Inflammatory bowel diseases (IBD), including ulcerative colitis and Crohn's disease, are chronic relapsing inflammatory conditions of unknown aetiology in the intestinal tract(Reference Mudter and Neurath1, Reference Yamamoto-Furusho and Podolsky2). Based on the present understanding of the pathogenesis of IBD, activated immune cells can destroy the intestinal barrier either directly via cytotoxicity or indirectly through the release of cytokines, reactive oxygen species and other metabolites(Reference Yamamoto-Furusho and Podolsky2). In addition, the risk of developing colorectal cancer increases approximately 0·5–1 % each year after 7 years in patients with IBD(Reference Rubin and Kavitt3). Despite the well-known functional link between inflammation and colon cancer, the pathways regulating the initiation of colon cancer in the presence of chronic inflammation remain unclear. Therefore, it is important to identify the regulatory mechanisms underlying inflammation and tumorigenesis in the colon.
Dietary long-chain n-3 PUFA found in fish oil (FO), specifically EPA (20 : 5 Δ5,8,11,14,17) and DHA (22 : 6Δ4,7,10,13,16,19), are capable of ameliorating critical determinants which link inflammation and cancer development and progression(Reference Davidson, Nguyen and Hokanson4–Reference Hudert, Weylandt and Lu6). In contrast, dietary maize oil (MO) rich in n-6 PUFA (found in vegetable oils), e.g. linoleic acid (18 : 2Δ9,12) and arachidonic acid (20 : 4Δ5,8,11,14), is considered as a pro-inflammatory dietary component that enhances the development of colon tumours(Reference Chang, Chapkin and Lupton7, Reference Whelan and McEntee8). To date, the effects of n-3 PUFA on susceptibility to colitis and colon cancer have not been determined, and a unifying mechanistic hypothesis addressing how n-3 PUFA selectively modulate colonic inflammation is lacking. Furthermore, it has been demonstrated that n-3 PUFA can inhibit NF-κB activation(Reference Hudert, Weylandt and Lu6, Reference Nowak, Weylandt and Habbel9). This is significant because NF-κB can modulate several steps in the inflammatory cascade by inducing the expression of pro-inflammatory cytokines such as TNF-α, IL-1β, IFN-γ, IL-12, cyclo-oxygenase (COX)-2 and inducible NO synthase(Reference Hollenbach, Vieth and Roessner10, Reference Waetzig, Seegert and Rosenstiel11), and is required for proliferation/apoptosis homeostatic regulation and the protection from acute inflammation of the intestine(Reference Karrasch and Jobin12, Reference Steinbrecher, Harmel-Laws and Sitcheran13). Therefore, it is important to further elucidate the link between NF-κB-dependent signalling pathways and dietary n-3 PUFA.
Curcumin is a natural polyphenol isolated from the dried rhizomes of Curcuma longa (turmeric). As the active component in turmeric, curcumin has been widely used in traditional medicine in India and Southeast Asia(Reference Duvoix, Blasius and Delhalle14). Moreover, curcumin has been shown to ameliorate inflammation associated with experimental colitis(Reference Deguchi, Andoh and Inatomi15–Reference Camacho-Barquero, Villegas and Sánchez-Calvo17). These data suggest that selective dietary polyphenolics may favourably modulate inflammatory responses in the colon. The chemoprotective effects of curcumin appear to be mediated, in part, through NF-κB inhibition(Reference Camacho-Barquero, Villegas and Sánchez-Calvo17–Reference Milacic, Banerjee and Landis-Piwowar19). Therefore, in the present study, we have evaluated the effect of curcumin supplementation, in the presence or absence of dietary n-3 PUFA, on NF-κB activation and the resolution of chronic inflammation in the colon.
Dextran sodium sulphate (DSS)-induced inflammation is an excellent preclinical mucosal wounding model of colitis that exhibits many phenotypic characteristics relevant to the human disease(Reference Seril, Liao and Yang20). Typically, repeated cycles of DSS treatment are used to induce chronic intestinal inflammation(Reference Xiao, Gulen and Qin21). In the present study, we exposed mice to either one or two cycles of DSS treatment to induce acute and chronic inflammation, respectively. We determined how dietary n-3 PUFA and curcumin influence (1) mortality, (2) colonic inflammation and injury scores, (3) epithelial cytokinetics, e.g. apoptosis and proliferation, (4) NF-κB activation and (5) global mucosal gene expression.
