Cytomegalovirus (CMV) is transmitted from mother to foetus in approximately 30–39% of pregnancies in which a maternal primary infection occurs.Reference Kenneson and Cannon 1 Transplacental transmission rates are lower, approximately 20%, with infection in the first trimester and increase with advancing gestational age to approximately 75% with third trimester infection.Reference Leruez-Ville, Sellier, Salomon, Stirnemann, Jacquemard and Ville 2 Congenital CMV infection is a leading cause of sensorineural hearing loss, and it is an important cause of developmental disability, cognitive impairment, cerebral palsy and impaired vision. The foetus is most likely to suffer permanent damage in the case of primary maternal infection.Reference Cannon, Schmid and Hyde 3 , Reference Fowler, Stagno, Pass, Britt, Boll and Alford 4 The disease burden from congenital CMV infection is greater than that of Down's syndrome and spina bifida combined; it has been estimated that approximately 6000 children born in the USA each year will have central nervous system, auditory, visual or motor disability because of congenital CMV infection.Reference Cannon and Davis 5 It is currently believed that transplacental transfer of CMV is a local phenomenon following viraemia. The ability of viruses to spread from the infected mother to the foetus arises from the architecture of the placenta, which anchors the foetus to the uterus. The notion that stress and stress-related disorders can interfere with the structure and the placental function is biologically plausible.Reference Brunton 6 Increasing evidence indicates that maternal physiological and pathological stress conditions may influence placental secretion of neurohormones, suggesting that the placenta takes an active role in response to stress-mediated adverse conditions.Reference Maccari, Krugers, Morley-Fletcher, Szyf and Brunton 7 Indeed, psychosocial adversity and stress-related symptoms during pregnancy have been previously associated with increased placenta weight, evidence of bacterial vaginosis, preterm birth and increased risk of placental abruption.Reference Tegethoff, Greene, Olsen, Meyer and Meinlschmidt 8 – Reference de Paz, Sanchez, Huaman, Chang, Pacora and Garcia 11 We challenged the hypothesis that psychological factors may also play a role in the mother-to-child transmission (MTCT) of infections. We aimed at evaluating the role of mental illness symptoms and personality traits in CMV MTCT, in a cohort of pregnant women with primary CMV infection enrolled in a prospective study (ClinicalTrials.gov NCT01659684) designed to assess the safety and efficacy of immunoglobulin (IVIG) therapy.Reference Polilli, Parruti, D'Arcangelo, Tracanna, Clerico and Savini 12 The study protocol was approved by the local ethical committee (Comitato etico per la Sperimentazione Clinica dei Farmaci ASL di Pescara).
Method
All eligible women with confirmed primary CMV infection during pregnancy, referring to the Infectious Disease Unit of Pescara General Hospital from 2010, were prospectively evaluated before the start of IVIG infusions for demographic, clinical – virological findings and psychological factors. Data from all screened patients were prospectively collected through an electronic database system. We collected age, gestational age, IgM and IgG CMV titres after and before each infusion, IgG CMV avidity index after and before each infusion, CMV viral load in the blood and in the urine performed at each time point of follow-up, number of infusions, adverse events, CMV DNA in amniotic fluid if available and clinical data of newborns: presence of CMV infection and clinical appearance of CMV sequelae. Among the psychological factors we included: (a) reactive psychopathological symptoms, such as current depressive symptoms and ongoing symptoms of post-traumatic stress disorder (PTSD), that is, the subjective stress caused by a traumatic event (stressful life events such as a high-risk pregnancy); (b) stable personality traits, such as alexithymia, which reflects the difficulty in the appreciation of one's own emotions, such as identifying, describing and analysing emotions, and Type D (distressed) personality, consisting of two stable personality traits: negative affectivity (NA) and social inhibition (SI). NA is the tendency of an individual to experience negative emotions across time and in various situations, while SI refers to the tendency to feel discomfort in social interactions, to exhibit a lack of social poise and to avoid confrontation.
