Introduction
Infants with single-ventricle physiology (∼15% of all CHD) face a significant challenge in terms of growth, both short and long term, particularly after the first palliative surgery during the interstage. Reference Anderson, Iyer and Schidlow1–Reference Vogt, Manlhiot, Van Arsdell, Russell, Mital and McCrindle4 Growth impairment is well documented in this population and is associated with adverse outcomes, both developmental and surgical. Reference Williams, Zak and Ravishankar5 Inadequate growth in this population is multifactorial and may be due to a combination of factors including inadequate calorie intake, high metabolic demands, gastrointestinal pathology, and genetic and extracardiac abnormalities. Reference Gingell, Hornung and Lebenthal6,Reference Hansen and Dorup7 Optimisation of growth with aggressive nutritional support may result in improved outcomes.
The use of feeding protocols in infants with CHD has been shown to positively impact feeding and growth outcomes both pre-operatively and post-operatively. Reference Venna, Reid and Davis8–Reference Newcombe and Fry-Bbowers13 In 2013, the Feeding Work Group (FWG) within the National Pediatric Cardiology Quality Improvement Collaborative (NPC-QIC) conducted a literature review along with expert opinion of centres within the collaborative to develop best practice guidelines around feeding infants with hypoplastic left heart syndrome. Reference Slicker, Hehir and Horsley14 The consensus statement suggested to begin fortification once targeted volume was achieved. Reference Slicker, Hehir and Horsley14 In a recent review, Salvatori and colleagues proposed a decisional algorithm for clinicians to guide nutrition and growth optimisation during the pre-operative and post-operative phases but does not provide protocolised steps for advancement of feeds. Reference Salvatori, De Rose and Massolo15 Further research has identified continued variation in feeding practices both perioperatively and following stage 2 palliation within this population. Reference Lambert, Pike and Medoff-Cooper16–Reference Hoch, Fatusin, Yenokyan and Thompson18 Slicker and colleagues surveyed 46 centres within the collaborative and reported a utilisation of < 10% of the previously published guidelines. Reference Slicker, Sables-Baus and Lambert17 Therefore, further development of standardisation around feeding practices remains warranted within this population.
The benefits of human milk use in infants with CHD along with extremely premature infants have been widely recognised and recommended to improve clinical outcomes. Reference Venna, Reid and Davis8,Reference Slicker, Hehir and Horsley14,Reference Sullivan, Schanler and Kim19–Reference Blanco, Hair and Justice22 In very low birth weight premature infants, an EHM diet has been shown to result in significantly lower rates of necrotising enterocolitis and decreased incidence of sepsis. Reference Sullivan, Schanler and Kim19,Reference Fleig, Hagan and Lee23 Many paediatric cardiac programmes have limited experience with utilisation of an EHM diet in term infants with single-ventricle physiology as the use of human milk-based fortifiers is often restricted to preterm infants; therefore, the cardiac programmes rely on bovine milk-based products for fortification.
A recent multi-centre randomised controlled trial showed that infants with single-ventricle physiology following stage 1 surgical palliation who received an exclusive human milk diet with early fortification had improved short-term growth and decreased risk of necrotising enterocolitis. Reference Blanco, Hair and Justice22 A criticism of the randomised controlled trial was a difference in the feed advancement protocol between the two groups where the formula fed patients progressed slower due to investigator concerns for tolerance and necrotising enterocolitis. The purpose of this study was to examine growth in a group of single-ventricle CHD patients whose feeding was not dictated by a study feeding algorithm compared to patients enrolled in the randomised controlled trial (both the TCG and EHM groups). We sought to assess the impact of protocolised feeding and an exclusive human milk diet on growth and feeding tolerance.
Methods
Study design
We conducted a retrospective cohort study to evaluate growth velocity and clinical outcomes in infants with single-ventricle physiology fed with cow’s milk formula (either primarily or as fortification added to maternal breast milk) using clinician driven fortification as per standard of care (the retrospective control group; RCG) compared to those fed a protocolised exclusive human milk diet (EHM) or cow milk-based formula in a previously published randomised controlled trial, of which Cincinnati Children’s Hospital Medical Center (CCHMC) was a participating site. Details regarding the feeding advancement and the timing of fortification between each group (TCG vs. EHM vs. RCG) are summarised in Table 1. The study was approved by the Institutional Review Board at CCHMC.
