According to the International Dairy Federation (IDF) recommendations subclinical mastitis in dairy cows is defined as a disease without clinical inflammatory signs of the udder and a SCC > 100 000 cells/ml with the presence or absence of pathogens. Subclinical mastitis is commonly detected by measuring SCC, but there is no standardized approach in terms of SCC cut-off values and evaluation on basis of individual composite SCC or QSCC. As a consequence, the reported prevalence of SCM varies widely between 13 and 53% (Fox et al., Reference Fox, Chester, Hallberg, Nickerson, Pankey and Weaver1995), since the chosen criteria vary with country (Hiitiö et al., Reference Hiitiö, Vakkamäki, Simojoki, Autio, Junnila, Pelkonen and Pyörälä2017), region (Hashemi et al., Reference Hashemi, Kafi and Safdarian2011), herd (Erskine et al., Reference Erskine, Wagner and DeGraves2003) and method of SCC count (Narayana et al., Reference Narayana, Miglior, Naqvi, Malchiodi, Martin and Barkema2018).
Subclinical mastitis causes substantial economic losses in dairy herds not least from reduced milk yield ranging from approx. € 1000 to 1300 per case annually (Seegers et al., Reference Seegers, Fourichon and Beaudeau2003; Halasa et al., Reference Halasa, Huijps, Østerås and Hogeveen2007; van den Borne et al., Reference van den Borne, Halasa, van Schaik, Hogeveen and Nielen2010). For every 100 000 somatic cells/ml increase exceeding 200 000 somatic cells/ml, in the bulk tank SSC the herd yield is estimated to decrease by 2.5% (Bradley and Green, Reference Bradley and Green2005). In addition, SCM leads to extra labour and treatment costs and represents a major reservoir of contagious pathogens, which are risk factors for uninfected cows in the herd (Down et al., Reference Down, Green and Hudson2013).
Therapeutic options for SCM are typically based on the use of antibiotics (McDougall et al., Reference McDougall, Parker, Heuer and Compton2009). These are used both during lactation as well as at the start of the drying-off period, often in combination with teat sealants (Golder et al., Reference Golder, Hodge and Lean2016). There are, however, limitations in cases of chronic infections, where antibiotic therapy is not considered to be cost-effective (Gruet et al., Reference Gruet, Maincent, Berthelot and Kaltsatos2001; Erskine et al., Reference Erskine, Wagner and DeGraves2003). Excessive use and misuse of antibiotics in agriculture, humans and animal husbandry accelerate the occurrence of antimicrobial resistance and have a negative impact on the environment worldwide (Sandgren et al., Reference Sandgren, Waller and Emanuelson2008). Pathogen induced SCM is a major cause for the worldwide consumption of antibiotics (Krömker and Leimbach, Reference Krömker and Leimbach2017). Due to the disadvantages of antibiotic treatment, recent research has explored the use of non-drug strategies, e.g. feeding of probiotics or plant extracts and immunoprophylaxis to improve udder health in SCM (Wu et al., Reference Wu, Liu, Yang, Wang and Zhu2015, Reference Wu, Zhu, Xu, Liu, Duan, Wang and Wang2018; Seroussi et al., Reference Seroussi, Blum, Krifucks, Lavon and Leitner2018; Catozzi et al., Reference Catozzi, Cuscó, Lecchi, De Carlo, Vecchio, Martucciello, D'Angelo, Francino, Sanchez Bonastre and Ceciliani2019).
