In developed countries most children consume too much Na with intakes frequently exceeding dietary recommendations( Reference Brown, Tzoulaki and Candeias 1 ). In addition to the more well-established links between Na intake and blood pressure( Reference He and MacGregor 2 ), Na has recently been associated with obesity risk in children and adolescents( Reference Grimes, Riddell and Campbell 3 – Reference Zhu, Pollock and Kotak 8 ). Early work in this area focused on the indirect association of Na intake with obesity through the greater consumption of sugar-sweetened beverages (SSB)( Reference Grimes, Riddell and Campbell 3 – Reference He, Marrero and MacGregor 5 , Reference Karppanen and Mervaala 9 ), which are a known risk factor for weight gain in children( Reference Hu 10 ). Across these population-based cross-sectional studies Na intake was found to be positively associated with total fluid intake as well as SSB intake in children and adolescents from the UK( Reference He, Marrero and MacGregor 5 ), the USA( Reference Grimes, Wright and Liu 4 ) and Australia( Reference Grimes, Riddell and Campbell 3 ). Informed by experimental evidence in animals( Reference Stricker, Hoffmann and Riccardi 11 ) and adults( Reference He, Markandu and Sagnella 12 , Reference Stachenfeld 13 ), which shows the activation of thirst and fluid intake to regulate body fluid balance when Na intake is increased, the observed association between Na and SSB intake in children is plausible.
However, in contrast to these findings, other studies have reported positive associations between Na intake and measures of adiposity independent of energy intake (EI)( Reference Libuda, Kersting and Alexy 6 – Reference Zhu, Pollock and Kotak 8 ). In Korean children Na density (Na intake (mg)/food intake (g) per d) was positively associated with obesity and abdominal obesity( Reference Yoon and Oh 7 ), and in US adolescents( Reference Zhu, Pollock and Kotak 8 ) Na intake was positively associated with BMI, waist circumference and percentage of body fat (%BF), independent of energy and SSB intake. In all of the aforementioned studies Na intake was assessed using dietary recall or food diary methods. These methods do not measure the amount of Na from table or cooking salt and therefore tend to underestimate total daily Na intake( Reference Espeland, Kumanyika and Wilson 14 ). To date, only one paediatric study has used 24-h urinary Na excretion to assess the relationship between total daily Na (i.e. food sources and discretionary salt) intake and obesity risk( Reference Libuda, Kersting and Alexy 6 ). In this study of German children 24-h urinary Na excretion was positively associated with BMI z score and %BF. Furthermore, the association remained after the adjustment of total EI or SSB intake. Overall, there is a growing evidence base to indicate that Na intake is associated with measures of adiposity in children. However, there is limited evidence using a reliable measure of total daily Na intake. Given the tracking phenomenon of overweight and obesity from childhood across the lifespan( Reference Singh, Mulder and Twisk 15 ), understanding dietary determinants of obesity is of paramount importance. Hence, the aim of this study was to assess the association between total daily Na intake, as assessed by 24-h urinary Na excretion, and measures of adiposity, including BMI z score, weight category and abdominal obesity, in a sample of Australian schoolchildren aged 4–12 years.
Methods
Data for this analysis come from the cross-sectional Salt and Other Nutrient Intakes in Children study, and the methods( Reference Grimes, Baxter and Campbell 16 ) have been reported previously.
Study invitations were sent to a convenience sample of 509 government and non-government schools, and fifty-six schools (11 %) agreed to participate. Within participating schools consent was obtained from 852 children (response rate=6 %); forty-one children withdrew from the study and children who did not agree to attend an off-school campus data collection day (n 25) or who were aged >13 years (n 6) were excluded, leaving 780 participants. The Deakin University Research Ethics Committee (Project No: EC 62-2009) approved the study. Written informed consent was obtained from the primary caregiver and the child.
Measures
Data collection occurred between June 2010 and May 2013. Parents completed a questionnaire reporting on children’s date of birth and sex. Socio-Economic Indexes for Areas, Index of Relative Socio-Economic Disadvantage( 17 ), was used to group schools, based on school postcode, into tertiles of socio-economic disadvantage. This marker was used to define socio-economic status (SES), whereby the participant was grouped as either low, mid or high SES with reference to the tertile that the school they attended fell within.