Materials and methods
Animals and diet
Male C57BL/6 mice, 6- to 8-weeks old, were obtained from Jackson Laboratories (Bar Harbor, ME, USA) and housed in a temperature- and humidity-controlled facility with a 12 h light–12 h dark cycle. All procedures followed the guidelines approved by Public Health Service Policy and the Institutional Animal Care and Use Committee at Texas A&M University. After 1 week of acclimatisation on a standard pelleted diet, animals were randomly grouped (n 15) and fed ad libitum one of the four experimental diets (2 × 2 design) (see Table S1 of the supplementary material, available online at http://www.journals.cambridge.org/bjn): MO diet containing 5 % MO; FO diet containing 4 % FO and 1 % MO (EPA and DHA content was 8·1 and 8·5 wt %, respectively); MO plus curcumin diet (MO-cur) containing 5 % MO and 2 % curcumin; FO plus curcumin diet (FO-cur) containing 4 % FO, 1 % MO and 2 % curcumin. The diets contained (per 100 g) 42 g sucrose, 20 g casein, 22 g maize starch, 0·3 g dl-methionine, 3·5 g 76A salt mix, 1·0 g American Institute of Nutrition (AIN) 76A mineral mix, 0·2 g choline chloride, 6 g fibre (cellulose), 5 g fat. The level of curcumin (2 %) has been shown to modulate disease pathology in the DSS mouse model(Reference Deguchi, Andoh and Inatomi15), and the relative dose is comparable with levels previously used in a human clinical trial(Reference Hanai and Sugimoto22). To prevent the formation of oxidised lipids, diets were stored at − 20°C and provided fresh to animals each day. MO was obtained from Dyets (Bethlehem, PA, USA), and vacuum deodorised menhaden FO was obtained from Omega Protein (Houston, TX, USA). Curcumin C3 complex, which contains 80 % curcumin, 3 % bisdemethoxy curcumin and 17 % demethoxy curcumin, was provided by Sabinsa Corporation (Payson, UT, USA). Heavy metals, including As and Pb, were below the limits of detection.
Colitis induction and histological scoring
Animals were fed experimental diets 1 week before DSS (molecular weight, 36 000–50 000; MP Biomedicals, Solon, OH, USA) treatment (see Fig. S1 of the supplementary material, available online at http://www.journals.cambridge.org/bjn). To induce chronic inflammation, 2·5 % DSS was administered in the drinking-water for 5 d, followed by 16 d of tap water. This cycle was repeated with a lower dose of DSS (3 d of 1·5 % DSS followed by a 14 d recovery period) after which mice were euthanised humanely. Acute inflammation was induced by 5 d of 2·5 % DSS, followed by a 3 d recovery period. At each necropsy interval, the entire colon was removed, flushed with PBS, fixed in 4 % paraformaldehyde and paraffin embedded. Sections were stained with haematoxylin and eosin. Histological examination was performed in a blinded manner by a board-certified pathologist (B. R. W.), and the degree of inflammation (score, 0–3) and epithelial injury (score, 0–3) in microscopic cross-sections of the colon was graded as described previously(Reference Jia, Lupton and Smith23). Briefly, the presence of occasional inflammatory cells in the lamina propria was assigned a value of 0; increased numbers of inflammatory cells in the lamina propria as 1; confluence of inflammatory cells, extending into the submucosa, as 2; and transmural extension of the infiltrate as 3. For the evaluation of epithelial injury, no mucosal damage was scored as 0; discrete lympho-epithelial lesions were scored as 1; surface mucosal erosion or focal ulceration was scored as 2 and extensive mucosal damage associated with the deeper structures of the bowel wall was scored as 3.
In situ apoptosis and proliferation measurement
Intestinal apoptosis was measured in paraformaldehyde-fixed, paraffin-embedded tissues using the terminal deoxynucleotidyl transferase-mediated uridine 5′triphosphate-biotin nick-end (Oncor, Dallas, TX, USA) labelling method(Reference Hong, Bancroft and Turner24). Cell proliferation was measured following bromodeoxyuridine injection (Zymed, South San Francisco, CA, USA) as described previously(Reference Ma, Finnell and Davidson25).
Assessment of NF-κB activity
NF-κB activation was measured by quantifying p65/Rel A activation as described previously(Reference Kim, Fan and Smith26). In brief, whole-cell protein from snap-frozen colonic mucosa scraping was extracted using a Nuclear Extraction Kit (Active Motif, Carlsbad, CA, USA) and subsequently incubated with oligonucleotides which comprise the NF-κB consensus DNA-binding site (5′-GGGACTTTCC-3′) to detect activated p65/Rel A.
Total RNA isolation
At each necropsy interval, mucosa scrapings from the colon were stored in mirVana™ reagent at − 80°C. Total RNA was extracted from each sample using mirVana™ miRNA Isolation Kit (Ambion, Austin, TX, USA) according to the manufacturer's instructions. The concentration and quality of total RNA were assessed using a Nanodrop spectrophotometer (Nanodrop, Wilmington, DE, USA) and an Agilent 2100 Bioanalyser (Agilent Technologies, Waldbronn, Germany), respectively.
Codelink mouse whole-genome microarray assay
Quadruplicate total RNA samples (including non-DSS, MO-fed control) were processed to generate biotin-labelled complementary RNA via a modified Eberwine RNA amplification protocol using a CodeLink iExpress Kit (Applied Microarray, Tempe, AZ, USA). Labelled complementary RNA was applied to CodeLink Mouse Whole Genome Bioarrays (Applied Microarray), which contain 34 967 unique probe sequences. After incubation, slides were washed, stained and scanned by Gene Pix. Array images were processed using CodeLink system software version 5.0. The resulting files were imported into zRMicroArray(Reference Zlatev27) for analysis. After median and log transformation, the gene expression was examined for normality (Shapiro–Wilk test) of the empirical distribution within each experimental group. Only genes that passed the significance test (P < 0·05) were considered for subsequent analyses(Reference Strimmer28).