Since the late 1990s, a number of studies have investigated the immunoregulatory role of the vagus nerve in different inflammatory disorders (Maier et al., Reference Maier, Goehler, Fleshner and Watkins1998; Bonaz et al., Reference Bonaz, Sinniger and Pellissier2017). The direct stimulation of the vagus nerve branches leads to a significant improvement of the clinical presentation in various severe inflammatory diseases (Meregnani et al., Reference Meregnani, Clarençon, Vivier, Peinnequin, Mouret, Sinniger, Picq, Job, Canini, Jacquier-Sarlin and Bonaz2011). In addition to a direct stimulation of the cervical branch of the vagus nerve (Koopman et al., Reference Koopman, Chavan, Miljko, Grazio, Sokolovic, Schuurman, Mehta, Levine, Faltys, Zitnik, Tracey and Tak2016) a non-invasive approach was established by stimulation of the auricular branch of the vagus nerve (ABVN) via electric current (Hong et al., Reference Hong, Zillekens, Schneiker, Pantelis, Jonge, Schaefer, Kalff and Wehner2018), acupuncture (He et al., Reference He, Wang, Shi, Shang, Li, Jing and Zhu2012) or the injection of substances (Litscher and Litscher, Reference Litscher and Litscher2016). These therapeutic methods are part of AT. The existence of associations between different regions or organs of the body and certain points or zones of the human ear was first published in the 1950s (Nogier, Reference Nogier1957). The development of AT for humans is described in detail (Wirz-Ridolfi, Reference Wirz-Ridolfi2019). Several studies used ABVN stimulation showing that AT influences the vagal tone to regulate cardiovascular, gastrointestinal and respiratory disorders in different species, e.g. rats (Gao et al., Reference Gao, Li, Liu, Rong, Ben, Li, Zhu and Zhang2011), mice (Hong et al., Reference Hong, Zillekens, Schneiker, Pantelis, Jonge, Schaefer, Kalff and Wehner2018), rabbits (Xu et al., Reference Xu, Liu, Liu, Chen and Dong1992), cats (Nomura and Mizuno, Reference Nomura and Mizuno1984) or humans (Bonaz et al., Reference Bonaz, Sinniger and Pellissier2017). To our knowledge, only a limited amount of research on AT has been performed in cattle, although a bovine ear chart containing 27 auricular acupuncture points (AAP) was published about 20 years ago (Fig. S1 in online Supplementary File) (Kothbauer, Reference Kothbauer1999). For cattle, Kothbauer (Reference Kothbauer1999) described relationships between certain diseases and active AAP. Provoking individual regions or organs triggers a reaction in the corresponding AAP, which can be directly observed by surface conductivity measurements (as reviewed in Kothbauer, Reference Kothbauer1986, Reference Kothbauer1999). Under clinical conditions or after manual irritation, the AAP shows signs of inflammation like marked reddening, swelling, occurrence of exudate or, under more severe clinical conditions, superficial skin lesions (Kothbauer, Reference Kothbauer1986). The projection zone for the udder is located below the plicae auriculares longitudinales mediales in the middle third of the ear (Ear tag position 10 in Fig. 1 and AAP of the udder in Fig. S1 in online Supplementary File) (Kothbauer, Reference Kothbauer1999). This location (position 10) corresponds largely with the position of the ear tags of cattle (Fig. 1).
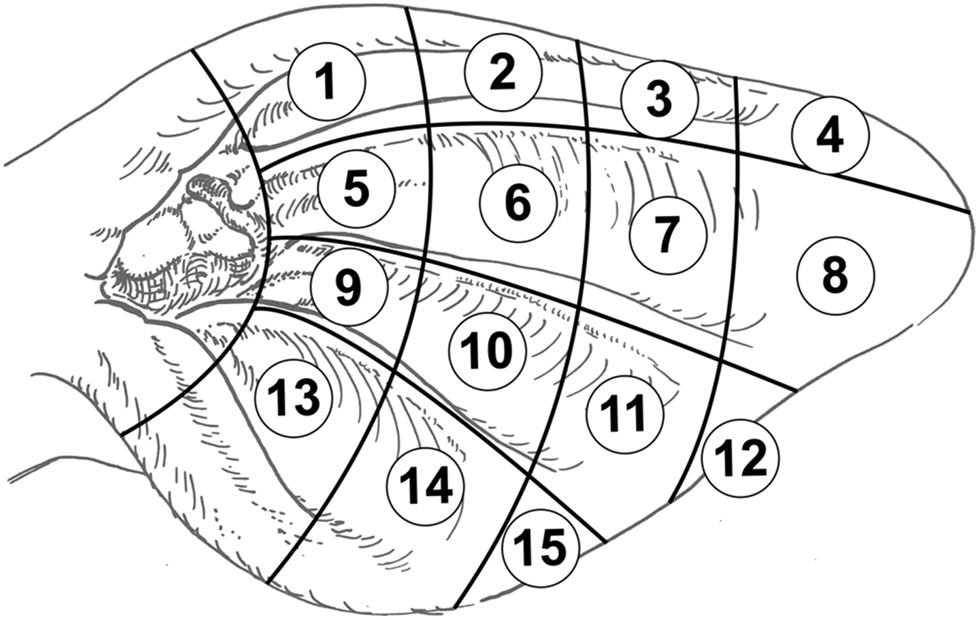
Fig. 1. Scheme to track the position of ear tags in dairy cows. Numbers 1–15 (in circles) represent positions. Position 10 usually represents the location of ear tags in cows. Figure based on the picture of cattle´s ear according to Kothbauer (Veterinary Acupuncture – Basic Principles and Their Clinical Applications with Ear Acupuncture on Cattle and Some References to the Horse. Zweimühlen-Verlag, Munich, Germany, 1999).
