide were rare in the cohort and as such, we could not state which SMI has a more elevated risk. However, both diagnoses convey a dramatically increased risk, relative to the general population.
Interpretation of our findings
During the study period, a number of factors could have differentially influenced mortality in people with bipolar disorder and schizophrenia compared with the general population. There is evidence that addressing negative health behaviours has been more effective in the general population, for example, population-level smoking cessation programmes have had less of an impact on people with SMI. Reference Lawrence and Kisely59 Although it has been suggested that second-generation antipsychotics reduce mortality overall, Reference Tiihonen, Lönnqvist, Wahlbeck, Klaukka, Niskanen and Tanskanen6 polypharmacy and higher drug dosages may increase it. Reference Weinmann, Read and Aderhold60 Polypharmacy is increasingly common in bipolar disorder Reference Hayes, Prah, Nazareth, King, Walters and Petersen61 and may be contributing to the worsening CVD mortality compared with the general population in the 2010–2014 period. Wahlbeck and colleagues speculated that the reducing mortality gap seen in Nordic countries up until 2006 reflected the success of deinstitutionalisation. Reference Wahlbeck, Westman, Nordentoft, Gissler and Laursen9 Although deinstitutionalisation in the UK has been a success in terms of integrating people into wider society, it has been argued that there is now too little support for people living with bipolar disorder and schizophrenia in the community. Reference Fakhoury and Priebe62,Reference Green and Griffiths63 and this may be reflected in mortality rates. Research into the health effects of recession has suggested that consequences will be most severe for the poorest groups in society and will have the most impact where social safety-nets are lacking and public hardship grows rapidly. Reference Cooper64,Reference Riumallo-Herl, Basu, Stuckler, Courtin and Avendano65 Given this, we could hypothesise that policies made in the UK following the 2008 financial crash (i.e. austerity) have had the hardest impact on those with SMI. A comprehensive understanding of the mechanisms by which SMI shortens life remains elusive, but potentially constitutes a syndemic including psychiatric and physical comorbidity, substance misuse, clustering of adverse social factors and lifestyle behaviours. Reference Liu, Daumit, Dua, Aquila, Charlson and Cuijpers66
Strengths and limitations
A major strength of this study, beyond its size and length of follow-up, is that the results are generalisable to individuals living with bipolar disorder and schizophrenia in the UK (whether or not they have been in-patients in psychiatric hospitals), because of the representativeness of the THIN database. The exposure groups (bipolar disorder and schizophrenia) Reference Nazareth, King, Haines, Rangel and Myers44,Reference Hardoon, Hayes, Blackburn, Petersen, Walters and Nazareth45 and the outcomes (mortality, CVD deaths, CVD, suicide and self-harm) Reference Ruigómez, Martín-Merino and Rodríguez46,Reference Hammad, McAdams, Feight, lyasu and Dal Pan47,Reference Arana, Wentworth, Ayuso-Mateos and Arellano50,Reference Hayes, Pitman, Marston, Walters, Geddes and King51 have been well validated. Despite this, there is potential under-recording of cause-specific deaths and morbidity. Cause of death from death certificates would have improved the study, but this information was not available. In particular, suicide rates were lower than would be expected from Office for National Statistics (ONS) data 67 but we would not expect these to be differential by diagnostic group and our HRs reflect recent standardised mortality ratio estimates from the UK. Reference Hoang, Stewart and Goldacre4,Reference Brown, Kim, Mitchell and Inskip68 The proportion of deaths from CVD in the general population comparison group (7%) is consistent with 2014 ONS data, where in a similar age range, CVD mortality represented 6% of all deaths. Reference Townsend, Bhatnagar, Wilkins, Wickramasinghe and Rayner69 There is no under-recording of all-cause mortality. Reference Maguire, Blak and Thompson39 This study used a mixture of prevalent and incident cases of bipolar disorder and schizophrenia. It has been stated that this is the ideal situation as it allows exploration of the effects of different durations of exposure and enhances information on long exposure durations. Reference Vandenbroucke and Pearce70 However, it means that there is the potential for both left truncation (that is, events occurring before we start to follow-up) and right censoring. Other cohort studies of this nature will invariably face the same problems.
Recording of smoking status, BMI, hypercholesterolemia, hypertension and type 2 diabetes mellitus may be incomplete and therefore there is potential for residual confounding. However, recording of a number of these CVD risk factors has been incentivised by the Quality and Outcomes Framework, Reference Doran, Kontopantelis, Valderas, Campbell, Roland and Salisbury11 and we have attempted to minimise ‘missingness’ by defining these as ever recorded during follow-up. Although this incentivisation potentially improved CVD recording after 2004, there is no evidence that this was differential with regard to bipolar disorder, schizophrenia or the comparison group. The measure of social deprivation included in the multivariable model was related to the area in which the individual lived, rather than their individual status. As such, this may not be sensitive enough to capture the impact of socioeconomic status on the association between SMI and mortality. However, our study goes further than others in this area, by adjusting for a number of recognised confounders, rather than simply using standardised mortality ratios that account for age and gender.
Implications
This study suggests that although there have been important reductions in overall mortality since 2000, interventions to improve health outcomes for those with bipolar disorder or schizophrenia have not reduced the mortality gap. Mortality trends in individuals with SMI are important indicators of outcome and quality of psychiatric and medical care. Reference Bushe, Taylor and Haukka8,Reference Ösby, Correia, Brandt, Ekbom and Sparén71 Our results underscore how continuous monitoring of mortality of people with bipolar disorder and schizophrenia, relative to the general population, might guide us in evaluating the impact of interventions to manage physical comorbidity, reduce disparities in medical care provision and prevent inequalities in background risk factors.







eLetters
No eLetters have been published for this article.