Akathisia is a distressing movement disorder associated with neuroleptic medication. Patients complain of a feeling of inner restlessness (Reference BarnesBarnes, 1989). The characteristic signs include constant moving when seated, rocking from foot to foot and pacing (Reference Adler, Angrist and ReiterAdler et al, 1989). Neuroleptic-induced akathisia (NIA) is usually an acute reaction, but a chronic akathisia can occur after prolonged treatment with neuroleptics (Reference Fleischhacker, Roth and KaneFleischhacker et al, 1990). It has been suggested that chronic NIA is related to tardive dyskinesia, and that like tardive dyskinesia it is difficult to treat (Reference BlaisdellBlaisdell, 1994). Reducing the neuroleptic dose may ease the symptoms (Reference BarnesBarnes, 1989). Other options include using an atypical neuroleptic, which may cause lower rates of akathisia (Reference Poyurovsky, Shardorodsky and FuchsPoyurovsky et al, 1999). Biochemical and pharmacological evidence has indicated the importance of serotonin in controlling movement (Reference Bersani, Grispini and MariniBersani et al, 1986). Serotonin antagonists, particularly mianserin, ritanserin and cyproheptadine, have been used to treat NIA. This article reviews the published trials and case reports retrieved by a Medline search from January 1985 to August 1999.
Published studies
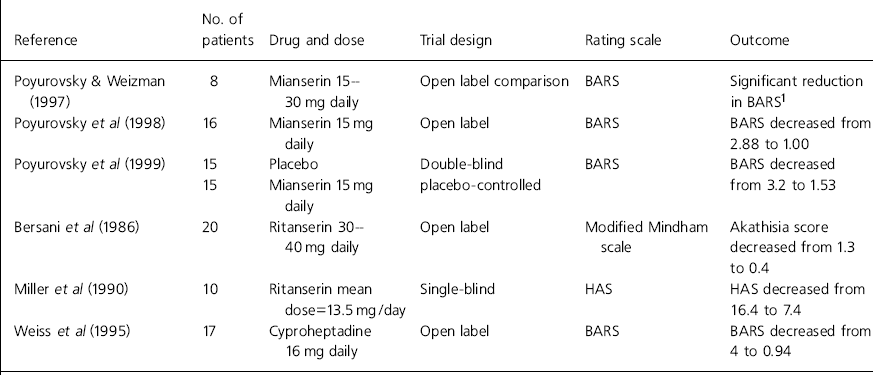
There are limitations with all the current studies. All used low patient numbers, were of short duration and usually open. The different studies lack uniformity, making it difficult to compare results (see Table 1 for an overview of results).
Table 1. Studies involving serotonin antagonists to treat akathisia
| Reference | No. of patients | Drug and dose | Trial design | Rating scale | Outcome |
|---|---|---|---|---|---|
| Poyurovsky & Weizman (Reference Poyurovsky and Weizman1997) | 8 | Mianserin 15–30 mg daily | Open label comparison | BARS | Significant reduction in BARS1 |
| Poyurovsky et al (Reference Poyurovsky, Fuchs and Weizman1998) | 16 | Mianserin 15 mg daily | Open label | BARS | BARS decreased from 2.88 to 1.00 |
| Poyurovsky et al (Reference Poyurovsky, Shardorodsky and Fuchs1999) | 15 | Placebo | Double-blind placebo-controlled | BARS | BARS decreased from 3.2 to 1.53 |
| 15 | Mianserin 15 mg daily | ||||
| Bersani et al (Reference Bersani, Grispini and Marini1986) | 20 | Ritanserin 30–40 mg daily | Open label | Modified Mindham scale | Akathisia score decreased from 1.3 to 0.4 |
| Miller et al (Reference Miller, Fleischhacker and Ehrman1990) | 10 | Ritanserin mean dose=13.5 mg/day | Single-blind | HAS | HAS decreased from 16.4 to 7.4 |
| Weiss et al (Reference Weiss, Aizenberg and Hermesh1995) | 17 | Cyproheptadine 16 mg daily | Open label | BARS | BARS decreased from 4 to 0.94 |
Three studies were identified but used mianserin, a tetracyclic antidepressant with 5-HT2A/2C antagonist activity, to treat akathisia. Two open studies investigated the effect of low-dose mianserin (Reference Poyurovsky and WeizmanPoyurovsky & Weizman, 1997; Reference Poyurovsky, Fuchs and WeizmanPoyurovsky et al, 1998). Both studies included patients who had a score of at least 2 on the Barnes Akathisia Rating Scale (BARS) (Reference BarnesBarnes, 1989) and met DSM—IV criteria for acute akathisia (Reference AdlerAmerican Psychiatric Association, 1994). Medication was not altered during either study, and the neuroleptic dose remained unchanged for at least 7 days before the trials.