Depressive symptoms were evaluated with Beck Depression Inventory-II (BDI-II score ≥15),Reference Castro, Couto, Martins Brancaglion, Nogueira Cardoso, Bergo Protzner and Duarte Garcia 13 the distress personality (Type D) with Italian validatedReference Gremigni and Sommaruga 14 DS-14 (NA score ≥9; SI score ≥9, as opposed to the originalReference Denollet 15 DS-14 cut-off of 10), alexithymic trait with Toronto Alexithymia Scale (TAS-20 score ≥50, includes borderline alexithymia + high alexithymia, a threshold previously employed in large population-based studiesReference Grabe, Schwahn, Barnow, Spitzer, John and Freyberger 16 and in agreement with the Italian TAS-20 user manual)Reference Caretti and La Barbera 17 and the post-traumatic stress symptoms (PTSS) with Impact of Event Scale-Revised (IES-R score ≥34).Reference Morina, Ehring and Priebe 18 Primary infection was defined by positive CMV IgM antibodies with absent or low titres of CMV IgG antibodies and low (<50%) CMV IgG avidity indexes, as previously reported.Reference Polilli, Parruti, D'Arcangelo, Tracanna, Clerico and Savini 12 Quantitative CMV DNA was amplified from whole-blood and urine samples using the real-time PCR (CMV ELITe MGB® Kit, ELITechGroup, Italy). Neonatal infection was diagnosed by CMV DNA amplification from blood and/or urine and saliva from newborn at birth. Variables that were significantly associated at univariate analysis were described as frequency (%) or mean (s.d.). They were included in the logistic regression equation to provide the adjusted estimates (odds ratio, OR) and corresponding 95% confidence interval (CI).
Results
At the time of data analysis, 306 pregnant women had been treated, and 280 of these had accepted to undergo psychological assessment and signed informed consent. Among them, 276 women had given birth while the remaining four were still pregnant. One hundred and eight (39.1%) newborns had CMV congenital infection. Compared with mothers of uninfected newborns, mothers of children with congenital infection exhibited higher gestational age at CMV diagnosis (16.9(s.d.=7.5) weeks v. 15.0(s.d.=6.5) weeks; P=0.029), higher gestational age at the start of IVIG therapy (23.2(s.d.=6.8) weeks v. 20.1(s.d.=6.4) weeks; P=0.0002), increased urinary CMV DNA level after the first infusion (1654 c/mL, IQR 237–6825 v. 528 c/mL, IQR 78–2451, P=0.004), higher frequency of alexithymic trait (TAS-20 ≥50: 21.8% v. 11.0%; P=0.02) and higher incidence of PTSS (IES-R ≥ 34: 30.2% v. 15.0%; P=0.0005), as reflected by higher IES-R total score evaluated both as a whole and as single subscales (Fig. 1). Moreover, congenital infection occurred more frequently in newborns of mothers who underwent amniocentesis during pregnancy (50% v. 25.6%; P<0.0001) and who received more doses of IVIG infusions (mean 2.8(s.d.=1.2) v. 2.4(s.d.=1.0), P=0.002).