Immunohistochemistry of pSTAT3
Sections (4 μm thick) were cut from paraffin-embedded colon ‘swiss rolls’ for immunohistochemistry of pSTAT3tyr705 (Cell Signalling, Danvers, MA, USA). In brief, tissue sections were deparaffinised and antigen retrieval was performed using EDTA buffer, pH 8. Primary antibody (1:50) was incubated overnight at 4°C followed by the addition of Signal Stain Boost IHC Detection Reagent (horseradish peroxidase, rabbit; Cell Signalling) for 30 min. Slides were developed using diaminobenzidine. Staining of pSTAT3 was quantified using a Nikon Cool-SNAP camera and NIS elements software. From the distal, middle and proximal regions of the colon, four representative stained areas (hot spots) were selected, corresponding to a total of twelve images from each slide per animal. Stained areas in the mucosa within the intensity threshold were recorded. On average, twelve images per animal were read in a blinded fashion.
Statistics
Data are expressed as means with their standard errors. Differences between experimental groups were analysed using ANOVA from the SPSS software package and ANOVA R-package which is embedded in zRMicroArray. Adjusted P values < 0·05 were accepted as significant.
Results
Body weight and colon length
Animal body weight and colon length were recorded at termination points. Body weight dropped significantly following acute DSS treatment but no differences were observed between diets (see Fig. S2(A) of the supplementary material, available online at http://www.journals.cambridge.org/bjn). Similarly, colon length was shortened significantly following both acute and chronic DSS treatment, and no differences between diets were detected (see Fig. S2(B) of the supplementary material, available online at http://www.journals.cambridge.org/bjn).
Dietary fish oil and curcumin, independently and in combination, enhance acute dextran sodium sulphate-induced mortality
Animal mortality was determined following both acute (one cycle) and chronic (two cycles) DSS exposure. The MO diet was used as a baseline control because it contains no EPA, DHA or curcumin. Therefore, curcumin supplementation to the MO diet (MO-cur) represents the isolated effect of curcumin. The FO diet represents the effect of FO alone, while the FO-cur diet represents the combined effects of FO and curcumin. During the acute inflammatory phase (Fig. 1(A) and (C)), FO-fed mice exhibited the highest mortality (40 %, seventeen of forty-three) compared with MO with the lowest mortality (3 %, one of twenty-nine) (P = 0·0008). Addition of curcumin to MO increased (P = 0·003) the mortality to 37 % (thirteen of thirty-five). FO and curcumin combination treatment also increased (P = 0·03) the mortality to 28 %. These results demonstrate that FO or curcumin supplementation increases the mortality rate following acute DSS exposure. Mice surviving acute colitis and exposed to a second round of DSS (chronic phase) died infrequently (MO, two of seventeen, FO, one of ten, MO-cur, one of eleven, FO-cur, two of fourteen) and exhibited no effect of diet (Fig. 1(B) and (D)). These data indicate that n-3 PUFA and curcumin supplementation exacerbate susceptibility to DSS-induced acute colitis.
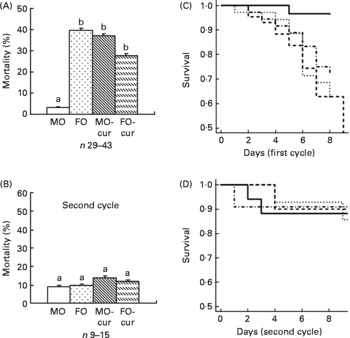
Fig. 1 Fish oil (FO) and curcumin feeding increase mortality. Animal mortality following dextran sodium sulphate (DSS) treatment was recorded during the (A) acute and (B) chronic inflammatory phases. Kaplan–Meier plots are shown for the (C) acute and (D) chronic experimental phases. Maize oil (MO, ![]() ), 5 % MO; FO (
), 5 % MO; FO (![]() ), 4 % FO+1 % MO; MO plus curcumin (MO-cur;
), 4 % FO+1 % MO; MO plus curcumin (MO-cur; ![]() ), 5 % MO+2 % curcumin; FO plus curcumin (FO-cur;
), 5 % MO+2 % curcumin; FO plus curcumin (FO-cur; ![]() ), 4 % FO+1 % MO+2 % curcumin. Acute phase represents treatment with DSS for 5 d followed by a 3 d recovery period; chronic phase represents two cycles of DSS exposure followed by a 14 d recovery period. Refer to Fig. S1 of the supplementary material, available online at http://www.journals.cambridge.org/bjn for details. a,b Mean values with unlike letters were significantly different (P < 0·05).
), 4 % FO+1 % MO+2 % curcumin. Acute phase represents treatment with DSS for 5 d followed by a 3 d recovery period; chronic phase represents two cycles of DSS exposure followed by a 14 d recovery period. Refer to Fig. S1 of the supplementary material, available online at http://www.journals.cambridge.org/bjn for details. a,b Mean values with unlike letters were significantly different (P < 0·05).
