Clinical Implications:
-
▪ Dysfunctions of the dopamine, serotonin, and glutamate systems contribute to the development and pathophysiology of schizophrenia and bipolar mania.
-
▪ Most clinically effective atypical antipsychotics are known to interact with the serotonin system, modulate the glutamate system, and induce region-specific changes in serotonin and glutamate receptor levels.
-
▪ Aripiprazole, a dopamine D2/D3 receptor partial agonist, was evaluated for its long-term effects on dopamine, serotonin, and glutamate receptor levels in comparison to cariprazine, a potent dopamine D2/D3 receptor partial agonist with preferential binding for the D3 receptors, in rat forebrain regions.
-
▪ Aripiprazole produced less-pronounced increases in D3 receptors as compared to those previously reported for another dopamine D2/D3 receptor partial agonist, cariprazine. Aripiprazole and cariprazine treatment induced similar changes in 5-HT1A, NMDA, and AMPA receptors in different rat forebrain regions, which suggests that these receptors constitute common targets that mediate the actions of both drugs.
-
▪ Similar to other atypical antipsychotic drugs, aripiprazole treatment induced significant decreases in cortical 5-HT2A receptors. In contrast, cariprazine failed to downregulate serotonin 5-HT2A receptors, which may suggest a minimal role for this receptor in mediating the actions of cariprazine.
-
▪ Cariprazine’s unique effects on dopamine and serotonin receptor subtypes, as well as its shared effects on glutamate receptor subtypes, may contribute to the improved clinical benefits of this novel atypical antipsychotic drug in the treatment of schizophrenia and bipolar mania.
Introduction
Dysfunctions in dopaminergic, serotonergic, and glutamatergic neurotransmissions are believed to play an important role in the pathophysiology and symptomatology of schizophrenia and other idiopathic psychotic disorders.Reference Baldessarini and Tarazi 1 – Reference Hu, MacDonald, Elswick and Sweet 4 In addition to their effects at dopamine receptor subtypes, atypical antipsychotics modulate serotonergic and glutamatergic neurotransmission, which may contribute to their clinical efficacy, safety, and tolerability.Reference Baldessarini and Tarazi 1 , Reference Meltzer and Massey 5 , Reference Ellenbroek and Cesura 6
The atypical antipsychotics cariprazine and aripiprazole are both dopamine D2/D3 receptor partial agonists; however, the drugs differ in that cariprazine is D3 receptor-preferring and has moderate affinity for serotonin 5-HT1A receptors.Reference Kiss, Horváth and Némethy 7 Moreover, cariprazine, but not aripiprazole, demonstrated high and balanced occupancy of both D2 and D3 receptors at pharmacologically effective and antipsychotic doses in both animalsReference Gyertyán, Kiss and Sághy 8 and schizophrenia patients.Reference Slifstein, Abi-Dargham and D’Souza 9 In animal behavioral models, cariprazine showed procognitiveReference Neill, Grayson, Kiss, Gyertyan, Ferguson and Adham 10 and antidepressant-like effects,Reference Papp, Gruca, Lason-Tyburkiewicz, Adham, Kiss and Gyertyán 11 suggesting the potential benefits of cariprazine for both the cognitive deficits and depressive symptoms of schizophrenia. These effects of cariprazine are likely to be mediated by D3 receptors, since they were absent in transgenic knockout mice that lack expression of functional D3 receptors.Reference Zimnisky, Chang, Gyertyan, Kiss, Adham and Schmauss 12 , Reference Duman, Duric, Banasr, Adham, Kiss and Gyertyán 13 We recently reported that long-term treatment with cariprazine increased D2 and D3 receptor levels in selected rat forebrain regions, whereas other typical and atypical antipsychotics upregulated D2 receptors with no effects on D3 receptors.Reference Tarazi, Kiss, Choi, Adham and Gyertyán 14 In the present study, we quantified the long-term effects of aripiprazole on D2 and D3 receptors and compared them to findings previously obtained for cariprazine and other antipsychotics.
Previous studies in rats have shown that long-term treatments with the atypical antipsychotics olanzapine, risperidone, quetiapine, and asenapine increased serotonin 5-HT1A receptor levels in the medial prefrontal cortex (MPC) and dorsolateral frontal cortex (DFC) while decreasing 5-HT2A receptor levels in the same regions.Reference Tarazi, Zhang and Baldessarini 15 , Reference Tarazi, Moran-Gates, Wong, Henry and Shahid 16 Additionally, antipsychotic exposure altered the levels of ionotropic glutamate N-methyl-d-aspartic acid (NMDA) and α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid (AMPA) receptors in the caudate putamen, nucleus accumbens, and hippocampus.Reference Tarazi, Florijn and Creese 17 – Reference Tarazi, Choi, Gardner, Wong, Henry and Shahid 19 However, all of the tested atypical antipsychotics displayed combined dopamine D2/serotonin 5-HT2A receptor antagonism, which is a common feature of atypical antipsychotic drugs.Reference Baldessarini and Tarazi 1 , Reference Meltzer and Massey 5 The chronic effects of D2/D3 receptor partial agonists on serotonin and glutamate receptors require further investigation. Accordingly, we investigated the effects of long-term aripiprazole and cariprazine treatment on serotonin (5-HT1A and 5-HT2A) and glutamate (NMDA and AMPA) receptor subtypes in the rat forebrain.