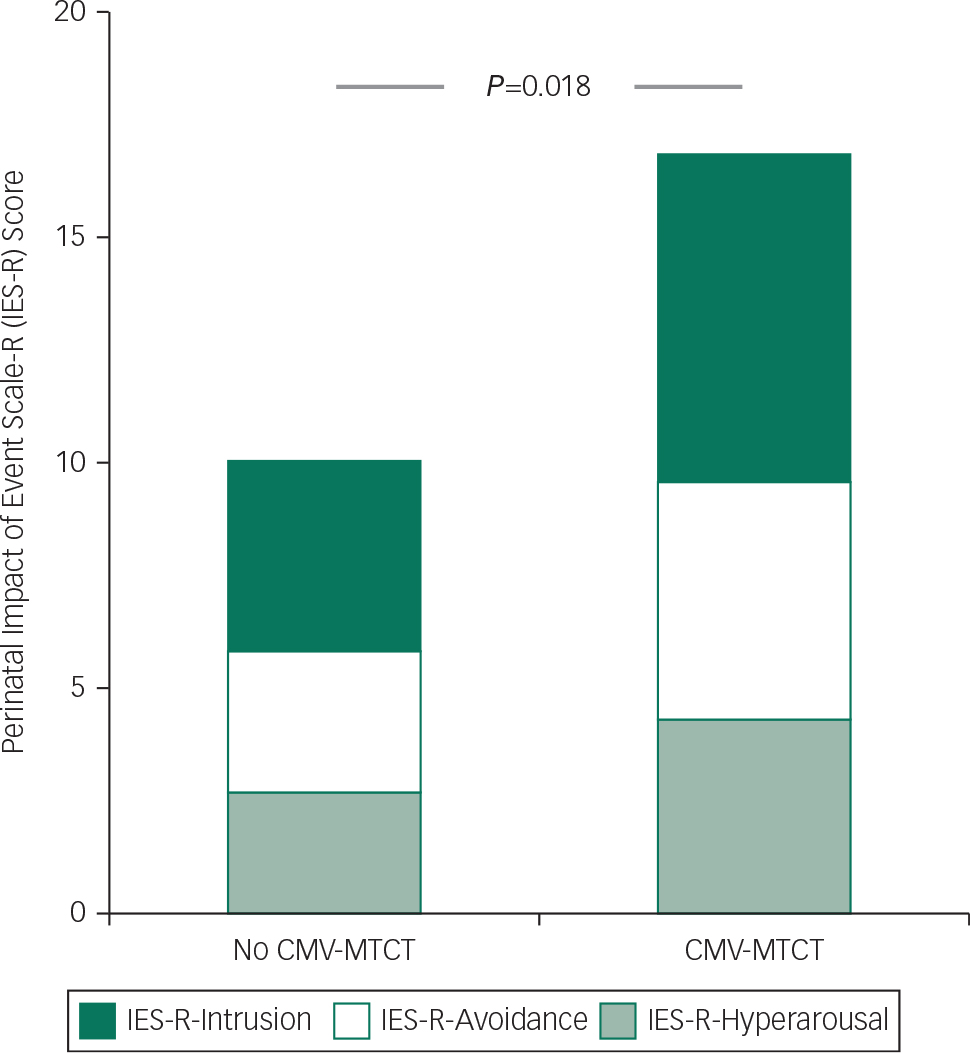
Fig. 1 Relationship between perinatal post-traumatic stress symptom (PTSS) and cytomegalovirus (CMV) congenital infection in newborns. Impact of Event Scale-Revised (IES-R) total score (whole column) and its components (IES-R-Intrusion, IES-R-Avoidance, IES-R-Hyperarousal) in women with and without mother-to-child transmission (MTCT) of CMV infection during pregnancy (P=0.018, P=0.014, P=0.020, P=0.039, for IES-R total score, intrusion score, avoidance score, hyperarousal score, respectively).
No association with congenital infection was detected with depressive symptoms (BDI-II ≥ 15) and distress personality evaluated during pregnancy.
At the multivariate analysis, PTSS (OR=2.31; 95% CI 1.10–4.85), amniocentesis (OR=3.55; 95% CI 1.79–7.02), IVIG doses (OR by 1 dose=1.60; 95% CI: 1.05–2.44), gestational age at the start of IVIG therapy (OR=1.13; 95% CI 1.04–1.24) and level of CMV DNA in the urine after the first infusion (trend by quartiles: OR=1.34; 95% CI 1.01–1.79) were independent predictors of occurrence of congenital disease (Table 1).