24-h urine collection
Details of the 24-h urine collection procedures have been previously described( Reference Grimes, Baxter and Campbell 16 ). In brief, children could opt to commence the 24-h urine collection at the start of a school day (i.e. approximately 09.00 hours Monday to Friday) or at any suitable time on a non-school day (i.e. weekends, public holidays and school holidays). At the commencement of the 24-h urine collection, children were instructed to empty their bladder, discard this urine and note this as the start time. Following this, all urine voided was collected up until the corresponding 24-h finish time. Parents and children were provided with written instructions on how to complete the 24-h urine collection. Parents were asked to report any missed collections or spillages on a urine collection slip, which was returned with the 24-h urine sample. Returned 24-h urine samples were transported to an accredited commercial pathology laboratory (Dorevitch Pathology) for urinalysis. Urinary Na and K concentration was assessed using indirect ion selective electrodes, and urinary creatinine concentration was assessed using the Jaffe reaction( Reference Jaffe 18 ) on the Siemens Advia 2400 analyser (Siemens Healthcare). The CV for Na and K was <1 % and for creatinine it was 3·25 %. The molecular weights of Na (23 g/mol) and sodium chloride (58·5 g/mol) were used to convert laboratory values in mmol of Na to mg( 19 ). If the duration of the collection was not exactly 24 h but within 20–28 h, urinary electrolytes, creatinine and total volume were standardised to a 24-h period (i.e. (24 h/urine duration (h))×urinary measure). Urine samples were considered incomplete if collection time was <20 or >28 h, total volume was <300 ml, the participant reported missing >1 collection, or urinary creatinine excretion was <0·1 mmol/kg body weight per d( Reference Grimes, Baxter and Campbell 16 ). Of the 780 participants, 763 returned urine specimens and 667 of these were deemed complete( Reference Grimes, Baxter and Campbell 16 ). One extreme outlier for urinary Na excretion (mean 531 (sd 8·7) mmol) was excluded, leaving an analytical sample of 666. There were no differences in age, sex, SES or weight category between completers and non-completers of the urine collection.
Anthropometric measurements
Height, weight and waist circumference were measured by trained research staff following standard protocols( Reference Grimes, Baxter and Campbell 16 ). BMI values were converted to age- and sex-adjusted BMI z scores using the 2000 US Centers for Disease Control and Prevention growth charts( Reference Flegal and Cole 20 , Reference Kuczmarski, Ogden and Guo 21 ). Participants were grouped into weight categories (underweight, healthy weight, overweight, obese) using the International Obesity Taskforce BMI reference cut-offs for children( Reference Cole 22 , Reference Cole, Flegal and Nicholls 23 ). The waist:height ratio (WHtR), which is not dependent on age or sex references, was used as a marker of central adiposity( Reference Garnett, Baur and Cowell 24 ). This was calculated as waist circumference (cm) divided by height (cm). A WHtR ≥0·5 has been associated with cardiovascular risk factors in the paediatric population( Reference Garnett, Baur and Cowell 24 , Reference Kahn, El ghormli and Jago 25 ), and this cut-off was used to define abdominal obesity( Reference Garnett, Baur and Cowell 24 ).
Dietary intake
One face-to-face three-pass 24-h dietary recall was completed with the child on the day of the testing procedures completed at the school site. This method includes the following stages: (i) provide a quick list of all foods and beverages consumed from midnight to midnight on the day before the interview; (ii) probe questions relevant to each quick list item to gather more detailed information – for example, the amount consumed, brand name, additions to the food item; and (iii) a recall review to cross-check information. In this analysis we report intakes of energy (MJ/d) and SSB (g/d) in the subsample of children aged ≥8 years who completed a three-pass 24-h dietary recall. Of these 498 children, six could not complete the dietary recall and were excluded from the analysis. Children <8 years of age do not reliably recall food( Reference Livingstone, Robson and Wallace 26 ) intakes and dietary data are not presented for this age group (n 168). SSB included sugar-sweetened soda, cordial, fruit drinks, flavoured mineral water, and sports and energy drinks( Reference Grimes, Riddell and Campbell 3 ).