To our knowledge, no systematic research is published relating ABVN stimulation to improve udder health or addressing any other organs in dairy cows. Therefore, the purpose of our study was to assess the potential effect of ABVN stimulation with local anaesthetics at the positions of the ear tags of dairy cows suffering from SCM. It was hypothesized that stimulation of the auricular branch of the vagus nerve by neural therapy has an anti-inflammatory effect on the subclinical inflamed udder and results in decreased SCC.
Material and methods
The study procedures were discussed and approved by the Ethics Committee of the University of Veterinary Medicine, Vienna, in accordance with Good Scientific Practice guidelines and the national authority according to §8 of Law for Animal Experiments, Tierversuchsgesetz-TVG (BMWF-68.205/0033-WF/V/3b/2017) of Austria.
Animals and study design
Nine conventional Simmental dairy farms in Lower Austria, which obtained monthly milk recording data from the regional Dairy Herd Improvement Service (Landeskontrollverband), participated in this study. The farm inclusion criteria were that at least 80% of cows showed SCC < 150 000 cells/ml and that a maximum of 15% had SCC > 200 000 cells/ml milk on average over the year. Only cows with an individual composite SCC of > 200 000 cells/ml milk in the latest milk record data were enrolled to increase the probability for finding cows with QSCC > 100 000 cells/ml in at least one quarter. All selected cows were clinically healthy and received no treatment with antibiotics and/or nonsteroidal anti-inflammatory drugs in the current lactation. In our study, cows with QSCC > 100 000 cells/ml independent of the bacteriological result were defined as having SCM, which is in accordance with the definition of the IDF. The Excel randomization function determined the allocation of cows to control group (CON) without any treatment, and treatment group (TRE) with application of local anaesthetics. Documentation of data was performed using Microsoft Excel 2010 (Microsoft Office Cooperation, Redmond, WA, USA). In addition, data relating days in milk (DIM), parity, farms, season, position of ear tag, affected udder side and bacteriological analysis were documented.
Collection of quarter milk samples for determination of QSCC and bacteriological examination
Quarter milk samples were collected on days 0 (before treatment), 2, 4, and 6 for measurement of QSCC. Furthermore, aseptic quarter milk samples were taken on d0 and d6 for bacteriological examination. After sampling, all milk samples were immediately stored at 4°C and delivered to an analytical laboratory (Qualitätslabor Niederöstereich, Gmünd, Austria) on the same day. An automated somatic-cell counter (Fossomatic™ FC, Foss, DK-3400 Hilleroed, Denmark) analysed the QSCC. Collection and microbiological procedures were defined and performed as outlined by the National Mastitis Council (Hogan and National Mastitis Council (U.S.), Reference Hogan1999).
Auriculotherapy with local anaesthetics
Neural therapy on the ABVN of the scar of the left ear was performed in TRE on d0, d2 and d4. For this purpose, each cow was restrained in the feeding fence and the head was tied to the right side of the animal using a halter. The whole ear, especially the area around the ear tag, was examined for pathological changes such as marked reddening, swelling, exudate and hyperalgesia by palpation. After clipping all long hair of the pinna, the area was cleaned and disinfected using 70% ethyl alcohol-soaked cotton balls (Ethanol 70%, Liquid Production GmbH, Flintsbach am Inn, Austria). Cows of TRE group were treated by intracutaneous infiltration of 0.1 ml procaine hydrochloride 2% (Procamidor® 20 mg/ml, Richterpharma AG, Wels, Austria) at each location clockwise at 12, 3, 6 and 9 around the ear tag (Fig. 2) using 1 ml syringes (Omnifix®-F, B. Braun Melsungen AG, Melsungen, Germany) and 0.6 × 30 mm needles (Henry Schein Inc. Melville, USA). Cows from CON group were handled identically, however, without injection of local anaesthetics.