Table 1 Results from the logistic regression models predicting congenital cytomegalovirus (CMV) infection
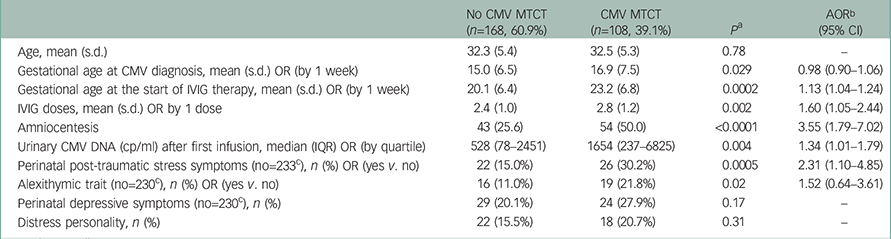
| No CMV MTCT (n=168, 60.9%) | CMV MTCT (n=108, 39.1%) | P a | AOR b (95% CI) | |
|---|---|---|---|---|
| Age, mean (s.d.) | 32.3 (5.4) | 32.5 (5.3) | 0.78 | – |
| Gestational age at CMV diagnosis, mean (s.d.) OR (by 1 week) | 15.0 (6.5) | 16.9 (7.5) | 0.029 | 0.98 (0.90–1.06) |
| Gestational age at the start of IVIG therapy, mean (s.d.) OR (by 1 week) | 20.1 (6.4) | 23.2 (6.8) | 0.0002 | 1.13 (1.04–1.24) |
| IVIG doses, mean (s.d.) OR by 1 dose | 2.4 (1.0) | 2.8 (1.2) | 0.002 | 1.60 (1.05–2.44) |
| Amniocentesis | 43 (25.6) | 54 (50.0) | <0.0001 | 3.55 (1.79–7.02) |
| Urinary CMV DNA (cp/ml) after first infusion, median (IQR) OR (by quartile) | 528 (78–2451) | 1654 (237–6825) | 0.004 | 1.34 (1.01–1.79) |
| Perinatal post-traumatic stress symptoms (no=233 c ), n (%) OR (yes v. no) | 22 (15.0%) | 26 (30.2%) | 0.0005 | 2.31 (1.10–4.85) |
| Alexithymic trait (no=230 c ), n (%) OR (yes v. no) | 16 (11.0%) | 19 (21.8%) | 0.02 | 1.52 (0.64–3.61) |
| Perinatal depressive symptoms (no=230 c ), n (%) | 29 (20.1%) | 24 (27.9%) | 0.17 | – |
| Distress personality, n (%) | 22 (15.5%) | 18 (20.7%) | 0.31 | – |
IQR=interquartile range.
a Analysis of variance for normally distributed variables, Mann–Whitney U test for not normally distributed variables, chi-square test for categorical variables.
b Adjusted odds ratio: Logistic regression equation includes urinary CMV DNA, PTSS, alexithymic trait, amniocentesis, gestational age at CMV infection diagnosis and gestational age at the start of IVIG therapy.
c The adherence to psychological evaluations was not complete: 43 women (15.6%) refused to undergo IES-R, 44 (15.4%) TAS-20, 46 (16.7%) BDI and 47 (17.0%) DS-14. Thirty-nine (14.1%) refused all evaluations. These women (n=39) were not different from those who underwent psychological evaluation in terms of age, week of seroconversion, week of first infusion, urinary DNA, amniocentesis frequency, number of infusions and newborn CMV infection.
Determination of viral load in amniotic fluid was available only for 92 women. Of those with the information, amniotic fluid viral DNA was retrieved in 45 (49.2%), and 43 (95.6%) gave birth to CMV-infected children. The corresponding figure for 47 women with viral DNA-negative amniotic fluid was 8 (17.0%, P<0.0001). Including in the multivariate logistic equation a summary variable (0=analysis not performed, 1=detectable DNA, 2=undetectable DNA), we found that having detectable DNA in amniotic fluid was associated with a 60-fold higher risk of CMV MTCT (95% CI 11.0–331.0), whereas the estimate for perinatal PTSS did not markedly change (OR=2.55, 95% CI 1.14–5.72). Women with amniocentesis that were not tested for DNA in amniotic fluid had an intermediate OR (1.6), not significantly different from those with undetectable viral DNA.