Methods
Materials
Tritium autoradiography standards and [2,3-3H]R(+)-8-hydroxy-N,N-di-n-propyl-2-amino-1,2,3,4-tetrahydronaphthalene ([3H]8-OH-DPAT; 135 Ci/mmol), [3H]3-[2-[4-(4-fluorobenzoyl)piperidin-1-yl]ethyl]quinazoline-2,4(1H,3H)-dione ([3H]ketanserin; 50 Ci/mmol), [3H](+)-5-methyl-10,11-dihydro-5H-dibenzo[a,d]cyclohepten-5,10-imine ([3H]MK801; 23 Ci/mmol), and [3H]2-amino-3-(3-hydroxy-5-methyl-isoxazol-4-yl)propanoic acid ([3H]AMPA; 58 Ci/mmol) were obtained from Amersham Life Science (Arlington Heights, IL, USA). Kodak Biomax MR film was purchased from Thermo Fisher Scientific (Waltham, MA, USA). D-19 photographic developer and fixative were obtained from Eastman Kodak (Rochester, NY, USA).
Aripiprazole HCl and cariprazine HCl were donated by Forest Laboratories, Inc. (New York, NY, USA). Pargyline HCl, ketamine, 7-nitro-2,3-dioxo-1,4-dihydroquinoxaline-6-carbonitrile (CNQX), ketanserin tartrate, prazosin, tetrabenzaine, serotonin (5-HT), methysergide, L-glutamate, glycine, and spermine were obtained from Sigma Research Biochemicals International (Natick, MA, USA). Cation hydrochlorides, guanosine-5′-triphosphate sodium (GTP), and tris-(hydroxymethyl)-aminomethane-HCl (Tris) were obtained from Sigma Chemicals (St. Louis, MO, USA).
Animals
Male Sprague-Dawley rats (Charles River Laboratories, Wilmington, MA, USA), initially weighing 200–225 g, were maintained under controlled artificial daylight (7:00 am–7:00 pm), temperature, and humidity, with free access to standard food and tap water in a USDA-inspected, veterinarian-supervised, small-animal research facility of the Mailman Research Center, with approval by the Institutional Animal Care and Use Committee (IACUC) of McLean Hospital.
Treatment and tissue preparation
For 28 days, 7 groups of rats (8 animals/group) received a daily intraperitoneal injection of vehicle control (distilled water, 1 ml/kg) or active treatment of aripiprazole (2, 5, or 15 mg/kg) or cariprazine (0.06, 0.20, or 0.60 mg/kg). The selected doses were active in different behavioral paradigms.Reference Gyertyán, Kiss and Sághy 8 , Reference Gao, Qin and Li 20 No gross effects on motor behaviors or significant changes in body weight were observed after repeated treatment with the different doses of aripiprazole or cariprazine compared with vehicle-treated animals.
At the end of the treatment period, rats were decapitated 24 hours after the last injection; their brains were quickly removed, frozen in chilled isopentane, and stored at –80°C. Brain regions assessed included the medial prefrontal cerebral cortex (mPFC), dorsolateral frontal cortex (DFC) (3.2–4.2 mm anterior to bregma), nucleus accumbens (NAc) (1.7–2.2 mm anterior to bregma), medial caudate putamen (CP–M) and lateral caudate putamen (CP–L) (0.7–1.2 mm anterior to bregma), hippocampus (HIPP), and entorhinal cortex (0.2–0.7 mm anterior to bregma).Reference Paxinos and Watson 21 The selected extrapyramidal, limbic, and cortical regions of interest mediate the cognitive, emotional, and motor behaviors that are typically disturbed in patients with schizophrenia and other psychotic disorders.Reference Baldessarini and Tarazi 1
Receptor autoradiography and image analysis
Coronal sections (10 μm) of these samples were cut in a cryostat at –20°C, mounted on gelatin-coated microscope slides, and stored at –80°C. To minimize experimental variability, brain sections from all vehicle- and drug-treated animals used for a given receptor subtype assay were evaluated at the same time.
Dopamine receptor autoradiography
For the D2 and D3 assays, sections were first preincubated for 1 h at room temperature (RT). We have previously shown that this preincubation step is effective in minimizing the effects of endogenous ligands and the potential interference of residual drugs.Reference Florijn, Tarazi and Creese 22
D2 receptor binding
Sections were first preincubated for 1 h at RT in 50 mM Tris-HCl buffer (pH 7.4) containing 120 mM NaCl, 5 mM KCl, 2 mM CaCl2, and 1 mM MgCl2. Sections were incubated for 1 h at RT in the same buffer containing 1.0 nM [3H]nemonapride with 0.5 µM DTG and 0.1 µM pindolol to mask sigma (σ1,2) and 5-HT1A sites, respectively. Nonspecific binding (NSB) was determined with 10 µM S(–)-sulpiride. After incubation, slides were washed twice for 5 min in ice-cold buffer, dipped in cold water, and air-dried. Radiolabeled slides and [3H] standards were exposed to Hyperfilm for 4 weeks at 4°C.Reference Tarazi, Zhang and Baldessarini 23 , Reference Tarazi, Yeghiayan, Baldessarini, Kula and Neumeyer 24
D3 receptor binding
Sections were preincubated for 1 h in 50 mM Tris-HCl buffer (pH 7.4) containing 0.3 mM GTP and 40 mM NaCl (no MgCl2 added) to minimize labeling of the high-affinity agonist binding state of D2 receptors, and then incubated for 1 h in the same buffer containing 3 nM [3H]7-OH-DPAT, with 5 µM DTG to mask sigma sites. NSB was determined with 1 µM S(–)-eticlopride. After incubation, slides were washed (2×3 min) in ice-cold buffer and dried. Radiolabeled slides and [3H] standards were exposed to Hyperfilm for 6 weeks at 4°C.Reference Tarazi, Zhang and Baldessarini 23 , Reference Tarazi, Yeghiayan, Baldessarini, Kula and Neumeyer 24