Poyurovsky & Weizman (Reference Poyurovsky and Weizman1997) used buspirone and mianserin to treat NIA. Initially, 10 patients received 30 mg daily of buspirone for 4 days. If there was no response, after a 1-day washout, patients received 15-30 mg daily of mianserin for 4 days.
Buspirone had no significant effect. By day 4 NIA symptoms had improved in two patients, but had worsened in two patients. (An improvement was a decrease in the BARS global score of at least 1; a worsening an increase of at least 1.) In seven of the eight patients who failed to respond to buspirone and received mianserin, the akathisia improved. By day 4 NIA symptoms completely disappeared in five patients taking mianserin.
The significance of the reduction in the BARS scores was evaluated using statistical analysis of covariance with repeated measurements (ANCOVA-RM). The baseline value was the covariate variable. After 4 days of mianserin there was a significant reduction in the global BARS score (ANCOVA-RM: F=12.24, d.f.=7; P<0.001). The only side-effect noted during mianserin therapy was 4 reports of mild sedation. The authors suggested that low-dose mianserin was effective for NIA, although this required confirmation in double-blind placebo-controlled studies.
A 2-week open study showed that 15 mg daily of mianserin treated akathisia in 14 out of 16 patients (Reference Poyurovsky, Fuchs and WeizmanPoyurovsky et al, 1998). Patients were rated at baseline; after 3, 7 and 14 days of mianserin therapy; and 2 days after mianserin had been stopped. The mean global BARS score at baseline was 2.88 (s.d.=0.86), and by day 14 it had decreased to 1.00 (s.d.=1.17). After mianserin had been withdrawn the BARS score increased to 1.67 (s.d.=1.01). Transition probability analysis showed that this decrease was significant (cumulative v. baseline: P<0.01). Psychotic symptoms were not induced, and mianserin was well tolerated.
The paper concluded that mianserin possessed anti-akathitic activity. The authors noted that in 14 patients, the akathisia had been resistant to traditional therapies.
The third trial conducted was a placebo-controlled double-blind study (Reference Poyurovsky, Shardorodsky and FuchsPoyurovsky et al, 1999). Thirty people with schizophrenia, who had received neuroleptics and met DSM—IV criteria for acute NIA (Reference AdlerAmerican Psychiatric Association, 1994), were given either placebo or 15 mg daily of mianserin. Akathisia was rated on the BARS at baseline and on days 3 and 5. The dosage of concomitant medication remained unchanged during the study and for at least 4 days before the baseline reading. Four patients on placebo dropped out of the trial. At day 5 the BARS global sub-scale score had dropped 1.67 (s.d.=0.82) points (52.2%) in the mianserin group, whereas the BARS had dropped 0.45 points (9.9%) (s.d.=0.82) in the placebo group. This difference was significant (P=0.006). Using a drop in the BARS score of at least 2 points as a response, six patients on mianserin responded (40%), compared with one patient on placebo (9.1%) (P=0.093 by Fisher's exact test (not significant); log odds ratio=1.897).
The low-dose mianserin was well tolerated, and psychotic symptoms were not aggravated. The main side-effect noted was mild and transient sedation, which occurred in eight patients on mianserin and three on placebo. Although the short duration and low numbers of patients involved limited the usefulness of the trial, the authors concluded that mianserin is an effective antikathitic agent, and that 5-HT2 antagonism is responsible for its activity.
Two studies used ritanserin to treat akathisia (Reference Bersani, Grispini and MariniBersani et al, 1986; Reference Miller, Fleischhacker and EhrmanMiller et al, 1990). One open trial involving 20 patients with schizophrenia, according to DSM—III criteria (American Psychiatric Association, 1980), treated extrapyramidal side-effects (EPSE) with ritanserin (Reference Bersani, Grispini and MariniBersani et al, 1986). All had received neuroleptics for periods ranging from 6 months to several years. Neuroleptic therapy was not modified during the study. The use of antimuscarinic medication was avoided, and patients already receiving it had a 15-day washout period. EPSE were evaluated at baseline, and then weekly by the Mindham scale (Reference Mindham1976), modified with the addition of a tenth scale to rate akathisia.