Table 1. Feeding advancement and fortification for TCG versus EHM versus RCG

TCG = trial control group; EHM = exclusive human milk; RCG = retrospective control group.
Participants
RCG patients (e.g., controls) were identified by searching EPIC for patients who underwent surgical palliation for single-ventricle physiology (Norwood procedure, BT shunt, PA band) and received clinical care at CCHMC between January 2016 and December 2020.
Term infants (≥ 37 and 0/7 weeks gestational age at birth) 7 days old with a diagnosis of single-ventricle physiology who required surgical palliation within the first 1 month of life were included in the study. Infants were excluded from the study if they were <37 weeks gestation, required cardiopulmonary resuscitation or extracorporeal membrane oxygenation prior to surgical repair, or if they had any of the following: major congenital abnormalities that could significantly affect survival (such as confirmed or suspected major genetic abnormalities (lethal or with extremely low probability for survival), major organ system abnormalities not related to a genetic syndrome that are lethal or have extremely low probability for survival, heterotaxy syndrome, metabolic disorders affecting growth, evidence of intracerebral haemorrhage and/or intraventricular haemorrhage ≥ Grade 3, or any comorbidity or significant clinical event prior to enrolment that was deemed by the investigator as likely to affect survival.
“Protocolised” study patients (trial controls and trial EHM) were enrolled during the same study period. Inclusion and exclusion criteria and variables of interest were similar to study patients. Details have been previously published. Reference Blanco, Hair and Justice22
Outcomes
The primary objective was to evaluate growth velocity (weight velocity [g/day]) from birth to 30 days after the first post-operative feed (or hospital discharge, whichever came first) for infants with single-ventricle physiology who were fed according to CCHMC standard of care (non-protocolised) compared to the protocolised TCG and those fed an EHM diet with early fortification in the randomised controlled trial.
The secondary objectives were to evaluate weight z-scores from birth to 30 days after the first post-operative feed (or hospital discharge, whichever came first), the rate of linear growth (cm/week and z-score from WHO growth charts), and incremental rate of head circumference growth (cm/week and z-score from WHO growth charts) over the duration of the initial 30-day feeding period after surgery or discharge, whichever came first.
Feeding intolerance was also evaluated and defined as nothing by mouth (NPO) for at least 24 hours in the 30-day feeding period (day 1 is the first day of feeding post-op) not related to procedures. Feeding intolerance was further classified based on intestinal versus non-intestinal causes. Intolerance was classified as being due to intestinal disease if the patient had abdominal clinical signs documented (such as abdominal distension or discoloration), radiographic evidence of intestinal disease, bloody stools, or progress notes specifying necrotising enterocolitis concerns. Intolerance was classified as being due to non-intestinal disease if the patient became NPO due to acute cardiorespiratory decompensation or related events (such as bradycardic episodes, hypoxic episodes, pulmonary oedema, respiratory distress, myocardial infarction, ischemia, and code event), cannulation for extracorporeal membrane oxygenation, other unrelated diseases (such as seizures or sepsis), or if they were placed NPO >24 hours and <72 hours and was resumed on full fortified feeds within this period.
Additional outcomes evaluated include post-operative length of stay in the hospital and in the ICU/cardiac unit, days of parenteral nutrition, and incidence of key morbidities in the 30days post-surgical period, such as necrotising enterocolitis (defined as stage 2 or greater per Bell’s criteria) and suspected necrotising enterocolitis, confirmed sepsis, wound infections, and/or wound dehiscence.
Statistical methods
Categorical variables were summarised using frequency and percentages and compared between groups using Chi-square/Fisher’s exact test with a Bonferroni correction for pairwise comparisons. Continuous variables were summarised using mean ± standard deviation and/or median/interquartile range and compared using ANOVA with Bonferroni or Kruskal–Wallis tests with the Dwass-Steel-Critchlow-Fligner (DSCF) correction. Length of stay outcomes, time from surgery to the end of study, and total parenteral nutrition days were analysed using Kaplan–Meier analysis with the log-rank test and adjusted for multiple comparisons using a Bonferroni correction. Growth velocity was compared between groups using multivariable linear regression adjusting for baseline factors (small for gestational age, hypoplastic left heart syndrome, and type of surgical repair). In all analyses, (p < 0.05) was considered significant.