Fish oil and curcumin modulate dextran sodium sulphate-induced pathology
To explore the effect of n-3 PUFA and curcumin on DSS-induced colitis, mice were treated with a single 5 d cycle of DSS followed by a 3 d recovery (acute) or two cycles of DSS (chronic), and colonic inflammation and mucosal injury were assessed. Mouse colon sections were haematoxylin and eosin stained for histological scoring (Fig. 2(A)). Injury and inflammatory scores represent the degree of ulceration and immune cell infiltration, respectively. FO with or without curcumin supplementation and MO with curcumin increased (P < 0·05) injury scores, compared with control (MO), following both acute and chronic DSS exposure (Fig. 2(B)). This is consistent with animal survival data indicating that dietary FO and/or curcumin promoted mucosal injury and mortality during acute-phase DSS exposure (Fig. 1(A) and (C)). With respect to the inflammatory score, the addition of curcumin to FO was protective, especially following chronic DSS exposure (Fig. 2(C)). These data suggest that n-3 PUFA and curcumin may affect the pathology of DSS-induced colitis by modulating immune cell infiltration and activation in both acute and chronic phases.
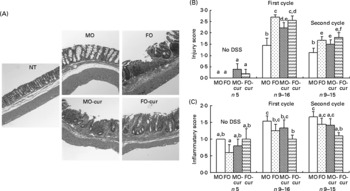
Fig. 2 Histological features of colonic inflammation and mucosal injury. Mice were treated with an acute or chronic dextran sodium sulphate (DSS) regimen, and colonic inflammation and mucosal injury were assessed. (A) Representative haematoxylin and eosin-stained colonic sections from mice were exposed to chronic DSS treatment (100 × magnification). Crypts were severely distorted and the epithelium was denuded in fish oil (FO), FO curcumin and maize oil (MO) curcumin treatments compared with MO (control) and non-DSS-treated animals fed MO (NT). (B) Injury scores and (C) inflammatory scores. Values are means, with their standard errors represented by vertical bars. No DSS, indicates no DSS treatment; first cycle, indicates 5 d DSS followed by a 3 d recovery period; second cycle, indicates two cycles of DSS exposure. a,b,c,d,e,f Mean values with unlike letters were significantly different (P < 0·05). MO-cur, MO plus curcumin; FO-cur, FO plus curcumin.
Fish oil and curcumin combination suppresses dextran sodium sulphate-induced NF-κB activation
We examined NF-κB activation status following chronic DSS exposure in order to elucidate the mechanism by which n-3 PUFA and curcumin modulate the inflammatory response in the colon. In comparison with the MO control, only the dietary FO and curcumin combination suppressed NF-κB activity in the colonic mucosa (Fig. 3). Together with inflammatory score data (Fig. 2), these results suggest a mechanistic link between the enhanced resolution of chronic inflammation and the suppression of NF-κB by FO-cur feeding.
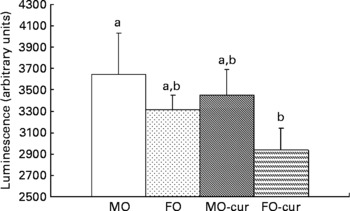
Fig. 3 Fish oil (FO) and curcumin suppress NF-κB activation in the colonic mucosa. Colonic mucosa was isolated following two cycles of dextran sodium sulphate treatment (chronic inflammatory phase) and p65 NF-κB nuclear activation was determined. Values are means, with their standard errors represented by vertical bars (seven mice per treatment). a,b Mean values with unlike letters were significantly different (P < 0·05). Refer to Fig. 1 for legend details. MO-cur, maize oil plus curcumin; FO-cur, FO plus curcumin.
Fish oil and curcumin combination promotes mucosal cell proliferation in the colon
Next, we investigated the ability of FO and curcumin to modulate mucosal cytokinetics following DSS-induced exposure. Epithelial apoptosis was measured by the terminal deoxynucleotidyl transferase (terminal deoxynucleotidyl transferase-mediated uridine 5′triphosphate-biotin nick-end labelling) assay. As shown in Fig. 4(A) and Fig. S3 of the supplementary material (available online at http://www.journals.cambridge.org/bjn), dietary FO enhanced epithelial apoptosis compared with the MO control, which may explain, in part, the elevated injury score associated with FO feeding (Fig. 2(B)). Since the initiation of cell proliferation is a prerequisite for mucosal repair(Reference Okamoto and Watanabe29), we examined epithelial cell proliferation by bromodeoxyuridine incorporation. As shown in Fig. 4(B), feeding FO-cur resulted in an enhanced rate of proliferation relative to MO (control). This suggests that the FO-cur diet promotes repair of the colonic epithelium.