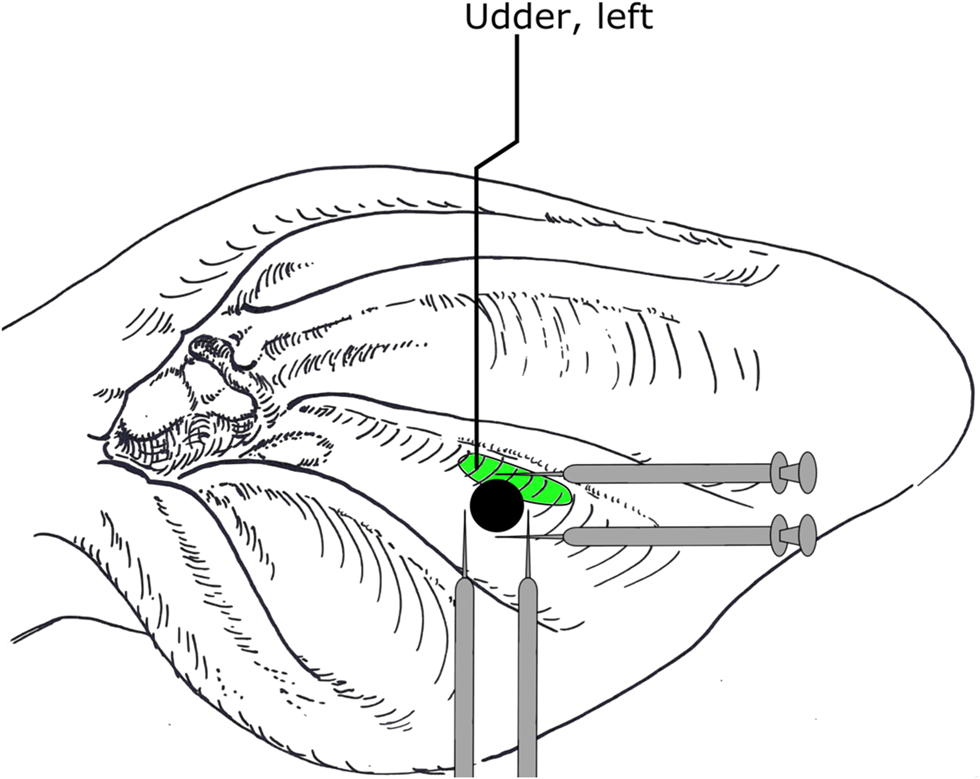
Fig. 2. Schematic illustration of intracutaneous application of local anaesthetics at each location clockwise at 12, 3, 6 and 9 around an ear tag. Black circle represents the ear tag corresponding with position number 10. Oval area represents the ear acupuncture point for the udder. Figure based on the picture of cattle´s ear according to Kothbauer (Veterinary Acupuncture – Basic Principles and Their Clinical Applications with Ear Acupuncture on Cattle and Some References to the Horse. Zweimühlen-Verlag, Munich, Germany, 1999).
Statistical analysis
All data were analysed with the statistical program SPSS (SPSS version 20; IBM Corporation, NY, USA). Because QSCC values were not normally distributed, data were log10 transformed and categorized for subsequent analysis. A general linear model with repeated measurements (one-way analysis of variance: ANOVA) was used to compare the change of QSCC over the sampling days (d0, d2, d4 and d6). The group variable CON and TRE was included in the model to exclude a potential confounding interaction. The change of QSCC over time within CON and TRE was compared using ANOVA and adjusted by Bonferroni post hoc test. The sampling days were defined as within-subject variable and d0 was taken as simple contrast to compare with. Independent variables (DIM, parity, farm, season, position of the ear tag, udder side, and bacteriological result) were included in the analysis of variance (ANCOVA). Moreover, effects on QSCC change in bacteriological positive samples on d0, AAP corresponding to position of ear tag (yes/no), side of udder quarter (left/right) were evaluated in CON and TRE. Bacteriological cure was defined as absence, on d6, of the bacteria identified on d0. Differences in bacteriological cure rate between CON and TRE were analysed by χ2-test. In general, values were considered significant when P ≤ 0.05.