Results
We identified 45 retrospective control patients who underwent cardiac surgical palliation. Demographics and growth characteristics are summarised in Tables 2 and 3, respectively. The retrospective patients were similar in gestational age, gender, race, birth size, method of delivery, and pre-operative support. The RCG consisted of a statistically significant lower proportion of patients with hypoplastic left heart syndrome compared to both the TCG and EHM group (51% vs. 77% vs. 84%, respectively, p = 0.0009). Growth velocity in g/day was similar for the RCG compared to the TCG (7.5 g/day [IQR 2.3–13.9 g/day] vs. 8.2 g/day [IQR 0–13.8 g/day]), but the RCG had significantly lower growth than the EHM group (12 g/day [IQR 4.7–18.4], p = 0.05). The RCG patients had higher head circumference growth but lower growth in length velocity and significantly lower length z-score at discharge. Although not statistically significant, the EHM group demonstrated superior length velocity by the end of study among all groups.
Table 2. Summary of demographic characteristics

TCG = trial control group, EHM = exclusive human milk, RCG = retrospective control group; SGA = small for gestational age, LGA = large for gestational age, HLHS = hypoplastic left heart syndrome, RV = right ventricle, AVSD = atrioventricular septal defect, DORV = double-outlet right ventricle, LV = left ventricle, BTT = Blalock–Taussig Thomas, MPA = main pulmonary artery, DHCA = deep hypothermic circulatory arrest.
Table 3. Summary of growth characteristics
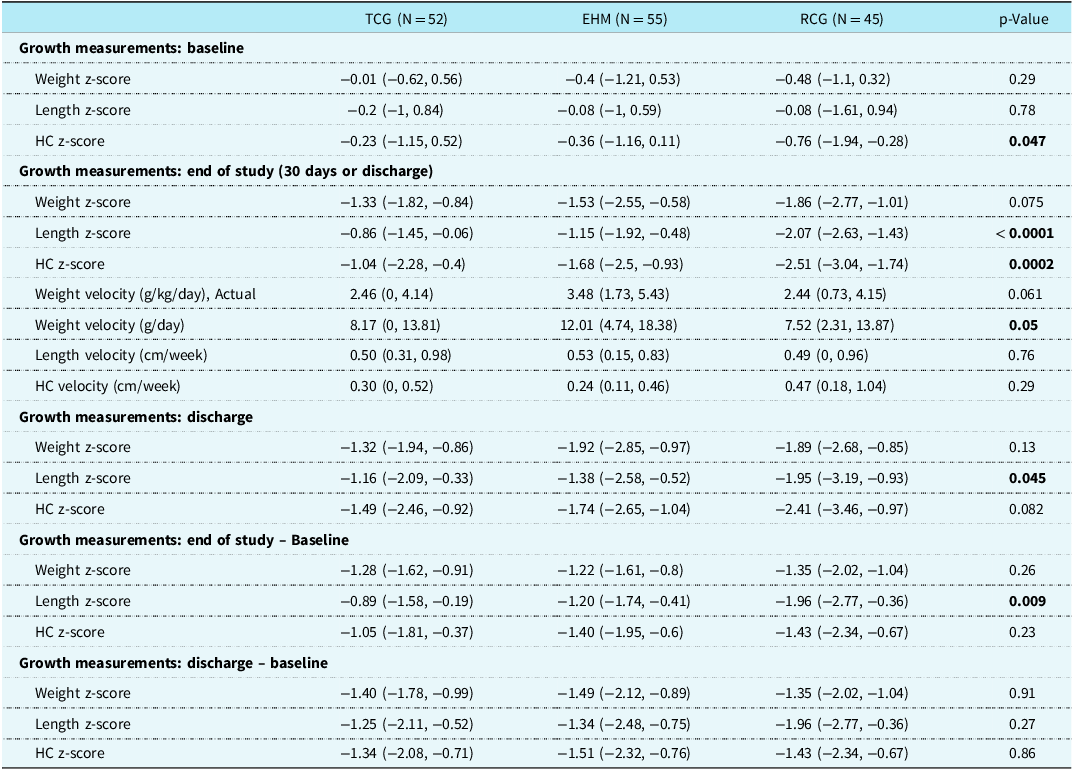
TCG = trial control group, EHM = exclusive human milk, RCG = retrospective control group; HC = head circumference.