The paediatric adjusted Goldberg cut-off method( Reference Sichert-Hellert, Kersting and Schoch 27 ) was used to identify participants with implausible EI. This method compares the participant’s ratio of reported EI:estimated BMR (estBMR) with the Goldberg cut-off 2 value. Age- and sex-specific Goldberg cut-off values were determined using the original adult Goldberg equation( Reference Goldberg, Black and Jebb 28 ), substituted with child-specific light physical activity levels (PAL)( Reference Torun, Davies and Livingstone 29 ) and revised estimates( Reference Black 30 ) for the CV in EI (CVwEI 23 %), repeated BMR measurements (CVwB 8·5 %) and variation in PAL (CVtp 15 %). On this basis, participants (n 34, 6·9 %) with an EI:estBMR ratio <0·87 for 8–12-year-old boys and 0·84 for 8–12-year-old girls were classified as a low-energy reporter and excluded from analyses with dietary data. It has been shown that under-reporting is more likely to occur with increased body fatness( Reference Magarey, Watson and Golley 31 ). Therefore, in this analysis, which includes adiposity as an outcome measure, we considered it important to identify and exclude low-energy reporters. An additional sensitivity analysis was performed, whereby no children were excluded on the basis of misreporting. In this analysis there was no appreciable change to the results when identified low-energy reporters were included (data not shown).
Logistically it was not possible to concurrently collect 24-h dietary recalls alongside the 24-h urine collection. Instead 24-h recalls were collected with children on the day of testing procedures completed at the school site (i.e. actual recalled food intake is for the day before testing procedures), whereas children could opt to commence the 24-h urine collection at school on the day of testing, or over a weekend day. Overall, 416 (91 %) children completed the 24-h dietary recall and 24-h urine collection within a week of one another. In some instances, because of the child being unwell on the day of testing or away for an extended holiday period the timing between the 24-h dietary recall and 24-h urine collection was greater than a week (i.e. n 29, 7 % collected within a 2-week period, and n 14, 3 % collected within a 6-week period).
Statistical analysis
Descriptive statistics (mean values and standard deviations or proportions and numbers) were used to describe participant characteristics. Because SSB (g/d) intake was skewed, medians and interquartile ranges were reported. Multiple linear regression was used to assess the association between urinary Na excretion (17 mmol/d, equivalent to 1 g/d of salt) and BMI z score. Logistic regression was used to assess the association between 24-h urinary Na excretion (17 mmol/d) and (i) weight category and (ii) abdominal obesity. The unadjusted and adjusted models (age, sex and SES) are presented. Where abdominal obesity was the outcome, additional adjustment for BMI z score was made. To assess whether the association between 24-h urinary Na excretion and adiposity outcome measures was independent of energy and SSB intake, additional models were constructed with these covariates. In this analysis EI was adjusted using the standard multivariable method, as opposed to the residual method, as there was no collinearity between 24-h urinary Na and EI (r 0·10, P=0·03). One outlier with an implausible waist circumference (i.e. 32 cm, which was 3·5 SD away from the mean waist circumference), was removed from the analyses related to WHtR. The normality of regression residual distributions was visually assessed and linearity checked using a component-plus-residual plot. To account for clustering of students within schools, clustered robust standard errors were used. Of note, the intra-class correlation (ICC) for the exposure variable (i.e. Na excretion) was 0·024 and the ICC for the primary outcome variable (i.e. BMI z score) was 0·030. Analyses were completed with STATA/SE 13.0 software (StataCorp LP), and a P value<0·05 was considered statistically significant.
Results
Table 1 shows the demographic characteristics of participants and information on urinary excretion and dietary intake. An overall 55 % were male, 62 % were of high socio-economic background, and the average age was 9·3 years. In total, 17 % of children were either overweight or obese and just under a quarter were classified as centrally obese. Na intake was 2378 mg/d and intakes were higher in older children.
Table 1 Descriptive characteristics of participants (Mean values and standard deviations; or numbers and percentages)

WHtR, waist:height ratio.
* One additional outlier for waist circumference removed from analyses.
Table 2 shows anthropometric measures and key characteristics by tertile of 24-h urinary Na excretion. BMI z score and waist circumference increased with increasing intakes of Na, as did the proportion of children who were overweight or obese. In both 4–7-year olds and 8–12-year olds the prevalence of abdominal obesity increased with higher intakes of Na. Concurrent with increases in Na were increases in 24-h urinary K excretion as well as the urinary Na:K ratio. Among 8–12-year-old children, EI and the amount of food and beverages consumed on the day of the 24-h diet recall increased across tertiles of Na intake.
Table 2 Anthropometry and key characteristics by tertile (T) of 24-h urinary sodium (mmol/d) excretion in Australian schoolchildren by age group (Mean values with their standard errors; numbers and percentages; or medians and interquartile ranges (IQR))

WHtR, waist:height ratio; SSB, sugar-sweetened beverages.