Serotonin receptor autoradiography
5-HT1A receptor binding
Sections were preincubated for 1 h at RT in 50 mM Tris-HCl buffer (pH 7.6) containing ascorbic acid (0.1%, w/v), CaCl2 (4 mM), and the monoamine oxidase inhibitor pargyline-HCl (10 μM). Sections were then incubated for another 60 min at RT in fresh buffer containing 2.0 nM [3H]8-OH-DPAT. NSB was determined with 1 μM 5-HT. After incubation, slides were washed (2×5 min) in ice-cold buffer and air-dried. Radiolabeled slides and [3H] standards were exposed to Hyperfilm for 5 weeks at 4°C.Reference Tarazi, Zhang and Baldessarini 15
5-HT2A receptor binding
Sections were preincubated for 60 min at RT in 50 mM Tris-HCl buffer (pH 7.7), and then incubated for 1 h at RT in fresh buffer containing 3.0 nM [3H]ketanserin, 1 μM prazosin (to block α1-adrenoceptors), and 100 nM tetrabenazine (to block monoaminergic nerve terminal sites). NSB was determined with 1 μM methysergide. After incubation, slides were washed (2×30 min) in ice-cold buffer, dipped in cold water, and air-dried. Radiolabeled slides and calibrated [3H] standards were exposed to Hyperfilm for 5 weeks at 4°C.Reference Tarazi, Zhang and Baldessarini 15
Glutamate receptor autoradiography
NMDA receptor binding
Sections were preincubated for 60 min in 50 mM Tris-HCl buffer (pH 7.4) to remove endogenous ligand and then incubated for 150 min at RT in buffer containing 10 nM [3H]MK-801, 100 μM L-glutamate, 100 μM glycine, 1 mM EDTA, and 75 μM spermine to enhance the binding of [3H]MK-801 to NMDA receptors. NSB was determined with 20 μM ketamine. After incubation, slides were washed in ice-cold buffer (2×20 min) and then air-dried. Radiolabeled slides and calibrated [3H] standards were exposed to Hyperfilm for 4 weeks at 4°C.Reference Tarazi, Florijn and Creese 17 , Reference Tarazi, Baldessarini, Kula and Zhang 18
AMPA receptor binding
Sections were preincubated for 60 min in 50 mM Tris-HCl buffer (pH 7.3) at 4°C and then incubated in fresh buffer containing 20 nM [3H]AMPA for 60 min at 4°C.Reference Tarazi, Baldessarini, Kula and Zhang 18 NSB was determined with 20 μM unlabeled CNQX. After incubation, slides were washed (3×15 sec) in ice-cold buffer and air-dried. Radiolabeled slides and [3H] standards were exposed to Hyperfilm for 3 weeks at 4°C.Reference Tarazi, Florijn and Creese 17 , Reference Tarazi, Baldessarini, Kula and Zhang 18
Image analysis
Biomax MR films were developed and fixed in Kodak D-19 for 5 min at RT; images were quantified using an MCID image analyzer and MCID–M4 Imaging Research software (St. Catharines, Ontario, Canada). A calibration curve was generated from [3H]-microscale standards that were exposed along with tissue sections. Regions of interest were outlined and their optical density measured, with the left and right sides of two contiguous sections representing total binding and matching samples representing nonspecific binding. Four determinations were averaged for each subject sample. Optical density of sampled regions was measured, and the amount of ligand bound was calculated as nCi/mg tissue and expressed as fmol bound/mg tissue.
Statistical analysis
Receptor binding data were analyzed first for overall effects of drug versus vehicle for all receptor types and brain regions using multiple regression modeling methods. Density measures were logarithmically transformed to achieve more Gaussian-like distributions before regression modeling. Model goodness-of-fit was checked using partial residual plot methods. Since individual brain specimens provided receptor-binding data for several brain regions, resulting in incomplete independence across observations, we used robust standard error estimates to adjust for this clustering effect. This estimation method permits relaxation of the assumption of independence of all observations and requires only that the observations be independent across specimens.Reference Greene 25 Estimates of interaction effects were employed for post-hoc tests of drug effects for specific receptors and brain regions, with adjustment of p values obtained from the regression analyses estimating these multiple comparisons by the standard method of Sidák.Reference Winer, Brown and Michels 26 Data are presented as means ± standard error of the mean (SEM). Comparisons were considered significant at p<0.05 (two-tailed test).
Results
Long-term effects of aripiprazole on dopamine receptors
Aripiprazole increased D2 receptor levels in mPFC (29–48%). However, only the high dose of aripiprazole (15 mg/kg) increased D2 receptors in NAc (40%), medial (35%), and lateral (50%) CPu, and HIPP (57%) (Table 1). Chronic aripiprazole treatment (5 and 15 mg/kg) significantly increased D3 receptors in the olfactory tubercle (OT) (15–37%) and Islands of Calleja (IC) (18–41%) (Table 1). However, aripiprazole, unlike cariprazine, did not alter the levels of D3 receptors in the shell of the NAc at any of the doses tested (Table 1).