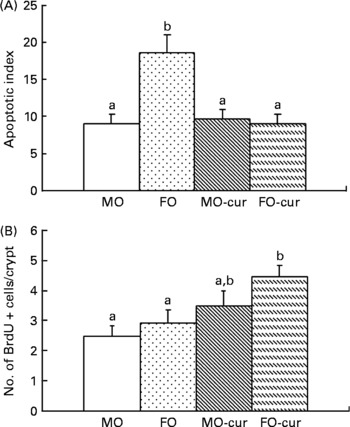
Fig. 4 Fish oil (FO) and curcumin modulate colonocyte cytokinetics. In situ detection of (A) epithelial apoptosis and (B) proliferation was quantified using terminal deoxynucleotidyl transferase-mediated uridine 5′triphosphate-biotin nick-end labelling and bromodeoxyuridine (BrdU) incorporation, respectively. Animals were terminated following two cycles of dextran sodium sulphate treatment (chronic inflammatory phase). Data are expressed as an apoptotic index (total number of apoptotic cells per 100 crypts). Values are means, with their standard errors represented by vertical bars (three to five mice per treatment, 115–338 crypts per column). a,b Mean values with unlike letters were significantly different (P < 0·05). MO-cur, maize oil plus curcumin; FO-cur, FO plus curcumin.
Diet and dextran sodium sulphate exposure modulate mucosal gene expression
Gene expression in chronic colitis was determined by comparing the treated groups (two cycles of DSS) with MO-fed no-DSS untreated control (MO-con) mice. As shown in Fig. 5, genes up- or down-regulated by FO, curcumin and FO-cur combination were contrasted in order to elucidate the biological processes contributing to the tissue injury and inflammation phenotypes. Because dietary FO-cur reduced the inflammatory score and NF-κB activation, and elevated intestinal cell proliferation (Figs. 2–4), we compared gene expression profiles from mice fed FO-cur with the mice fed the MO control diet without DSS treatment (MO-con) (Fig. 5(A)). Specifically, genes that were differentially expressed in the MO, MO-cur and FO groups relative to the MO-con following chronic colitis but whose change was blocked by the FO-cur diet were identified. Examples include a hypoxic perinecrotic marker of tumour angiogenesis, neuritin precursor (Nrn1)(Reference Le Jan, Le Meur and Cazes30), which was down-regulated by 0·4–0·5-fold in the MO-DSS, FO-DSS, MO-cur-DSS groups relative to the MO-con and FO-cur-DSS treatment. In addition, the FO-cur combination prevented the up-regulation of pro-inflammatory genes (e.g. lymphocyte antigen 9 (Slamf3), suppressor of cytokine signalling 3 (Socs3), TNF receptor superfamily member 1B precursor (Tnfrsf1b) and macrophage colony-stimulating factor 1 receptor precursor (Csf1r)), expressed in immune cells(Reference Kingsbury, Feeney and Nong31–Reference Patel and Player33). These observations are consistent with the ability of FO-cur to resolve inflammation (Fig. 2). Up-regulation of the secretory phospholipase A2 precursor (Pla2g2c) and myosin regulatory light chain 2 (Myl7) was also prevented in the FO-cur-DSS group.
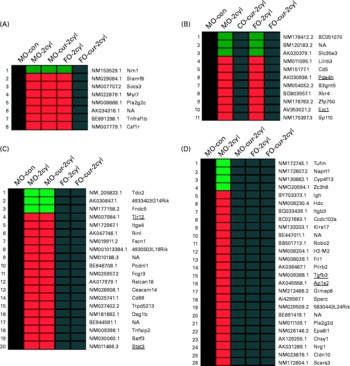
Fig. 5 Fish oil (FO) and curcumin supplementation differentially regulate intestinal gene expression. Distinctive gene expression signatures are found in the colonic mucosa following FO plus curcumin (FO-cur) dietary treatments and exposure to dextran sodium sulphate (DSS). Functional annotation of gene sets with similar profiles are compared: (A) expression pattern for genes strictly affected by FO-cur combination therapy; i.e. no DSS treatment maize oil (MO) control (con) diet (MO-con) and FO-cur-DSS combination v. other, (B) expression pattern for genes strictly affected by curcumin therapy; i.e. MO-con, MO plus curcumin (MO-cur)-DSS, and FO-cur-DSS supplementation v. other, (C) expression pattern for genes strictly affected by FO feeding; i.e. MO-con, FO-DSS, FO-cur-DSS v. other, (D) expression pattern for genes affected by FO or curcumin treatment; i.e. MO-DSS effect, MO-DSS (positive con) v. other. Significantly (P < 0·05) up-regulated genes are shown in red, and down-regulated genes are shown in green. Black boxes indicate no significant difference (P>0·05) compared with MO-con. NF-κB-related genes are underlined. n 5 Mice for MO-con, n 9–10 for all other groups. All animals were exposed to two DSS cycles (cyl) except for MO-con. NA, gene name not available.
Genes that were affected by curcumin alone are shown in Fig. 5(B). In this comparison, diets containing curcumin (MO-cur-DSS and FO-cur-DSS) reduced the expression of ELKS/RAB6-interacting/CAST family member 1 (Erc1) during chronic inflammation to levels similar to the MO-con diet. Erc1 is an essential regulatory subunit of the IκB kinase complex and important for NF-κB activation(Reference Ducut Sigala, Bottero and Young34), suggesting that curcumin is capable of modulating chronic inflammation via the inhibition of NF-κB activation in the colon mucosa. Curcumin also blocked the DSS-induced increase of pro-inflammatory genes such as T-cell surface glycoprotein CD5 precursor (Cd5), paired Ig-like receptor B (Lilrb3), cAMP-specific 3′,5′-cyclic phosphodiesterase 4B (Pde4b), nuclear body protein (Sp110) and solute carrier family 35, member E3 (Slc35e3). It is noteworthy that Pde4b inhibitors exhibit profound anti-inflammatory effects and have utility in the treatment of cancer and the regulation of energy balance(Reference Smith, Wilkinson and Savage35, Reference Zhang, Maratos-Flier and Flier36).