* For continuous variables P value for trend across tertiles, for categorical variables P value for χ 2 analysis.
† Dietary data apply only to those aged 8–12 years with a valid 24-h recall.
Association between 24-h urinary sodium excretion and measures of adiposity
Across the whole sample, there was a positive association between 24-h urinary Na excretion and BMI z score. An additional 17 mmol/d of Na (1 g salt/d) was associated with a 0·10 higher BMI z score, and Na intake accounted for 7 % of the variance in BMI z score. The association remained after adjustment for age, sex and SES (Table 3). When stratified by age group, the positive association remained in both age groups. In 8–12-year-old children the association remained after adjustment for energy and SSB intake.
Results from logistic regression analysis showed that a higher Na intake was associated with increased risk of being overweight or obese. Including all participants and with adjustment of confounders a 1 g/d higher salt intake was associated with a 23 % increase in the odds of being overweight or obese (Table 4). The association remained in age-stratified models and independent of energy and SSB intake in 8–12-year-old children.
Table 3 The association between 24-h urinary sodium excretion (17 mmol/d) and BMI z score in Australian schoolchildren by age groupFootnote * (Regression coefficients and 95 % confidence intervals)

* Clustered robust standard errors used to adjust for clustering by school.
† Model 1: unadjusted.
‡ Model 2: adjusted for age, sex and socio-economic status.
§ n 458 as only those children with valid dietary recall information are included.
|| Model 3: adjusted for model 2+energy intake (kJ/d).
¶ N/A: not available as dietary information was not collected in 4–7-year olds.
** Model 4: adjusted for model 2+sugar-sweetened beverages intake (g/d).
Including all participants, Na intake was positively associated with abdominal obesity (Table 5). After adjustment for age, sex and SES an additional 1 g/d of salt was associated with a 15 % increase in the risk of being centrally obese. In age-stratified models, the positive association remained. In all models, the association between salt intake and abdominal obesity was not independent of BMI z score.
Table 4 The association between 24-h urinary sodium excretion (17 mmol/d) and weight category (underweight/healthy weight v. overweight/obese) in Australian schoolchildren, by age group Footnote * Footnote † (Odds ratios and 95 % confidence intervals)

* Clustered robust standard errors used to adjust for clustering by school.
† Reference category=underweight/healthy weight.
‡ Model 1: unadjusted.
§ Model 2: adjusted for age, sex and socio-economic status.
|| n 458 as only those children with valid dietary recall information are included.
¶ Model 3: adjusted for model 2+energy intake (kJ/d).
** N/A: not available as dietary information was not collected in 4–7-year olds.
†† Model 4: adjusted for model 2+sugar-sweetened beverages intake (g/d).
Table 5 The association between 24-h urinary sodium excretion (17 mmol/d) and abdominal obesity in Australian schoolchildren, by age group* † (Odds ratios and 95 % confidence intervals)
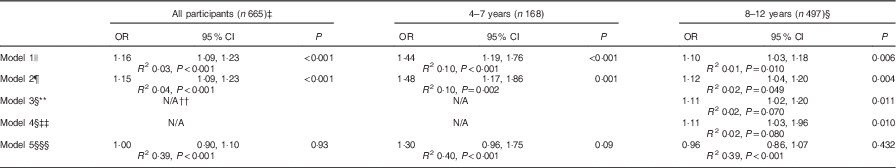
WHtR, waist:height ratio.
* Clustered robust standard errors used to adjust for clustering by school.
† Abdominal obesity defined as WHtR ≥0·5. Reference group=WHtR <0·5.
‡ One outlier for waist circumference removed from analysis.
§ Among 8–12 years n 457 as only those children with valid dietary recall information are included.
║ Model 1: unadjusted.
¶ Model 2: adjusted for age, sex and socio-economic status.
** Model 3: adjusted for model 2+energy intake (kJ/d).
†† N/A: not available as dietary information was not collected in 4–7-year olds.
‡‡ Model 4: adjusted for model 2+sugar-sweetened beverages intake (g/d).
§§ Model 5: adjusted for model 2+BMI z score.
Discussion
Among primary schoolchildren we found that 24-h urinary Na excretion was associated with a higher BMI z score, greater risk of overweight/obesity and abdominal obesity. Of note the association between Na and abdominal obesity was not independent of BMI z score, suggesting that the potential adipogenic effect of Na relates to total body weight and is not specific to central fat distribution. Overall associations were consistent across stratified age groups; however, the magnitude of the association was greatest in the 4–7-year-olds on all three adiposity outcome measures. Some caution should be applied when interpreting the findings in the 4–7-year-old children because of the small number of children classified as centrally obese (n 24) and overweight/obese (n 14).