Table 1 Changes in forebrain receptors following long-term exposure to cariprazine and aripiprazole
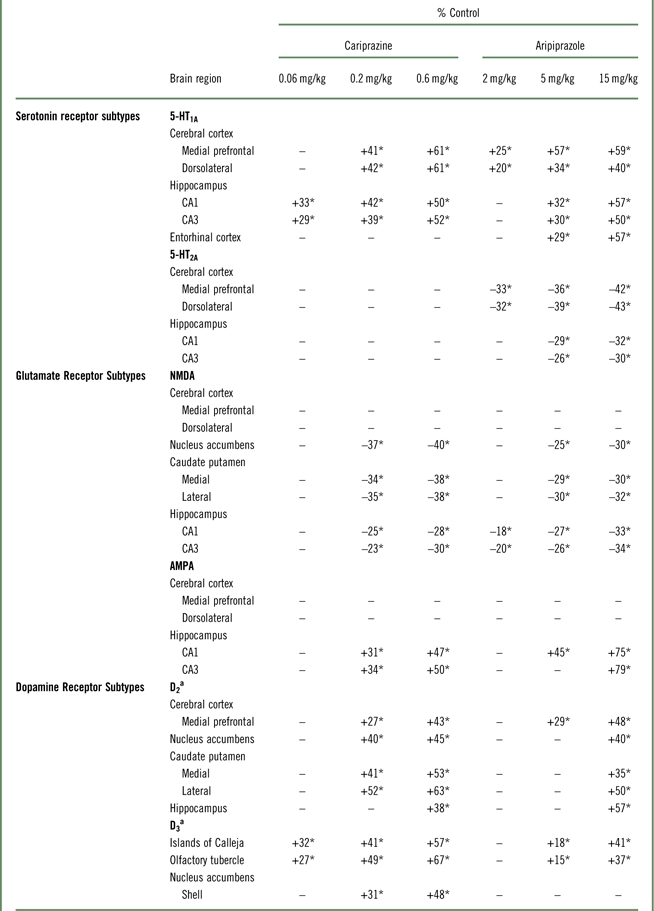
*p<0.05 versus respective control; –=not significantly different from respective control.
a Cariprazine data are reproduced from Choi et al.Reference Choi, Adham, Kiss, Gyertyán and Tarazi 27
Long-term effects of aripiprazole and cariprazine on 5-HT1A receptors
Aripiprazole (2, 5, or 15 mg/kg) treatment dose-dependently increased 5-HT1A receptors in the MPC (24–59%) and DFC (20–39%). Higher doses of aripiprazole (5 and 15 mg/kg) also increased 5-HT1A receptors in the HIPP–CA1 (31 and 56%) and HIPP–CA3 (30 and 50%) regions (Table 1, Figure 1A). Similar to aripiprazole, cariprazine (0.06, 0.2, or 0.6 mg/kg) treatment dose-dependently increased 5-HT1A receptor binding in the hippocampal CA1 (33, 42, and 50%, respectively) and CA3 (29, 39, and 52%, respectively) regions. In addition, repeated treatment with higher doses of cariprazine (0.2 and 0.6 mg/kg) increased 5-HT1A receptors in the MPC (41 and 61%) and DFC (42 and 61%) (Table 1, Figure 1B).
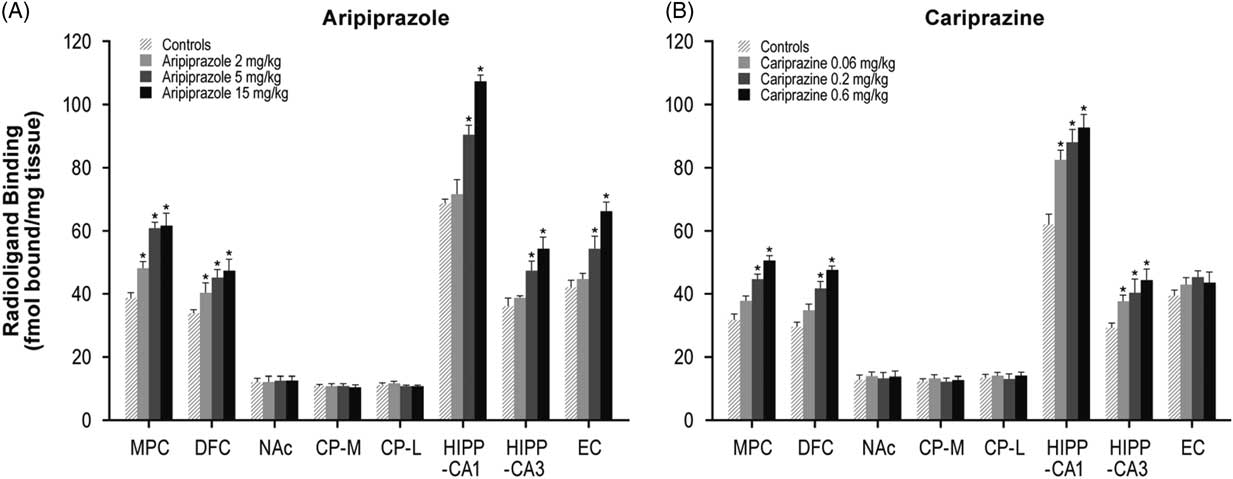
Figure 1 Long-term effects of cariprazine and aripiprazole on serotonin 5-HT1A receptors. Data are mean±SEM (n=8/group). * p<0.05 vs. control. CP–L indicates caudate putamen–lateral; CP–M=caudate putamen–medial; DFC=dorsolateral frontal cortex; EC=entorhinal cortex; HIPP=hippocampus; MPC=medial prefrontal cortex; NAc=nucleus accumbens.
Long-term effects of aripiprazole and cariprazine on 5-HT2A receptors
Aripiprazole (2, 5, or 15 mg/kg) decreased 5-HT2A receptors in the MPC (33, 36, and 42%, respectively) and DFC (32, 39, and 43%, respectively). Aripiprazole (5 and 15 mg/kg) also decreased 5-HT2A receptors in the HIPP–CA1 (28 and 32%) and HIPP–CA3 (26 and 30%) regions (Table 1, Figure 2A). In contrast, long-term administration of cariprazine (0.06, 0.2, and 0.6 mg/kg) failed to alter 5-HT2A receptor levels in all forebrain regions examined (Table 1, Figure 2B).