Genes that were selectively affected by FO feeding are shown in Fig. 5(C). In this comparison, mice that received FO-DSS and FO-cur-DSS treatment produced an expression profile similar to MO-con. Of interest in Fig. 5(C), two NF-κB-regulated genes had reduced the expression in response to FO, i.e. Toll-like receptor 12 (Tlr12) and signal transducer and activator of transcription 3 (Stat3). An immunohistochemical analysis of pSTAT3 Tyr 705 performed to confirm the microarray results showed that FO-DSS and FO-cur-DSS treatment had suppressed levels of pSTAT3 compared with MO-cur-DSS treatment but not MO-DSS (see Fig. S4 of the supplementary material, available online at http://www.journals.cambridge.org/bjn). Although the function of Tlr12 in colonic inflammation is not known, Stat3 has been reported to be a required regulator for the survival of intestinal epithelial cells and the development of colitis-associated tumorigenesis(Reference Bollrath, Phesse and von Burstin37, Reference Grivennikov, Karin and Terzic38). FO feeding also reduced the expression of pro-inflammatory genes including low-affinity Igγ Fc region receptor III precursor (Fcgr3), carcinoembryonic antigen-related cell adhesion molecule 14 (Ceacam14), early T-cell activation antigen p60 (Cd69), TNFα-induced protein 2 (Tnfaip2) and basic leucine zipper transcriptional factor ATF-like 3 (Batf3). Interestingly, Batf3 may be required for the differentiation of IL17-producing T-helper (TH17) cells(Reference Schraml, Hildner and Ise39).
We also examined genes that were uniquely affected by either FO or curcumin treatment, i.e. changes that were exclusive to MO-DSS treatment (Fig. 5(D)). Using this comparison, two NF-κB target genes, transforming growth factor β-3 precursor (Tgfb3) and AP-1 complex subunit sigma-2 (Ap1s2), were selectively up-regulated in MO-DSS mice. This is noteworthy because TGFβ3 is a well-known pro-inflammatory marker of IBD(Reference Kanazawa, Tsunoda and Onuma40). Other effector pro-inflammatory genes which were up-regulated include scavenger receptor class A member 3 (Scara3), paired Ig-like type 2 receptor β 2 precursor (Pilrb2), killer cell lectin-like receptor, subfamily A, member 17 (Klra17)(Reference Balla, Kósa and Kiss41–Reference Tai, Goulet and Belanger43), Ig heavy-chain C gene segment (Igh) and histidine decarboxylase (Hdc). Interestingly, Hdc is modulated by TLR agonists(Reference Funayama, Huang and Asada44). MO plus DSS treatment also increased additional genes that may be involved in mucosa wound repair including pro-neuregulin-1 (Nrg1)(Reference Buac, Xu and Cronin45), chondroitin sulphate synthase 1 (Chsy1)(Reference Zou, Jiang and Zhang46) and epidermal growth factor receptor kinase substrate 8-like protein 1 (Eps8l1)(Reference Offenhäuser, Borgonovo and Disanza47). Genes specifically down-regulated by MO feeding in the presence of DSS include elongation factor Tu, mitochondrial precursor (Tufm), nicotinate phosphoribosyltransferase (Naprt1), cytochrome P450, family 4, subfamily f, polypeptide 13 (Cyp4f13) and Zn finger CCCH domain-containing protein 8 (Zc3h8).
Discussion
Dietary n-3 PUFA and curcumin are receiving substantial attention for their anti-tumorigenic and anti-inflammatory properties(Reference Jia, Lupton and Smith23, Reference Chapkin, McMurray and Lupton48–Reference Weaver, Ivester and Seeds50). In the present study, we assessed the combined effects of these two bioactive dietary components on the resolution of both acute and chronic experimental colitis. FO and curcumin, both alone and in combination, unexpectedly enhanced animal mortality and exacerbated colonic mucosa injury following an acute inflammatory episode (Figs. 1(A), (C) and Fig. 2(B)). Wound repair occurs in three overlapping but distinct stages: inflammation; new tissue formation; remodelling(Reference Gurtner, Werner and Barrandon51). Inflammation occurs immediately after tissue damage and is essential for wound repair. In refractory cases of IBD or experimental chronic inflammation, mucosal repair is disrupted. Repair of damaged intestinal mucosa is regulated by multiple factors at both molecular and cellular levels including innate and adaptive immune cells(Reference Okamoto and Watanabe29, Reference Blikslager, Moeser and Gookin52, Reference Barrientos, Stojadinovic and Golinko53). Although many elements of the acute inflammatory response influence wound repair, particular attention has been paid to growth factors, PG and cytokines(Reference Barrientos, Stojadinovic and Golinko53, Reference Rakoff-Nahoum and Medzhitov54). Interestingly, there is a growing body of evidence indicating that n-3 PUFA(Reference Chapkin, McMurray and Lupton48, Reference Chapkin, Kim and Lupton55) and curcumin(Reference Kim, Fan and Smith26, Reference Koeberle, Northoff and Werz56, Reference Chakravarti, Myers and Aggarwal57) inhibit these same mediators, which are required for protection from DSS-induced colonic injury. Although there is a significant redundancy in the inflammatory response(Reference Gurtner, Werner and Barrandon51), it is possible that FO and curcumin delay epithelial repair mechanisms, which would enhance absorption of bacterial toxins and, therefore, increase mortality. There is also a remote possibility that dietary lipid source may influence curcumin bioavailability. However, there is no precedent in the literature to suggest that the lipid source can influence curcumin absorption/metabolism/bioactivity.