Every patient received 30-40 mg daily of ritanserin for 4 weeks. There was a progressive reduction in all EPSE, including akathisia. The baseline score for akathisia was 1.3 and by week 4 it had decreased to 0.4. This change was significant (P<0.0004).
In a single-blind trial, 10 patients received a mean ritanserin dose of 13.5 mg (Reference Miller, Fleischhacker and EhrmanMiller et al, 1990). Eight patients had schizophrenia, and two were bipolar in the manic phase according to DSM—III—R criteria (Reference AdlerAmerican Psychiatric Association, 1987). Akathisia was assessed at baseline and after 3 days, using the Hillside Akathisia Scale (HAS) (Reference Fleischhacker, Bergmann and PerovichFleischhacker et al, 1989). A placebo-controlled discontinuation of ritanserin was undertaken in five patients. All medication including neuroleptics remained unchanged for at least 1 week before the study and while it was in progress. It is unclear for how long patients had been receiving neuroleptics prior to the study.
The mean HAS baseline score was 16.4 (s.d.=6) and after 3 days of ritanserin it had decreased to 7.4 (s.d.=5.2). This difference was statistically significant (P=0.0069). Akathisia returned after the placebo-controlled discontinuation; the HAS score increased from 7.4 to 14.6. The paper concluded that ritanserin effectively treated akathisia, although this required confirmation.
One open trial used 16 mg daily of the potent 5-HT2 antagonist cyproheptadine to treat akathisia (Reference Weiss, Aizenberg and HermeshWeiss et al, 1995). The 4-day trial involved 17 patients; 15 had schizophrenia, according to DSM—III—R criteria (Reference AdlerAmerican Psychiatric Association, 1987), one had a schizoaffective disorder and one had psychotic depression. The akathisia was acute rather than chronic, as every patient had been receiving neuroleptics for only 3-7 weeks.
Patients receiving beta-blockers or benzodiazepines were excluded from the trial. Concomitant medication was not altered for at least 3 days prior to baseline recordings and during the trial. Akathisia was assessed using the BARS, which was measured at baseline, days 2 and 4, and 4 days after discontinuation of cyproheptadine.
By day 4 there was a marked improvement in the akathitic symptoms in all 17 patients. The initial mean global BARS score was 4.00 (s.d.=0.79), by day 4 this had reduced to 0.94 (s.d.=0.97). The mean reduction in the BARS score was at least 75%, with 15 patients demonstrating at least a 50% reduction. After cyproheptadine was discontinued the symptoms of akathisia returned, and the BARS score increased to 2.65 (s.d.=1.46).
The data were analysed using ANCOVA-RM, to assess any effect of possible changes in depressive or psychotic symptoms. The Brief Psychiatric Rating Scale (BPRS; Reference Overall and GorhamOverall & Gorham, 1962) and the 23-item version of the Hamilton Rating Scale for Depression (HRSD; Reference HamiltonHamilton, 1960) were the covariate variables. From baseline to day 4 the ANCOVA-RMs for the BARS global and sub-scale scores dropped significantly (F>32.12, d.f.=1.15, P<0.0001).
Cyproheptadine did not induce psychotic symptoms and was well tolerated. The main side-effects were sedation, dry mouth and blurred vision. The authors concluded that cyproheptadine possessed anti-akathitic activity, but that trials against placebo and a non-specific sedative agent were required to substantiate this action.
Current therapies
There are problems with the main treatments for NIA: beta-blockers, antimuscarinic agents and benzodiazepines. Of these, only antimuscarinic agents are licensed to treat akathisia, beta-blockers and benzodiazepines, like the serotonin antagonists, are not licensed to treat it (Association of British Pharmaceutical Industry, 1999). One review concluded that the effectiveness of antimuscarinic agents is not established (Reference Adler, Angrist and ReiterAdler et al, 1989). It has been suggested that antimuscarinics are effective only when the akathisia is accompanied with parkinsonism (Reference Fleischhacker, Roth and KaneFleischhacker et al, 1990).