Results
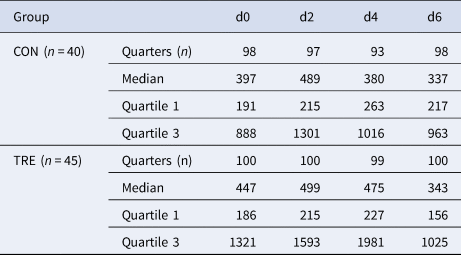
Based on the monthly milk record data over all four seasons of the nine participating dairy farms, 99 cows were initially enrolled into the study. Finally, 85 dairy cows met the inclusion criteria, whereas 14 were excluded from further analyses because of clinical mastitis (n = 7) or QSCC < 100 000 cells/ml at all quarters on d0 (n = 7). The milk samplings on days d0, d2, d4 and d6 resulted in a total of 784 QSCC, corresponding to 385 QSCC of CON (40 cows) and 399 QSCC of TRE (45 cows). Cows were grouped according to parity into primiparous (n = 21), two lactation (n = 19) and more than two lactation cows (n = 45). Moreover, the stage of lactation was then stratified into thirds, resulting in categories, SCC ≤ 100 (n = 16), 100–199 (n = 24) and ≥200 (n = 45) DIM. Since sampling in summer was performed at one farm only, the data of the season summer was restricted to those of the one specific farm. The position of the ear tag was categorized according to Fig. 1. None of the cows in the study showed signs of inflammation at the scar around the ear tags. Bacteriological examinations of milk samples were performed on days 0 and 6. Median QSCC on d0 was higher in TRE (447 000) than in CON (397 000). The changes of median QSCC over time demonstrated an increase from d0 to d2 and a decrease from then on in both groups (Table 1). Descriptive statistics of QSCC of previously mentioned categories (variables) in groups CON and TRE are shown in online Supplementary File Table S1.
Table 1. Median, first and third quartile of quarter somatic cell count (in 1.000 cells/ml milk) of control (CON) and treatment group (TRE) on days (d) 0, 2, 4 and 6
Effect of AT over time on the change of QSCC in TRE compared with CON
The initial analysis included the groups (CON, TRE) as potential confounding variables into the model. The results indicated an interaction between the groups and time revealing significant differences in QSCC over the time (P < 0.05). The analysis of repeated measurements within groups showed a significant change of QSCC over the time (d0 to d6; P = 0.001) in TRE, whereas within CON (P > 0.05) no differences were found (Fig. 3). The pairwise comparison between the sampling days resulted in a significant reduction of QSCC after the third treatment from d0 to d6 (P < 0.01) (Table S2 in online Supplementary File). Including independent variables (DIM, parity, bacteriological result, farm, season, position of the ear tag, udder side) into the analysis (ANCOVA) affected the results, indicating different effects of farms and season on QSCC in CON and of ear tag position on QSCC in TRE (Table S3 in online Supplementary File). Including the covariates into the pairwise comparison, however, resulted in a significant reduction of QSCC from d0 to d6 only in TRE (P < 0.01).
