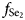Gachingite, Au(Te1–xSex), 0.2 ≈ x ≤ 0.5, is a new mineral discovered in the Gaching ore occurrence of the Maletoyvayam epithermal deposit, Kamchatka, Russia. Gachingite forms individual droplet-like grains of sizes from 2 to 10 μm included in native gold (Au–Ag), associated with calaverite, maletoyvayamite, watanabeite and Au–Sb oxides. The aggregates do not exceed 100 μm in diameter. In plane-polarised light, gachingite is grey with a bluish tint, has bireflectance (bluish-grey to deep grey), and strong anisotropy with rotation tints blue to dark blue to brown. Reflectance values for gachingite in air (Rmin, Rmax in %) are: 39.9, 40.3 at 470 nm; 41.6, 43.3 at 546 nm; 42.0, 43.7 at 589 nm; and 43.0, 44.0 at 650 nm. Eighteen electron-microprobe analyses of gachingite gave an average composition: Au 62.40, Ag 0.57, Se 9.78, Te 27.33 and S 0.01, total 100.09 wt.%, corresponding to the formula (Au0.96Ag0.02)Σ0.98(Te0.65Se0.37)Σ1.02 based on 2 apfu, the simplified formula is Au(Te0.65Se0.35); the average analyses of its synthetic analogue is Au 65.7, Se 13.1 and Te 21.1, total 99.9 wt.%, corresponding to Au1.00(Te0.50Se0.50). The calculated density is 10.47 g/cm3. The mineral is orthorhombic, space group Cmce (#64) with a = 7.5379 Å, b = 5.7415 Å, c = 8.8985 Å, V = 385.12 Å3 and Z = 8. The crystal structure was solved and refined from the single-crystal X-ray-diffraction data of synthetic Au1.00(Te0.50Se0.50). The crystal structure of gachingite represents a unique structure type, containing linear [Au–Au–Au] chains running along the b-axis indicating strong metallic interaction in one direction. The structural identity of gachingite and its synthetic analogue Au1.00(Te0.50Se0.50) was confirmed by electron back-scatter diffraction and Raman spectroscopy. The formation of gachingite requires an abundant source of Au and Se and a high oxidising environment. Gachingite is related to the gold-bearing productive stage of ore mineralisation, which is stable at 250°C in log$f_{{\rm S}{\rm e}_ 2}$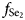 range of −12.4 and −5.7. The mineral is named after its type locality.
range of −12.4 and −5.7. The mineral is named after its type locality.