Beta-blockers, particularly lipophilic agents such as propranolol, have been suggested as the most effective anti-akathitic agent (Reference Fleischhacker, Roth and KaneFleischhacker et al, 1990). One review concluded that propranolol usually causes a mean improvement in subjective and objective NIA of 50% (Reference Adler, Angrist and ReiterAdler et al, 1989). Some of these studies were placebo-controlled, whereas others were single-blind crossover studies. The contraindications and side-effect profile, particularly bronchospasm and vasospasm, may limit the usefulness of beta-blockers (Reference Dumon, Catteau and LanvinDumon et al, 1992). Inter-estingly, it has been suggested that beta-blockers may partly treat akathisia via an antiserotonin effect (Reference Power and CowenPower & Cowen, 1992). Furthermore, the beta-blocker pindolol, which is a presynaptic 5-HT1A antagonist, has been shown to treat NIA (Reference Adler, Reiter and AngristAdler et al, 1987).
Benzodiazepines, especially clonazepam, have also been used to treat NIA (Reference Adler, Angrist and ReiterAdler et al, 1989). Pujalte et al (Reference Pujalte, Bottai and Hue1994) conducted a double-blind placebo-controlled study in which six patients received clonazepam. The mean improvement in the BARS scores for patients on clonazepam was 71%, whereas the overall mean improvement in the placebo group was 38%. Tolerance and the abuse potential of benzodiazepines may limit their usage (Reference BlaisdellBlaisdell, 1994).
Any further studies should compare two or more agents. A very useful future study would be a doubleblind three-arm trial comparing the efficacy of a serotonin antagonist such as cyproheptadine with a betablocker and placebo. Future trials should last longer and involve a greater number of patients than the currently published trials. It would also be useful if the studies involved patients with both acute and chronic akathisia, to see whether there is any difference in the response to therapy.
Evidence supporting the use of serotonin antagonists
Serotonin modulates central dopamine activity. The interaction is complex, but it is thought that 5-HT2 antagonism potentiates dopamine activity (Reference Lipinski, Mallaya and ZimmermanLipinski et al, 1989). This leads to the hypothesis that 5-HT2 antagonists treat the symptoms of akathisia (Reference Weiss, Aizenberg and HermeshWeiss et al, 1995).
Clinical evidence points to serotonergic involvement in the pathophysiology of NIA. First, serotonin antagonistic atypical neuroleptics such as olanzapine cause less akathisia than typical neuroleptics such as haloperidol, which have little activity at serotonin receptors (Reference Tran, Tollefson and SangerTran et al, 1999). Second, there are reports of akathisia with selective serotonin reuptake inhibitors such as fluoxetine (Reference Lipinski, Mallaya and ZimmermanLipinski et al, 1989).
Serotonergic involvement is supported by the apparent efficacy of serotonin antagonists in NIA. Most of the trial data are from open or placebo-controlled small-scale studies. Although this severely limits the usefulness of the results. It does appear that serotonin antagonists effectively treat akathisia. The trials also used many patients with akathisia resistant to traditional therapies.
There are no published studies comparing the different classes of therapeutic agent. Individual studies lack uniformity, making it difficult to draw conclusions on the relative effectiveness of the various agents. Of the three agents discussed above (mianserin, ritanserin and cyproheptadine) there is more evidence supporting the use of low-dose mianserin. As noted, one study demonstrated at least a 40% reduction in the symptoms of NIA compared to placebo. Thus, from the current evidence it can be concluded that mianserin is at least as effective as propranolol and clonazepam.
Two studies showed that ritanserin effectively treats NIA. Unfortunately, neither study was double-blind. Ritanserin is not licensed in the UK, and therefore availability may be limited.
While the study involving cyproheptadine showed a mean symptom reduction of more than 75%, this study was not placebo-controlled. Double-blind placebo-controlled studies are required to confirm this reported efficacy. More long-term data are also required supporting the use of mianserin and cyproheptadine in the treatment of chronic akathisia.
One major advantage of the serotonin antagonists is that they appear relatively well tolerated, have few contraindications and a low potential for misuse.
Summary
The available evidence indicates that serotonin antagonists effectively treat acute NIA. The studies lack uniformity, and there are no studies comparing serotonin antagonists with each other or with alternative agents. It is therefore difficult to form conclusions regarding the relative effectiveness of the various agents.
The side-effects profile and low potential for misuse of serotonin antagonists may offer advantages over existing agents. If controlled clinical trials show that serotonin antagonists are as effective as other agents, they should be considered as first- or second-line antiakathetic agents.




eLetters
No eLetters have been published for this article.