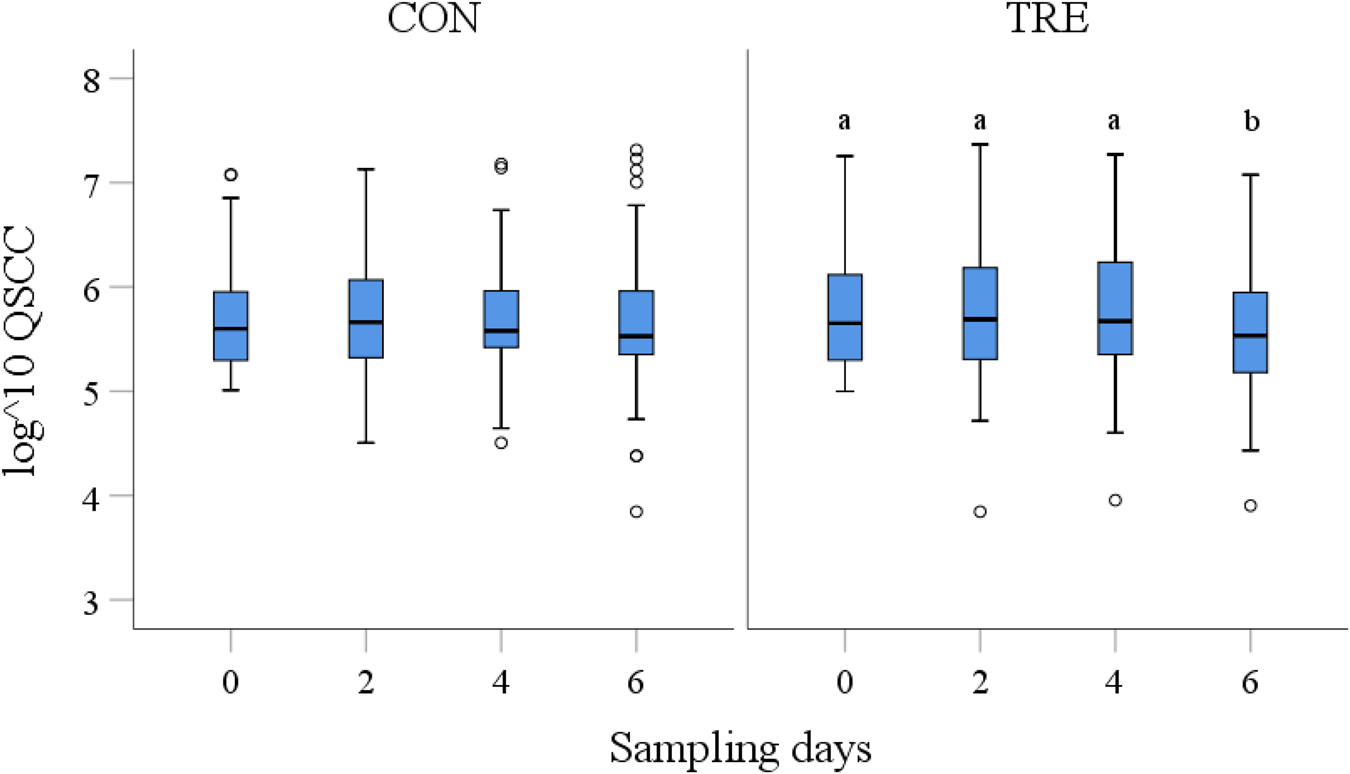
Fig. 3. Log transformed somatic cell counts (QSCC) of control (CON) and treatment (TRE) group on sampling days 0, 2, 4 and 6 (x-axis). General linear regression model with Bonferroni-adjustment indicates differences between QSCC over the time in TRE. Different superscripts indicate significant differences (P < 0.05). Outliers are diagrammed as circles (o).On line 158 Fig.1 corrected to Fig. 3
Effect of AT over time on the change of QSCC in bacteriological positive samples of TRE compared with CON
The bacteriological analysis of quarter milk samples on d0 and d6 revealed various bacteria, which were grouped into environmental (i.e. Enterobacteriaceae, Streptococcus dysgalactiae, Enterococcus, Acinetobacter), major contagious (i.e. Streptococcus uberis, Streptococcus agalactiae), and minor contagious species (i.e. Non-aureus Staphylococcus spp., Corynebacterium spp.). Major pathogens with higher importance, e.g. Escherichia coli, Staphylococcus aureus, and fungi (Aspergillus fumigatus) were listed separately. Bacteriological samples with more than one isolate were defined as multiple isolates. The proportion of the bacteriological findings in CON and TRE samples are presented in Table S4 in online Supplementary File. In total, 52.0% (51 of 98) samples in CON and 59.0% (59 of 100) of samples in TRE were bacteriological positive at d0. Minor contagious isolates were predominantly found in both groups. The bacteriological cure rate, defined as the absence of bacteria on d6 compared with d0, was not different between CON (27.5%, 14 of 51) and TRE (20.3%, 12 of 59) (P = 0.38). Using the group variable (CON, TRE) in the statistical model, an interaction between the groups and time was evident (P < 0.05). The multiple comparison in bacteriological positive milk samples on d0 or d6 or both over time showed a significant reduction of QSCC in TRE (P = 0.01), but not in CON. Pairwise comparisons of QSCC in TRE resulted in reduction of QSCC after the third treatment from d0 to d6 (P 0.05) (Table S5 in online Supplementary File). In the bacteriological negative samples (negative on d0 and d6) the reduction in QSCC was also significant in TRE (P < 0.05) but not in CON (P > 0.05). The pairwise comparison, however, indicated a significant difference between d2 and d6 (P = 0.01) (Table S6 in online Supplementary File).