The magnitude of the effect of 1 g/d of salt on BMI z score is small (i.e. 0·10), and the potential health significance of this is unclear. In obese children a reduction of at least 0·5 in BMI z score is needed to achieve reductions in waist circumference, body fat and cardiovascular risk( Reference Ford, Hunt and Cooper 32 ). However, given intensive obesity lifestyle interventions report changes of only −0·10 in BMI z score( Reference Ho, Garnett and Baur 33 ), our finding for an effect of Na alone is not unexpected. A change of this magnitude may still be important at the population level and in slowing down the trajectory of excess weight gain during childhood. The variance in BMI z score and weight status explained by Na intake alone was 7 and 5 %, respectively. Given the large number of determinants for childhood obesity, this level of variance explained by a single nutrient is notable. Comparatively sweetened beverages, which are often targeted in childhood obesity efforts, explained only 1 % of the variance in body weight status among US children( Reference Nicklas, Yang and Baranowski 34 ).
Overall our findings are consistent with previous studies conducted in children( Reference Libuda, Kersting and Alexy 6 – Reference Zhu, Pollock and Kotak 8 , Reference Ma, He and MacGregor 35 ) as well as adults( Reference Yoon and Oh 7 , Reference Ma, He and MacGregor 35 – Reference Yi and Kansagra 39 ). For example, in a large representative sample of Korean children aged 7–18 years participants in the highest quintile for Na density, as determined by 24-h dietary recall, were 1·78 (95 % CI 1·13, 2·80) and 2·13 times (95 % CI 1·16, 3·91) more likely to be obese or centrally obese, respectively( Reference Yoon and Oh 7 ). Unlike our study, the authors did not adjust for BMI z score in the Na and abdominal obesity model; hence, it is unclear whether this association was independent of overall body size. Similarly in US adolescents, after adjustment for important confounders including energy, Na intake assessed by repeated 24-h dietary recalls was positively associated with BMI, waist circumference and fat mass( Reference Zhu, Pollock and Kotak 8 ). In contrast, among Canadian schoolchildren no difference in Na intake, as assessed by a web-based 24 h recall, across weight categories was found( Reference Woodruff, Fryer and Campbell 40 ). In adults, a positive association between Na intake, from 24-h dietary recall methods, and BMI was reported in nationally representative samples in the USA( Reference Yi, Firestone and Beasley 38 ) and Korea( Reference Yoon and Oh 7 ).
Only one longitudinal paediatric study has assessed the association between 24-h urinary Na and obesity risk( Reference Larsen, Angquist and Sorensen 41 ). In this study of German children aged 3–18 years positive baseline associations between Na intake and BMI z score and %BF were reported; however, there was limited evidence for a temporal relationship. Specifically, in the unadjusted model, baseline Na was associated with an increase in %BF; however, in adjusted models this was only a trend (P=0·073). Conversely there was no association between baseline Na and change in BMI z score. Similarly, there was no association between change in Na and change in either BMI z score or %BF. In adults, in a Danish longitudinal cohort with 6 years of follow-up, higher 24-h urinary Na excretion was associated with increased body fat and decreased fat-free mass, but not with body weight or waist circumference( Reference Larsen, Angquist and Sorensen 41 ); however, the positive associations were observed only in adjusted models.