RCG participants started enteral feeds on average by post-operative day 4 (range of 1–14). The feeding modalities consisted of oral, nasogastric, or transpyloric tube or a combination of these. Most patients (69% or 31/45) were fed by a combination of oral and tube (nasogastric or transpyloric) throughout their post-operative course. Approximately 29% of patients were fed only by a tube (either nasogastric or transpyloric) and only one patient fed exclusively by mouth within the 30 days after surgery. Eighty per cent (36/45) of patients were fed a combination of expressed breastmilk and formula. Only one patient received all expressed breastmilk and two patients received standard formula with the remaining six patients on all formula (either partially hydrolysed or elemental) during the post-operative period. The maximum concentration prescribed was 28 calorie/oz with a mean concentration among all patients of 24 calorie/oz.
Secondary outcomes are shown in Table 4. Total parenteral nutrition days, ICU length of stay and hospital length of stay were all significantly shorter in the RCG compared to both the TCG and the EHM group. Necrotising enterocolitis and suspected necrotising enterocolitis were similar in the RCG versus the TCG but significantly higher in the RCG compared to the EHM group (20.5% vs. 3.6%, p = 0.033). At the time of necrotising enterocolitis diagnosis, patients in the RCG were mostly fed by nasogastric tube. All three were on different types of feeds. One patient was receiving fortified human milk with a partially hydrolysed fortifier. Another patient was on a partially hydrolysed formula, and the remaining patient was on a term formula specifically intended to treat chylothorax. Two of the three patients were at 24 calories concentration at the time of developing necrotising enterocolitis, which is the same as the trial control group in which two patients who developed necrotising enterocolitis were on 24 calories concentration and one patient was on 20 calories concentration. All patients were greater than 2 weeks post-operative with a mean onset of necrotising enterocolitis at 2.8 weeks. All patients diagnosed with necrotising enterocolitis were less than 3 kg in size (Table 5).
Table 4. Safety and other event data
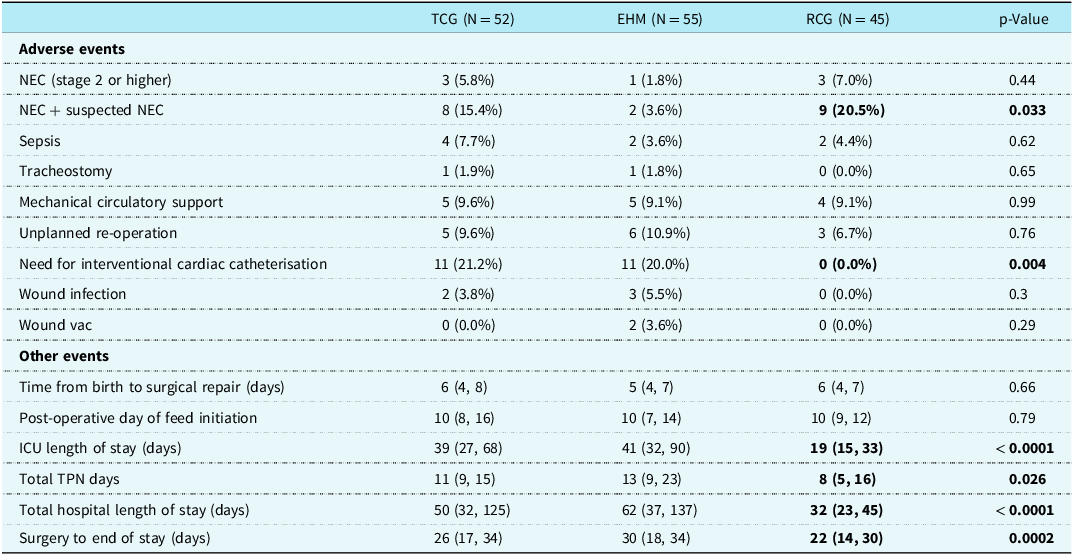
TCG = trial control group, EHM = exclusive human milk, RCG = retrospective control group, NEC = necrotising enterocolitis, ICU = intensive care unit, TPN = total parenteral nutrition.