The second stage of repair of injured intestinal mucosa, new tissue formation, is a process of organised restitution, proliferation and differentiation of mucosal epithelium. Within hours of injury, re-epithelialisation is initiated and epithelial cell proliferation is stimulated in crypts near the damaged mucosal area(Reference Okamoto and Watanabe29, Reference Blikslager, Moeser and Gookin52). Our data show that FO-cur enhanced colonic epithelial proliferation compared with other diets (Fig. 4(B)), which suggests that this nutritional combination may promote mucosal repair during chronic inflammation. In contrast, FO alone increased epithelial cell apoptosis (Fig. 4(A)). This is noteworthy because sustained epithelial apoptosis can preclude mucosal healing and decrease animal survival(Reference Davidson, Nguyen and Hokanson4), which may explain the elevated injury scores observed in FO-fed mice during acute-phase inflammation. Along these lines, it has been shown that the balance between colonic epithelial cell proliferation and apoptosis can be modulated by dietary n-3 PUFA, conferring resistance to toxic carcinogenic agents(Reference Davidson, Nguyen and Hokanson4, Reference Ma, Finnell and Davidson25, Reference Courtney, Matthews and Finlayson58). Additional experiments are required to further address the impact of diet on colonic cytokinetics following acute DSS exposure.
With respect to mechanisms which mediate intestinal cell cytokinetics, it has been shown that Toll-like receptor-4 (TLR4)-PG-dependent signalling directly regulates proliferation and apoptosis in response to acute colitis(Reference Rakoff-Nahoum and Medzhitov54, Reference Fukata, Michelsen and Eri59, Reference Fukata, Chen and Klepper60). Although TLR4 activation is beneficial in the short term, chronic signalling may lower the threshold for inflammation-associated colon cancer(Reference Rakoff-Nahoum and Medzhitov61). With respect to diet, DHA and curcumin appear to be pan-inhibitors for various TLR(Reference Wong, Kwon and Choi62, Reference Huang, Zhao and Kim63). Additional studies are needed in order to determine if these observations can be validated in vivo. It is also well documented that n-3 PUFA (EPA and DHA) antagonise arachidonic acid-derived PG (PGE2 and PGD2) in the colonic mucosa(Reference Hong, Bancroft and Turner24, Reference Lee, Lupton and Aukema64). Curcumin has also been reported to suppress PGE2 formation by blocking the expression of COX-2 and microsomal PGE2 synthase-1 and inhibiting the activity of microsomal PGE2 synthase-1(Reference Koeberle, Northoff and Werz56). This is noteworthy because PGE2 is capable of enhancing cell proliferation, angiogenesis, cell migration and invasion as well as inhibiting apoptosis and enhancing tumour growth(Reference Buchanan and DuBois65). Recently, it has been demonstrated that PGE2 is important for the healing of ulcers and epithelial injury(Reference Hatazawa, Tanaka and Tanigami66, Reference Ungaro, Fukata and Hsu67). Taken together, these data suggest that n-3 PUFA and curcumin modulate the resolution of inflammation and mucosa repair, in part, by suppressing the TLR4/COX-2/PGE2 signalling axis.
We demonstrate for the first time that FO with or without curcumin exacerbates acute inflammatory responses in the DSS wounding mouse model. Specifically, MO (control diet)-fed, DSS-treated mice were protected against acute death and mucosal damage compared with the FO with or without curcumin treatments (Figs 1 and 2). In contrast, FO-cur afforded the greatest protection with respect to mucosal (acute and chronic) inflammation (Fig. 2). Overall, these data indicate an enhanced ability of FO-cur-fed mice to resolve chronic inflammation of the colon. From a mechanistic standpoint, dietary FO-cur combination significantly suppressed NF-κB activity in the colon (Figs. 2(C) and 3), suggesting that FO and curcumin act in a combinatory manner. The separate effects of these two bioactive components have been reported previously(Reference Nowak, Weylandt and Habbel9, Reference Bharti, Donato and Singh18, Reference Fan, Ly and Barhoumi68). NF-κB is an important intracellular regulator of both the inflammatory response and mucosal integrity via the TLR4/COX-2/PGE2 signalling axis, which is important for the healing of epithelial injury(Reference Ungaro, Fukata and Hsu67). Recent findings have indicated that NF-κB can exert either a deleterious or a protective function in the intestine, depending on the stimuli encountered(Reference Waetzig, Seegert and Rosenstiel11–Reference Steinbrecher, Harmel-Laws and Sitcheran13, Reference Chapkin, McMurray and Lupton48). NF-κB regulates the gene expression of pro-inflammatory mediators including IL-1β, TNF-α, IL-12p40 and IL-23p19, which contribute to the pathophysiology of chronic intestinal inflammatory diseases(Reference Waetzig, Seegert and Rosenstiel11–Reference Steinbrecher, Harmel-Laws and Sitcheran13).