Effect of ear tag position on the results of AT application
The position of the ear tags were documented, indicating ‘correct’ position if the ear tag corresponded to the AAP of the udder (position 10). In consequence, 28 ear tags of CON and 32 of TRE were at the position 10 (approximately 70% for both), whereas both in CON and TRE eleven were found at other positions. Ear tags at the position 9, located at the area of the meatus acusticus, were found eight in CON and three in TRE. Other ear tags were located at position 6 (one in CON and five in TRE), position 7 (one in TRE), position 13 (one in TRE) and position 14 (two in CON and one in TRE).
There was a significant reduction of QSCC when treatment was performed at the ear tag position 10 (P < 0.01). Including the group variable (CON, TRE), an interaction with time was identified (P < 0.05). The pairwise comparison of the sampling days revealed that the reduction of QSCC occurred from d2 to d6 (P < 0.01) (Table S7 in online Supplementary File). An increased QSCC on d2 after the first treatment was apparent, however, this pattern was also present in CON (Table S1 in online Supplementary File). Moreover, there was an effect on QSCC when treatment was performed at the ear tag position 9 (TRE: P = 0.001) demonstrating reduction of QSCC after the third treatment from d0 to d6 (Table S8 in online Supplementary File). Including the group variable in the analyses, however, showed no interaction between the groups and time of QSCC change (P > 0.05). Because of the low number of ear tags positions other than 9 and 10 no analyses on position-level were performed.
Effect of the udder (quarter) side on the results of AT application
The bovine ear chart indicates that the AAP for the left udder is on the left ear and the AAP for the right udder is on the right ear (Fig. S1 in online Supplementary File). In CON, 184 samples were taken from the left udder side and 201 from the right udder side. In TRE, 191 samples were taken from the left udder side and 208 were taken from the right udder side. A significant reduction of QSCC was only found in TRE, but independent of the udder side (left: P = 0.001, right: P < 0.05).
Discussion
The aim of this study was to evaluate the effect of AT in dairy cows with SCM. Both CON and TRE contained a comparable number of quarters, so that the stochastic basis for statistical evaluations was given. Our findings support the hypothesis that there is a positive effect of ABVN stimulation on the QSCC after the third treatment. Including several variables into the model indicated significant interactions of a few variables. The systematic effect, however, was not confounded because results revealed a group effect on QSCC reduction indicating significant differences between treated and control cows. The significant variable in the data set of TRE was the position of the ear tags. Analysing the ear tag position as dependent variable identified the ear tag position at the AAP as the significant factor on the reduction of QSCC. Among the controls, however, we assume a confounding effect of the different farms and seasons on QSCC as data from the summer season was generated on one farm only. Furthermore, we observed that under certain conditions, e.g. in bacteriologically negative samples or AT at the AAP (position 10), an increase of QSCC after the first treatment was obvious, but three consecutive treatments resulted in a significant reduction of QSCC. In the field, the application of AT in cows suffering from SCM was easy to achieve. Nevertheless, intracutaneous infiltration of LA into the ear can cause the animals some pain and since repeated applications are required for a successful therapy, defensive reactions need to be taken into consideration. Therefore, a tight fixation of the cow's head to the feeding fence is crucial for the safety of both cow and veterinarian.