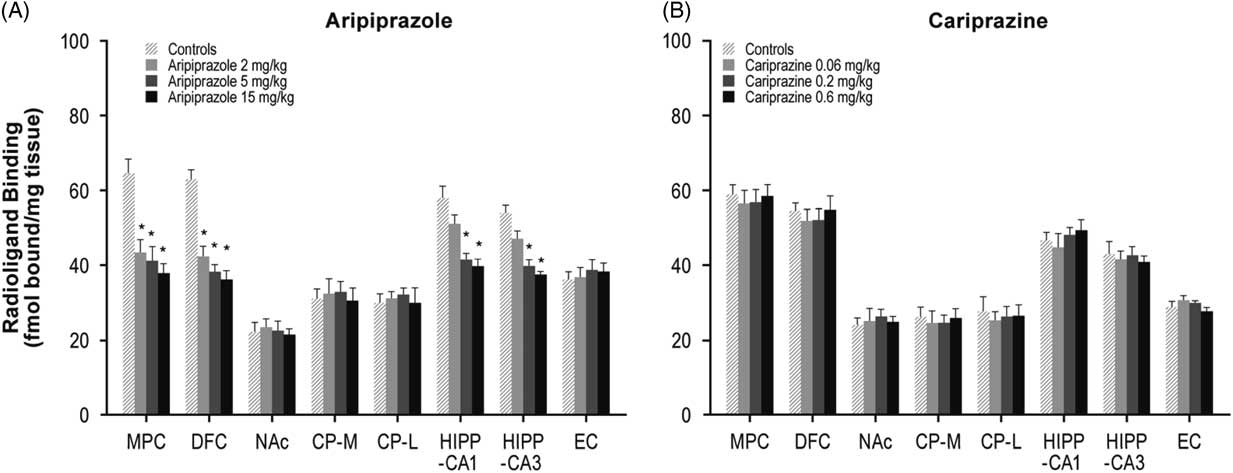
Figure 2 Long-term effects of cariprazine and aripiprazole on serotonin 5-HT2A receptors. Data are mean±SEM (n=8/group). * p<0.05 vs. control. CP–L indicates caudate putamen–lateral; CP–M=caudate putamen–medial; DFC=dorsolateral frontal cortex; EC=entorhinal cortex; HIPP=hippocampus; MPC=medial prefrontal cortex; NAc=nucleus accumbens.
Long-term effects of aripiprazole and cariprazine on NMDA receptors
Long-term administration of higher doses of aripiprazole (5 and 15 mg/kg) decreased NMDA receptors in the NAc (26 and 30%), CP–M (29 and 31%), and CP–L (30 and 32%) regions. Aripiprazole also dose-dependently decreased NMDA receptors in the HIPP–CA1 (18, 27, and 33%) and HIPP–CA3 (20, 25, and 34%) regions (Table 1, Figure 3A). Similarly, long-term administration of higher doses of cariprazine (0.2 and 0.6 mg/kg) significantly reduced NMDA receptor binding in the NAc (37 and 40%), CP–M (34 and 38%), and CP–L (35 and 38%) regions. In addition, repeated treatment with 0.2 and 0.6 mg/kg cariprazine decreased NMDA receptor binding in hippocampal CA1 (25 and 28%) and CA3 (23 and 30%) (Table 1, Figure 3B).
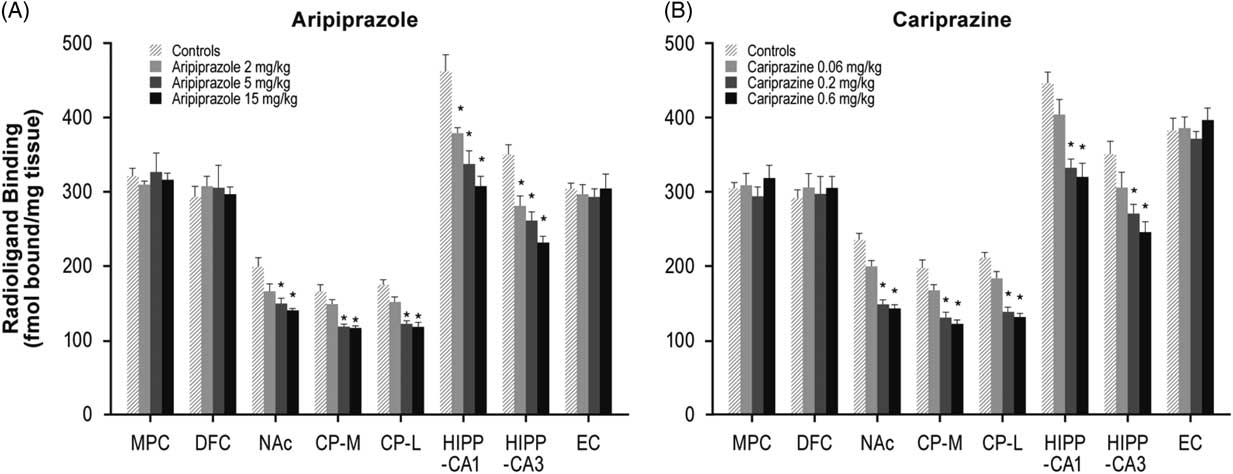
Figure 3 Long-term effects of cariprazine and aripiprazole on NMDA receptors. Data are mean±SEM (n=8/group). * p<0.05 vs. control. CP–L indicates caudate putamen–lateral; CP–M=caudate putamen–medial; DFC=dorsolateral frontal cortex; EC=entorhinal cortex; HIPP=hippocampus; MPC=medial prefrontal cortex; NAc=nucleus accumbens.