Mucosal microarray analysis revealed that dietary FO, curcumin and FO-cur combination differentially modulated the expression of genes induced by DSS treatment (Fig. 5). A regulator of NF-κB activation, Erc1, was suppressed at the transcriptional level following supplementation with curcumin (Fig. 5(B)). As an essential regulatory subunit of the IκB kinase (IKK) complex(Reference Ducut Sigala, Bottero and Young34), Erc1 may be a new target for curcumin which modulates NF-κB activation in addition to inhibiting IKK and Akt activation(Reference Aggarwal, Ichikawa and Takada69). The suppression of NF-κB target genes by FO or curcumin is consistent with the critical role of this transcription factor as a key regulator of intestinal inflammation. Interestingly, FO feeding blocked DSS-induced Stat3 up-regulation (Fig. 5(C)). Stat3 has been reported recently as a key regulator in IBD and colon cancer(Reference Bollrath, Phesse and von Burstin37, Reference Grivennikov, Karin and Terzic38). Specifically, Stat3 activation in enterocytes is required for cell survival and its hyperactivation promotes colitis associated tumorigenesis and growth(Reference Bollrath, Phesse and von Burstin37, Reference Grivennikov, Karin and Terzic38). In addition to intestinal epithelial cells, Stat3 is constitutively activated in mucosa immune cell types including dendritic cells, macrophages and T-cells(Reference Nishihara, Ogura and Ueda70–Reference Kortylewski, Xin and Kujawski72). The Stat3 pathway in CD4+ T cells promotes IL-17-producing Th cell (Th17) development which mediates immune responses in autoimmune disease and IBD-induced cancer development(Reference Nishihara, Ogura and Ueda70, Reference Wu, Rhee and Albesiano71). Stat3 activation in immune cells promotes an IL-23-mediated procarcinogenic immune response while inhibiting IL-12-dependent Th1-mediated antitumour immunity(Reference Kortylewski, Xin and Kujawski72). Our results suggest that Stat3 may be a new target of the antitumorigenic n-3 PUFA. We are currently investigating whether n-3 PUFA modulate Th17 polarisation in the intestine. Overall, FO and curcumin suppressed the up-regulation of pro-inflammatory gene expression relative to the MO (control) diet, which is consistent with human studies showing that EPA+DHA intake decreases the gene expression of inflammatory pathways, including NF-κB signalling(Reference Weaver, Ivester and Seeds50, Reference Bouwens, van de Rest and Dellschaft73, Reference Farzaneh-Far, Harris and Garg74).
It has been reported that different strains of mice may have differential susceptibility to, and pathogenesis of, DSS-induced colitis and colon cancer(Reference Mähler, Bristol and Leiter75–Reference Melgar, Karlsson and Michaëlsson77). DSS-induced chronic colitis in C57BL/6 mice is believed to be a robust model for validating future therapies for the treatment of IBD because of the similarities of the pathology compared with symptoms in human disease(Reference Melgar, Karlsson and Michaëlsson77). Thus, although different results might be obtained with another mouse strain, we believe that our findings make an important contribution to the consideration of using FO and curcumin as therapeutic agents in chronic IBD. We propose that n-3 PUFA and curcumin may be best suited to maintain IBD remission(Reference Uchiyama, Nakamura and Odahara78).
In conclusion, our data show that dietary FO and curcumin differentially modulate the pathology of DSS-induced chronic colitis in mice. Feeding FO-cur together enhanced the resolution of chronic inflammation but disrupted mucosa repair during the acute phase of DSS exposure in part due to the inhibition of NF-κB activity in the colonic mucosa. FO and curcumin also differentially modulated mucosa cytokinetics, which may be attributed to the regulation of the TLR4/COX-2/PGE2 and Stat3 signalling pathways in the colon. Collectively, these findings contribute to a better understanding of the ability of n-3 PUFA and curcumin to modulate the inflammation–mucosa repair–carcinogenesis axis in the colon.
Acknowledgements
The present study was financially supported by NIH grants DK071707, CA59034, CA129444 and USDA 2008-34402-19195 Vegetable & Fruit Improvement Center. Curcumin C3 complex was provided by Sabinsa Corporation. Q. J. performed the experiments, data collection, analysis and interpretation of the data. I. I. and Z. Z. Z. performed the microarray data analysis. R. C. A., J. R. L., D. N. M. and R. S. C. assisted with the study design and data interpretation. B. R. W. scored the histology sections. E. S. C., J. S. G., L. A. D. and Y.-Y. F. contributed to the sample collection/reagents/materials and the development of analytical tools. L. Z. supervised the statistical analyses. None of the authors has a conflict of interest.