Table 5. Details regarding RCG patients with NEC

NEC = necrotising enterocolitis, POD = post-operative day, mL = milliliters, kcal = kilocalories, kg = kilograms.
Feeding intolerance developed in 40% (n = 18) of patients in the RCG compared to 27% (n = 14) of TCG patients and 31% (n = 17) of patients in the trial EHM group. There were 23 episodes of feeding intolerance (intestinal vs. non-intestinal) among the 18 patients. Of those episodes, 8/23 were intestinal signs of intolerance which included abdominal clinical signs (emesis, distention, and residuals), blood in stool, and radiographic evidence (bowel dilation or pneumatosis or perforation). The remaining 15 episodes of intolerance were due to acute cardiorespiratory decompensation (Table 6).
Table 6. Details regarding feeding intolerance episodes for RCG patients

cal = calories, oz = ounce, NG = nasogastric, EBM = expressed breastmilk, TP = transpyloric, PO = by mouth.
Discussion
The use of an EHM diet for our most vulnerable critical infants has been demonstrated to be most protective from necrotising enterocolitis or suspected necrotising enterocolitis while promoting optimisation of nutritional intake. In this study, patients in the RCG grew similar to the patients in the TCG, but both groups exhibited inferior growth compared to the EHM group of the randomised controlled trial. These growth findings are comparable to the growth outcomes of EHM diets published in the very premature literature. Sullivan and colleagues conducted a randomised trial in infants weighing 500–1250 g at birth looking at the benefits of an EHM diet compared with a diet of both human milk and bovine milk-based products and found significantly lower rates of necrotising enterocolitis in those infants receiving an EHM diet. Reference Sullivan, Schanler and Kim19 Furthermore, an article by Fleig and colleagues looked at the impact of an EHM diet in small for gestational age preterm infants on growth outcomes and neonatal morbidities from birth to discharge. Reference Fleig, Hagan and Lee23 They noted similar growth compared to cow’s milk diet but overall decreased incidence of necrotising enterocolitis and sepsis. Reference Fleig, Hagan and Lee23 Although growth was not statistically significant in these studies, there was a positive trend in growth parameters of those fed an EHM diet along with improved neonatal morbidities.
Interestingly, the TCG patients did not demonstrate superior growth despite being on a feeding protocol compared to the RCG patients who were not on a feeding protocol. This finding is discrepant with multiple studies that have shown that the use of feeding protocols positively impacts feeding and growth outcomes. Reference Venna, Reid and Davis8–Reference Newcombe and Fry-Bbowers13 We postulated that despite the lack of a feeding protocol in our retrospective control patients, we had dedicated dietitian support on rounds every day, which provided our RCG patients with consistency, or essentially a pseudo-protocol based on their recommendations for nutritional management.
This study showed that our RCG patients received a shorter duration of total parenteral nutrition compared to both trial groups. For patients to have fewer total parenteral nutrition days, they would either have their feeds initiated sooner or advanced more rapidly or have fewer feeding complications requiring NPO (such as necrotising enterocolitis). However, given that the RCG had a significantly higher occurrence of necrotising enterocolitis, and all three groups had feeds started on median post-operative day 10, it can be inferred that the RCG patients had fewer total parenteral nutrition days because in the absence of a feeding protocol the feeding volume advancement likely occurred faster. However, this more aggressive feeding approach occurred at the expense of significantly more necrotising enterocolitis. Surprisingly, despite receiving a high-calorie concentration, infants enrolled in the trial EHM group had a significantly lower risk of necrotising enterocolitis compared to the RCG (37/55 infants in EHM group received >26 calorie/oz, max of 30 calorie/oz concentration compared to 2/45 with a maximum of 28 calorie/oz in the RCG). Furthermore, one may have predicted infants in the EHM group would have higher risk of necrotising enterocolitis due to larger proportion of hypoplastic left heart syndrome diagnosis; however, they received higher caloric concentration enterally and had a lower incidence of necrotising enterocolitis and better growth.