Long-term effects of aripiprazole and cariprazine on AMPA receptors
Aripiprazole at 5 and 15 mg/kg increased [3H]AMPA in the NAc (26 and 30%), CP–M (29 and 31%), and CP–L (30 and 32%) regions. Aripiprazole also dose-dependently increased AMPA receptors in the HIPP–CA1 (18, 27, and 33%) and HIPP–CA3 (20, 25, and 34%) regions (Table 1, Figure 4A). Repeated treatment with the higher doses of cariprazine (0.2 and 0.6 mg/kg) dose-dependently increased [3H]AMPA binding in the HIPP–CA1 (31 and 47%) and HIPP–CA3 (34 and 50%) regions. Cariprazine treatment did not alter AMPA receptors in the other brain regions examined (Table 1, Figure 4B).
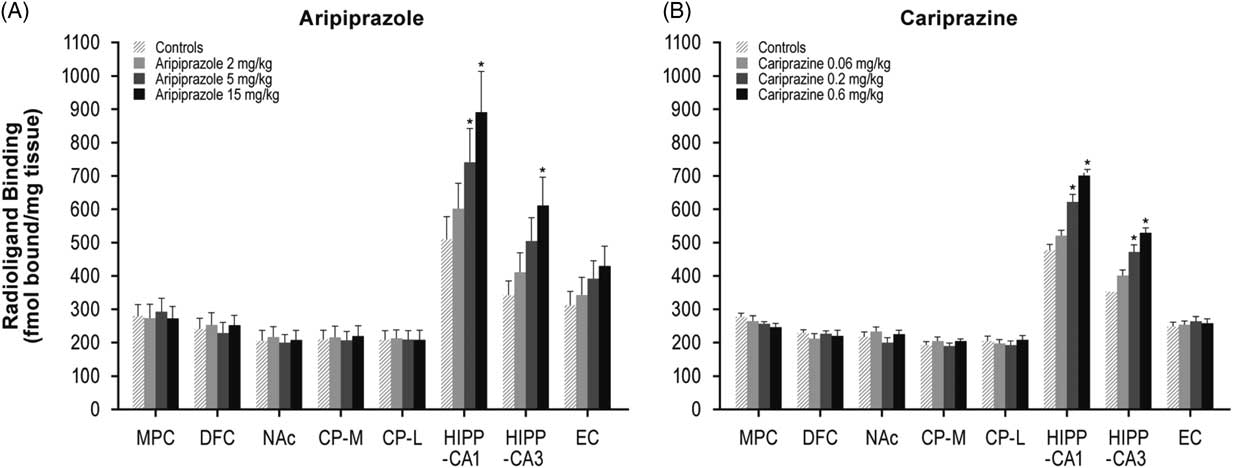
Figure 4 Long-term effects of cariprazine and aripiprazole on AMPA receptors. Data are mean±SEM (n=8/group). * p<0.05 vs. control. CP–L indicates caudate putamen–lateral; CP–M=caudate putamen–medial; DFC=dorsolateral frontal cortex; EC=entorhinal cortex; HIPP=hippocampus; MPC=medial prefrontal cortex; NAc=nucleus accumbens.
Discussion
Long-term effects of aripiprazole and cariprazine on dopamine D2/D3 receptors
Chronic aripiprazole treatment upregulated D2 receptor levels in different brain regions, with a maximum increase similar to that previously described for cariprazine and other antipyschotics (Table 1; Choi et al.Reference Choi, Adham, Kiss, Gyertyán and Tarazi 27 ). However, aripiprazole was at least 25-fold less potent than cariprazine (Table 1; Choi et al.Reference Choi, Adham, Kiss, Gyertyán and Tarazi 27 ). Aripiprazole also increased D3 receptor levels in selective brain regions; however, these effects were less pronounced (cariprazine had ~80-fold or greater potency for D3 receptors) and were produced in fewer brain regions than previously described for cariprazine (Table 1; Choi et al.Reference Choi, Adham, Kiss, Gyertyán and Tarazi 27 ). In particular, aripiprazole produced no effect at any dose tested on D3 receptors in the shell of the NAc, whereas cariprazine significantly increased these receptors in the same brain region. Increases in D3 receptor expression in the shell of the NAc has been proposed to be a common neurobiological mechanism of antidepressant treatments.Reference Lammers, Diaz, Schwartz and Sokoloff 28 These findings suggest that cariprazine is more potent than aripiprazole in upregulating D3 receptor subtypes, and that D3 receptors play a more prominent role in mediating the actions of cariprazine compared to aripiprazole. Moreover, the unique upregulation of dopamine D3 receptors in areas related to antidepressant action may offer therapeutic benefits against various mood disorders, including depression and the negative symptoms of schizophrenia.