The underlying principle by which stimulation of the ABVN in cows leads to a decrease of SCC is uncertain. A neural response to inflammation, however, described by a cholinergic anti-inflammatory pathway via the vagal nerve was already reported (Tracey, Reference Tracey2002). Furthermore, the stimulation of the vagal nerve led to significantly decreased amounts of circulating TNF in the serum and, therefore, suppressed inflammation (Borovikova et al., Reference Borovikova, Ivanova, Zhang, Yang, Botchkina, Watkins, Wang, Abumrad, Eaton and Tracey2000). This mechanism might be the underlying principle of the vagal acupuncture, reducing the QSCC in the udder of dairy cows suffering from SCM.
While AT at the ear tag position had no effect on the bacterial cure rate of infected quarters, this effect even in animals with bacteriological positive samples might again be explained by a general support of the immune system. This aspect, however, was not the aim of our study. Even in antibiotic therapy of mastitis, the bacterial cure rate is influenced by many different factors, such as animal- or farm-specific factors, application, dosage and duration of therapy. Furthermore, antibiotics often fail to eliminate bacteria completely (Krömker and Leimbach, Reference Krömker and Leimbach2017). Further research would be desirable to investigate the potential anti-inflammatory effect of AT on longer-term bacteriological cure in cows suffering from mastitis.
The study was designed to perform AT on the scar around the ear tag, assuming that most ear tags are located in the region of the AAP for the udder below the plicae auriculares longitudinales mediales in the centre of the cattle´s ear. Results showed a significant reduction of QSCC in TRE at the AAP (position 10). In humans, the vagus nerve mainly supplies the area around the auditory meatus via the ABVN (He et al., Reference He, Wang, Shi, Shang, Li, Jing and Zhu2012). This area corresponded to a large extent to the ear tag position 9 in the present study. The result that QSCC reduction was also observable in cows treated at ear tag position 10 could be explained because anatomical structures appear to be too different from humans to transfer AAP directly to bovine (Artmeier and König, Reference Artmeier and König1978; Kothbauer, Reference Kothbauer1986). To our knowledge, the exact course of the nerves in ears of cattle has not been studied yet. Overall, it is not easy to locate the acupuncture points accurately (He et al., Reference He, Wang, Shi, Shang, Li, Jing and Zhu2012), for example, the effect of AT on smoking cessation in humans was independent of the chosen acupuncture point (White and Moody, Reference White and Moody2006).
The ear chart by Kothbauer shows many AAP for several organs and regions of the body. It describes an AAP for the ‘udder, left’ in the left ear and an APP for the ‘udder, right’ in the right ear (Kothbauer, Reference Kothbauer1999). Performing AT on the left ear exclusively, allowed us to compare the effect on the left-side quarters (ipsilateral) as well as on the right-side quarters (contralateral). Our results showed a reduction of QSCC in both the left and the right quarters, which does not support the theory of a side related APP of the udder in bovine by Kothbauer (Reference Kothbauer1999), but support the pathway theory of ABVN stimulation to the site of inflammation (Bonaz et al., Reference Bonaz, Sinniger and Pellissier2017). The main points in AT to the contrary were the contralateral side, however, there were no significant differences in the effect of a single side or both sides therapy (reviewed by Bao et al., Reference Bao, Wang, Jia, Yang, Kong, Sun, Li, Xu, Zhang, Zhang, Du, Li, Yang, Feng, Shi, Jia and Wang2017).
In conclusion, applying neural therapy to the scars at the ear tags of lactating cows suffering from subclinical mastitis resulted in a significant reduction of the somatic cell counts in both udder sides, even though the treatment was performed on the left ear exclusively. The effect of neural therapy was found at the position of the cow's ear tag, indicating that the area of vagal innervation at the cow's ears is different compared with humans. Auricular therapy was successful both in bacteriological negative and positive samples. However, the bacterial cure rate was not affected.
Supplementary material
The supplementary material for this article can be found at https://doi.org/10.1017/S002202992100087X
Acknowledgment
This work was partially supported by ‘Österreichische Gesellschaft der Tierärztinnen & Tierärzte’ (ÖGT), Sektion Ganzheitsmedizin. Technical assistance was given by ‘Qualitätslabor Niederöstereich’ in A-3950 Gmünd, Austria, whose employees evaluated the milk samples. Technical assistance was also given by the farmers, who voluntarily provided their cows for this study and assisted in the practical part. We like to thank Professor D. Logue (Glasgow University) for his help preparing the manuscript.