There are several mechanisms that may explain the relationship between Na intake and obesity risk. First, as Na intake is highly correlated to EI (r 0·7)( Reference Willet 42 ) it is possible that overall greater consumption of foods that are high in both energy and Na promotes weight gain. Likewise, foods that contain higher levels of salt may enhance the palatability of foods, which are also high in fat and energy. In addition, through the effects of Na on thirst, a salty diet may encourage greater consumption of high-energy SSB when these are readily available( Reference Grimes, Riddell and Campbell 3 – Reference He, Marrero and MacGregor 5 ). Given that increased EI is a plausible factor that may link Na to obesity risk, we adjusted for both energy and SSB intake in the subsample of children with dietary data. Consistent with others( Reference Libuda, Kersting and Alexy 6 – Reference Zhu, Pollock and Kotak 8 , Reference Yi, Firestone and Beasley 38 ), we found that the association between Na intake and BMI z score and weight category was independent of energy and SSB intake, suggesting other mechanisms may be at play. However, an important consideration within our study is that we found no association between EI and BMI z score or weight status (data not shown), and the association between 24-h urinary Na and EI was very weak (r 0·10, P=0·03). Therefore, it is not surprising that the additional adjustment for energy or SSB intake did not alter our results. It is possible that, because of limitations associated with 24-h dietary recall methods – for example, estimating portion size and memory bias( Reference Livingstone, Robson and Wallace 26 ) – the degree of error in our measure of EI is large. Comparatively, a urinary biomarker is not subject to these limitations. In adults important differences in relation to the assessment method of Na and the relationship to BMI have previously been reported( Reference Zhang, Temme and Sasaki 43 ). In this large study of adults who completed both a 24-h diet recall and 24 h urine collection, it was shown that 24-h dietary recall Na was inversely associated with BMI, yet conversely 24-h urinary Na was positively associated with BMI( Reference Zhang, Temme and Sasaki 43 ). This emphasises important differences between the two methodologies.
It may be that the objective measure of 24-h urinary Na acts as a surrogate marker for overall poorer diet quality( Reference Navia, Aparicio and Perea 36 ), which in turn is linked to obesity risk. In 6-year-old Danish children greater diet quality was associated with lower 24-h urinary Na excretion and higher 24-h urinary K excretion( Reference Kristbjornsdottir, Halldorsson and Thorsdottir 44 ). Our finding whereby the urinary Na:K ratio concurrently increased across tertiles of urinary Na lends some support to the notion that overall diet quality may be an important factor in explaining the Na and obesity relationship.
Alternatively it has been suggested that Na may influence the formation of adipocyte tissue via increased lipogenic activity( Reference Fonseca-Alaniz, Brito and Borges-Silva 45 , Reference Fonseca-Alaniz, Takada and Andreotti 46 ). In rats, those fed a high-Na diet, compared with those fed a normal-Na diet, had a greater increase in adipocyte mass despite the same amount of overall food being consumed. In addition, rats fed the high-Na diet displayed greater uptake of glucose and conversion into lipids within adipocyte tissue( Reference Fonseca-Alaniz, Brito and Borges-Silva 45 ). Whether higher salt intake in humans has a similar effect on glucose metabolism is unknown. In adults it has been shown that higher 24-h urinary Na excretion is associated with the metabolic syndrome( Reference Ge, Guo and Chen 47 , Reference Hoffmann and Cubeddu 48 ), including abdominal obesity; however, neither of these studies adjusted for EI.
Limitations of the present study include the cross-sectional design, convenience sample and the low response rate, which limits the generalisability of the findings to the wider population. Compared with the Australian population our sample contained fewer overweight and obese children( 49 ). We had no dietary data for children aged 4–7 years and were unable to determine whether the associations between Na and adiposity outcomes were independent of energy. Furthermore, the collection of dietary data and urine samples was not concurrent and there may have been a positive association between EI and BMI that we were unable to detect. The major strength of this study is the use of a urinary biomarker to assess Na intake, removing potential bias from mis-reporting, a behaviour frequently observed in dietary studies( Reference Livingstone, Robson and Wallace 26 ); further, this method captures Na from food sources as well as discretionary salt use.
In conclusion, our study is in agreement with previous findings indicating a cross-sectional positive association between Na intake and measures of adiposity in Australian schoolchildren. A greater understanding of the potential mechanistic action is required in conjunction with longitudinal studies.
Acknowledgements
The authors acknowledge the Victorian Department of Early Childhood and Development for their support in allowing the study to be conducted within the government school sector. The authors thank all schools and children who participated in the study.
This study was funded by a National Heart Foundation of Australia Grant-in-Aid (G 10M 5021) and a Helen MacPherson Smith Trust Fund Project Grant (6002). At the time of this work C. A. G. was supported by a National Heart Foundation of Australia Postgraduate Scholarship (PP 08M 4074); C. A. G. is currently supported by a National Heart Foundation of Australia Postdoctoral Fellowship (Award ID: 100155). None of the funders had any role in the design, analysis or writing of this article.
C. A. G., L. J. R., K. J. C. and F. J. H. designed the research. C. A. G. analysed the data and drafted the manuscript. L. J. R., K. J. C., F. J. H. and C. A. N. secured project funding and reviewed and revised the manuscript. All authors approved the final manuscript as submitted.
The authors declare that there are no conflicts of interest.