Long-term effects of aripiprazole and cariprazine on serotonin 5-HT1A receptors
Repeated treatment with the three doses of aripiprazole (2, 5, and 15 mg/kg) and higher doses of cariprazine (0.2 and 0.6 mg/kg) dose-dependently increased the binding of the 5-HT1A radioligand [3H]8-OH-DPAT in MPC and DFC (Table 1, Figure 1A). This finding is consistent with the effects of the atypical antipsychotics olanzapine, risperidone, quetiapine, and asenapine on the same receptor in the same brain region.Reference Tarazi, Zhang and Baldessarini 15 , Reference Tarazi, Moran-Gates, Wong, Henry and Shahid 16 , Reference Newman-Tancredi, Assie, Leduc, Ormiere, Danty and Cosi 29 Both aripiprazole and cariprazine have substantial affinity for 5-HT1A receptors (Ki=5.6 and 2.6 nM, respectively) and display partial agonist activity against this receptor subtype.Reference Kiss, Horváth and Némethy 7 , Reference Shapiro, Renock and Arrington 30 Observed increases in cortical 5-HT1A receptors by both drugs may reflect direct blockade of 5-HT1A receptors or secondary changes resulting from more potent effects on dopamine D2 and D3 receptors.Reference Kiss, Horváth and Némethy 7 , Reference Tarazi, Kiss, Choi, Adham and Gyertyán 14 , Reference Shapiro, Renock and Arrington 30
Earlier studies reported that 5-HT1A receptors are more highly expressed in cortical and limbic brain regions than in the basal ganglia.Reference Pazos and Palacios 31 , Reference Pompeiano, Palacios and Mengod 32 In the frontal cortex, 5-HT1A receptors are located on neocortical glutamatergic pyramidal neurons that are typically implicated in different cognitive functions.Reference Francis, Pangalos, Pearson, Middlemiss, Stratmann and Bowen 33 Additional studies have suggested that 5-HT1A partial agonism can stimulate cortical dopamine releaseReference Rollema, Lu, Schmidt, Sprouse and Zorn 34 – Reference Bantick, Deakin and Grasby 36 and minimize the incidence of antipsychotic-induced extrapyramidal side effects.Reference Christoffersen and Meltzer 37 , Reference Daniel, Zimbroff, Potkin, Reeves, Harrigan and Lakshminarayanan 38 5-HT1A receptors have increasingly become attractive targets for novel agents to treat schizophrenia and other psychiatric disorders.Reference Millan 39 , Reference Celada, Bortolozzi and Artigas 40 Aripiprazole- and cariprazine-induced increases in cortical 5-HT1A receptors further validate the hypothesis that the 5-HT1A receptor may contribute, at least in part, to the beneficial therapeutic effects of these two agents as well as other dissimilar atypical antipsychotic drugs.Reference Tarazi, Zhang and Baldessarini 15
Repeated administration of aripiprazole (5 and 15 mg/kg) and cariprazine (0.06, 0.2 or 0.6 mg/kg) dose-dependently increased binding of the 5-HT1A radioligand [3H]8-OH-DPAT in hippocampal CA1 and CA3 regions. Asenapine is the only atypical antipsychotic that shares this effect, as other atypical antipsychotics, including olanzapine, risperidone, and quetiapine, did not alter 5-HT1A receptors in rat hippocampus.Reference Tarazi, Zhang and Baldessarini 15 , Reference Tarazi, Moran-Gates, Wong, Henry and Shahid 16 These findings suggest that aripiprazole and cariprazine can influence hippocampal 5-HT1A receptors and consequently normalize the increased hippocampal activity postulated to occur in schizophrenic patients and ameliorate the psychotic symptoms of the disease.Reference Heckers and Konradi 41
Long-term effects of aripiprazole and cariprazine on serotonin 5-HT2A receptors
Long-term treatment with all three aripiprazole doses significantly and equipotently decreased 5-HT2A receptor binding in the MPC and DFC regions. Aripiprazole has high affinity for 5-HT2A receptors (Ki=8.7 nM),Reference Shapiro, Renock and Arrington 30 and the decrease in 5-HT2A binding may reflect direct blockade of 5-HT2A receptors by the different doses of aripiprazole. We have previously reported that long-term administration of several atypical antipsychotics—including clozapine, olanzapine, risperidone, quetiapine, and asenapine—but not the typical antipsychotic haloperidol, resulted in decreased 5-HT2A receptor labeling in rat frontal cerebral cortex.Reference Tarazi, Zhang and Baldessarini 15 , Reference Florijn, Tarazi and Creese 22 , Reference O’Dell, La Hoste, Widmark, Shapiro, Potkin and Marshall 42 , Reference Kuoppamaki, Seppala, Syvalahti and Hietala 43 5-HT2A receptor downregulation may minimize the higher incidence of extrapyramidal side effects associated with typical antipsychotic drug treatment.Reference Baldessarini and Tarazi 1 , Reference Meltzer 44 , Reference Tarsy, Baldessarini and Tarazi 45 Aripiprazole (5 and 15 mg/kg) also decreased 5-HT2A receptors in the hippocampal CA1 and CA3 regions. Such downregulation may further contribute to the stabilization of dopamine neurotransmission in the limbic system and, consequently, to the improvement of affective and emotional behaviors typically disturbed in schizophrenia patients.Reference Baldessarini and Tarazi 1
In contrast, cariprazine failed to alter 5-HT2A receptor binding in all the forebrain regions examined. The lack of effect of cariprazine on 5-HT2A receptors may result from its lower in vitro affinity to these receptors, resulting in insufficient occupancy of 5-HT2A receptors to the level required to induce receptor changes. These findings suggest that unlike several of the atypical antipsychotics, which downregulate cortical 5-HT2A receptors,Reference Tarazi, Zhang and Baldessarini 15 , Reference Florijn, Tarazi and Creese 22 , Reference O’Dell, La Hoste, Widmark, Shapiro, Potkin and Marshall 42 , Reference Kuoppamaki, Seppala, Syvalahti and Hietala 43 5-HT2A receptors are less likely to contribute to the mechanism of action of cariprazine in vivo.