Discussion
The main finding of our study is the observation that the occurrence of congenital CMV disease in newborns is independently predicted by PTSS in mothers. To the best of our knowledge, this is the first study examining the association of stress-related symptoms during pregnancy with MTCT of CMV. CMV is the most common congenital infection and can follow a primary or recurrent maternal infection. MTCT of CMV occurs transplacentally (congenital infection), during birth, or by ascending route from the birth channel, or through breast milk, although the latter two modes of transmission are not associated with the central nervous system sequelae that occur with congenital infection. The mechanisms involved in the transmission of CMV in utero remain poorly understood. It is currently believed that transplacental transfer of CMV is a local phenomenon following viraemia. In this regard, CMV-placental changes, including an enhanced placental thickness related to inflammatory stimuli, reflecting placental inflammation and insufficiency, have been hypothesised as a major risk factor for CMV MTCT.Reference La Torre, Nigro, Mazzocco, Best and Adler 19 , Reference Hamilton, Scott, Naing, Iwasenko, Hall, Graf and Arbuckle 20 Psychosocial stress alters cytokine production across pregnancy and has been associated with placental abnormalities such as increased placental weight.Reference Tegethoff, Greene, Olsen, Meyer and Meinlschmidt 8 Increased levels of peripheral markers of inflammation have frequently been observed in patients with PTSD, partly because stress hormone dysregulation related to PTSD may lead to alterations in the immune system and inflammatory signaling.Reference Baker, Nievergelt and O'Connor 21 – Reference Eraly, Nievergelt, Maihofer, Barkauskas, Biswas and Agorastos 23 It is thus conceivable that psychological stress-related disturbances, through neuroendocrine mechanisms, may weaken the physical and immunological barrier against the haematogenous transmission of viruses from mother to foetus. Therefore, we here hypothesise that CMV infection and PTSD symptoms might concur to foster an inflammatory milieu favouring, in turn, placental abnormalities that increase the likelihood of transplacental viral transmission.
Additional independent predictors of congenital infection included having performed amniocentesis and the number of IVIF doses. Most women (88/97, 90.7%) underwent amniocentesis after seroconversion, as the procedure was mainly performed to measure viral DNA in the amniotic fluid. The major reason for performing or not performing amniocentesis was willingness of pregnant women, who were all invited.
Several seminal experiences and early experimental models documented that repeated infusions of immunoglobulins, either with hyperimmune or standard preparations, may help to quench foetal disease upon transmission and possibly to reduce maternal–foetal CMV transmission.Reference Nigro, Adler, Parruti, Anceschi, Coclite and Pezone 24 , Reference Parruti, Polilli, Ursini and Tontodonati 25
The counterintuitive finding here that a higher number of IVIG infusions is associated with an increased risk of congenital infection is likely due to the bias that this variable depends on both gestational age at diagnosis of infection and gestational age at start of IVIG therapy, as well as on the response to the first and subsequent infusions. Women with worse response to treatment and/or with proven foetal infection at amniocentesis were routinely invited to receive monthly infusions until deliveryReference Polilli, Parruti, D'Arcangelo, Tracanna, Clerico and Savini 12 , Reference Parruti, Polilli, Ursini and Tontodonati 25 and are therefore more likely to have received more IVIG infusions.
Strengths of our study included the large sample size and the ample geographical representation of our cohort of pregnant women, who were referred to our centre from all over Italy, and, in some cases, from abroad. This allows generalisability of our present results. Conversely, incomplete adherence to psychological evaluation may be a limitation of this study. However, women who refused to undergo complete psychological evaluation (n=39) were not different from responders for any of the clinical characteristics. Thus, it is unlikely that this type of selection bias may have affected our findings.
In summary, our results, despite needing confirmation, may suggest that the risk of MTCT of CMV is increased in pregnant women with high levels of PTSD symptoms. If these results are confirmed in different and larger cohorts, public health efforts should be undertaken to assess mental disturbances in women affected by primary CMV infection, in order to provide timely interventions limiting the biological impact of traumatic stress on the maternal–foetal dyad.
Acknowledgements
The authors thank all the nurses of the Infectious Disease Unit, Teresa Moschino and Angela Pisciella in particular, for their assistance with our pregnant patients during infusions. The authors also thank Antonina Sciacca for her support with our patients. Alessia Memmo and Elena Di Vera performed control echography whenever necessary, and Armando Tartaro and Claudio Celentano supported the management of patients needing magnetic resonance imaging or cordocentesis.
Funding
E.P. and E.T. were funded by an educational grant from the Fondazione Camillo de Lellis per l'Innovazione e la Ricerca in Medicina, Pescara, Italy.






eLetters
No eLetters have been published for this article.