Interestingly, there is a disconnect between weight velocity and weight z-score results. Specifically, the weight velocity (measured in g/day) was significantly better in the EHM group, yet there is no significant difference among groups in change in weight z-score from baseline to end of study. This was also present in the prior randomised controlled trial. Reference Blanco, Hair and Justice22 We hypothesise that the disconnect between weight velocity (the rate at which a child’s weight changes over time) and weight z-score (a measure of how a child’s weight compares to a reference population) arises because they measure different aspects of growth. Weight velocity measures the rate of weight gain or loss over a specified period. It provides a short-term view of how quickly a child’s weight is changing. The weight z-score measures a child’s weight relative to a reference population, standardised for age and sex. It provides a longer-term view, showing how a child’s weight compares to a population norm, which assume a normal distribution of weight for a given age and sex. Furthermore, this can be skewed because there are no reference norms for children with CHD.
There was a higher incidence of necrotising enterocolitis noted in both non-EHM groups (both the RCG and the TCG). These findings are consistent with prior studies that show infants fed using cow’s milk-based fortifiers/formulas tend to have higher incidence and severity of necrotising enterocolitis. Reference Sullivan, Schanler and Kim19,Reference Elgersma, Spatz and Fulkerson21,Reference Cristofalo, Schanler and Blanco24 This finding supports the aforementioned studies among extremely premature infants regarding the use of an EHM diet and the associated significant reduction in necrotising enterocolitis rates, including surgical necrotising enterocolitis. Reference Sullivan, Schanler and Kim19–Reference Fleig, Hagan and Lee23 It is likely that the combination of a higher enteral caloric concentration with an EHM diet translated into improved growth without increasing intestinal risk.
We noted several important centre-specific differences in the retrospective patients compared to the trial patients. Those patients had longer bypass and cross-clamp times than the trial data, but these times are consistent for this specific centre (data not shown). Retrospective patients also experienced shorter ICU and hospital length of stay related to centre-specific interstage discharge practices. 25,Reference Kumar, Gaynor and Jones26 Institutions that perform Norwood operations have variable practices regarding progression to discharge, and some centres maintain all single-ventricle patients in the hospital until after their second stage palliation, but our centre progresses patients to discharge unless remaining inpatient is clinically indicated, and our length of stay is comparatively short.
Our study has several important limitations. The retrospective nature of the review introduces the potential for selection bias of the retrospective patients; however, patients were selected in reverse order of presentation (from most recent backward) and all eligible patients were included. The non-contemporary nature of the controls could introduce biases in care based on the time frame they were managed and any particular institutional practices at that time. However, there were no new protocols initiated or significant changes in personnel during all time points, and some of the time frame was overlapping between the retrospective patients and the trial patients. In addition, the infants in the RCG were not on any sort of feeding protocol which would allow for provider variability in feeding practices, and therefore there could have been potential under-estimation in feeding intolerance as adjustments in their feeding regimen could have been made at any time based on provider preference rather than any objective criteria.
In conclusion, this study shows that neonates with single-ventricle physiology after stage I palliation have improved short-term growth and a decreased risk of necrotising enterocolitis or suspected necrotising enterocolitis when receiving an exclusive human milk diet with early fortification compared to standard of care. Additionally, even though the control group of the randomised controlled trial had a slower feeding advancement (due to standards of care and clinician concerns about tolerance when fortifying sooner when feeding non-EHM), the retrospective control group was not limited by a feeding protocol and grew similarly to the trial control group, but both groups exhibited inferior growth compared to the trial EHM group. A randomised controlled trial in which patients are fed either standard of care or an EHM diet utilising equivalent feeding protocols may be warranted.
Financial support
Funding was provided by Prolacta Bioscience. No individuals employed by the funders played any role in study design, decision to publish, or preparation of the manuscript. Research support was provided by Prolacta Bioscience to Megan Horsley, Amiee Trauth, and Lindsey Justice. Megan Horsley, David Cooper, and Lindsey Justice are consultants for Prolacta Bioscience. Cynthia Blanco received funding to conduct a prior randomised controlled trial from Prolacta Bioscience.