Long-term effects of aripiprazole and cariprazine on glutamate NMDA receptors
Continuous treatment with the higher doses of aripiprazole (5 and 15 mg/kg) and cariprazine (0.2 and 0.6 mg/kg) significantly decreased binding of [3H]MK-801 to NMDA receptors in the NAc and the medial and lateral CPu. These effects were similar to those previously reported for the atypical antipsychotics clozapine, olanzapine, risperidone, and asenapine, and the experimental atypical antipsychotic JL13, but not the typical agent haloperidol.Reference Tarazi, Florijn and Creese 17 – Reference Tarazi, Choi, Gardner, Wong, Henry and Shahid 19 , Reference Spurney, Baca, Murray, Jaskiw, Kleinman and Hyde 46 Aripiprazole- and cariprazine-induced reductions in striatal NMDA receptors may result from indirect interactions with dopamine and serotonin systems, both of which modulate glutamatergic neurotransmission.Reference Aghajanian and Marek 47 , Reference Carlsson, Waters, Holm-Waters, Tedroff, Nilsson and Carlsson 48 Such reductions in striatal NMDA receptors may reflect posttranscriptional modifications, since dissimilar atypical antipsychotics did not alter expression of mRNA-encoding NMDA-forming subunits in rat striatum.Reference Tascedda, Blom and Brunello 49 , Reference Tascedda, Lovati and Blom 50
We have demonstrated previously that acute and subchronic administration of cariprazine, as well as other atypical antipsychotics, reverses both NMDA receptor antagonist-mediated hypermobilityReference Gyertyán, Kiss and Sághy 8 and PCP-induced cognitive and social deficits in animal models,Reference Neill, Grayson, Kiss, Gyertyan, Ferguson and Adham 10 , Reference Zimnisky, Chang, Gyertyan, Kiss, Adham and Schmauss 12 , Reference Watson, King, Gyertyan, Kiss, Adham and Fone 51 indicating an ability to normalize abnormal glutamatergic transmission; however, the potential neurochemical and behavioral consequences of long-term aripiprazole- and cariprazine-induced reductions in striatal NMDA receptors are not clear. Studies reported that stimulation of NMDA receptors promoted typical antipsychotic-induced catalepsy, an animal model predictive of extrapyramidal side effects.Reference Schmidt and Bubser 52 In contrast, blockade of NMDA receptors attenuated the cataleptic effects of typical antipsychotic drugs.Reference Yoshida, Ono, Kizu, Fukushima and Miyagishi 53 Downregulation of striatal NMDA receptors by aripiprazole, cariprazine, and several dissimilar atypical antipsychotic drugs,Reference Tarazi, Baldessarini, Kula and Zhang 18 , Reference Florijn, Tarazi and Creese 22 as well as the subsequent modification of glutamate neurotransmission in the basal ganglia, may contribute, at least in part, to the benign extrapyramidal profile of atypical antipsychotic agents.Reference Tarsy, Baldessarini and Tarazi 45
Repeated treatment with aripiprazole and cariprazine reduced NMDA receptor binding in the hippocampal CA1 and CA3 regions. These effects are similar to the observed reduction in hippocampal NMDA receptors after repeated administration of the atypical antipsychotics olanzapine and risperidone.Reference Tarazi, Baldessarini, Kula and Zhang 18 Reductions in hippocampal NMDA receptors may normalize the abnormalities in glutamatergic neurotransmission postulated to occur in the hippocampus of schizophrenia patients,Reference Tsai and Coyle 54 and thus subsequently improve their psychotic symptoms.Reference Gao, Sakai, Roberts, Conley, Dean and Tamminga 55
Long-term effects of aripiprazole and cariprazine on glutamate AMPA receptors
The three doses of aripiprazole and the higher doses of cariprazine (0.2 and 0.6 mg/kg) dose-dependently increased AMPA receptors in the hippocampal CA1 and CA3 regions. These findings are in agreement with the atypical antipsychotic asenapine, which induced increases in hippocampal AMPA receptors.Reference Tarazi, Choi, Gardner, Wong, Henry and Shahid 19 Such increases in AMPA receptors may result from the actions of both aripiprazole and cariprazine on D2 receptors, which are coexpressed with AMPA in rat brains.Reference Ariano, Larson, Noblett, Sibley and Levine 56 Increases in hippocampal AMPA receptors may further contribute to the improvement of psychotic symptoms in schizophrenia patients. Interestingly, long-term administration of aripiprazole and cariprazine produced opposite effects on NMDA versus AMPA receptors, suggesting that ionotropic glutamate receptors may respond differently to repeated treatment with partial D2/D3 receptor agonists.
Conclusions
Long-term treatment with aripiprazole induced increases in D3 receptors in OT and IC. In contrast, repeated treatment with cariprazine produced more profound increases in D3 receptors in OT and IC, and extended its effect to induce increases in D3 receptors in the shell of the NAc. These findings are in agreement with the potent affinity of cariprazine at D3 receptors, and provide a distinction in the action mechanisms of cariprazine versus aripiprazole in selectively targeting D3 receptors as leading sites that mediate the beneficial actions of cariprazine.
Repeated treatment with aripiprazole and cariprazine induced similar changes in 5-HT1A, NMDA, and AMPA receptors in different rat forebrain regions. In contrast, aripiprazole downregulated 5-HT2A receptors in the frontal cortex, an effect not seen with cariprazine. Accordingly, 5-HT2A receptors may contribute more to the molecular actions of aripiprazole and less to the actions of cariprazine. In conclusion, the combined effects of cariprazine on serotonin and glutamate receptors, together with its unique actions on dopamine D3 receptors, may confer the clinical benefits, safety, and tolerability of cariprazine in improved treatment of schizophrenia and bipolar mania.
Disclosures
Yong Kee Choi hereby states that he has no conflicts of interest to declare. Nika Adham is an employee of Allergan. Béla Kiss is an employee of Gedeon Richter Plc. István Gyertyán was an employee of Gedeon Richter Plc at the time of the study. Frank I. Tarazi has received research grants from Forest Laboratories LLC, H. Lundbeck A/S, and Shire Plc.







