INTRODUCTION
Q fever is an important zoonosis with a worldwide distribution that is caused by Coxiella burnetii, a strict intracellular Gram-negative bacterium [Reference Maurin and Raoult1]. The organism infects people and a wide range of wild and domesticated animals including sheep, cattle, goats, dogs, cats, pigeons and rabbits. Although most C. burnetii infections in animals are asymptomatic, they can sometimes result in late-stage abortion, stillbirths, or delivery of weak offspring. Infected animals shed C. burnetii into the environment in milk, colostrum, birth products and urine [Reference Rodolakis2] and thus play an important role in maintaining the organism in the environment [Reference Kelly, Coetzer, Thompson and Tustin3]. C. burnetii is extremely resistant to desiccation, low and high pH, and ultraviolet radiation so it can remain infective in soil for many months.
Humans are very susceptible to C. burnetii and infections can result from exposure to only a few organisms [Reference Beslagic4]. Most infections are asymptomatic (60%) but some cause acute signs including influenza-like illness and atypical pneumonia. In chronic cases there can be endocarditis, chronic hepatitis and osteomyelitis with mortalities of up to 11% [Reference Maurin and Raoult1, Reference Delgado5]. Infections are often an occupational hazard in people working with livestock and exposed to highly infectious aerosols from birth products, infectious dust particles or contaminated wool [Reference Marrie6]. These include workers in slaughterhouses, meat-packing plants and tanneries as well as veterinarians and farmers.
Q fever often occurs in outbreaks in people, for example in Spain [Reference Errasti7], Switzerland [Reference Dupuis8], Great Britain [Reference Guigno9] and Germany [Reference Schneider10]. A recent outbreak in The Netherlands affected over 3700 people and lead to the loss of over 50 000 dairy goats [Reference Schimmer11]. This has led to a renewed interest in Q fever worldwide which seems justified as the prevalence in domestic ruminants around the world has increased from 7·4% to 10% between 2007 and 2008, with the greatest increase in goats, from 9·7% to 15·7% [12].
Although Q fever occurs worldwide and has been classified as a notifiable animal disease by the World Organization for Animal Health (OIE) [13], details on the disease are scant in most countries. This might be due to under-reporting as in many countries the disease is not considered economically significant. In other cases it is because local reports are not in English and may therefore not be incorporated into international publications. As this is the case in China, we reviewed the local Chinese publications on Q fever and also international publications relating to the disease in China. In this report we provide a summary of the more recent data collected on Q fever in China.
OVERVIEW OF Q FEVER IN CHINA
Q fever was first reported in China in the 1950s when patients with atypical pneumonia were diagnosed serologically with the complement fixation test (CFT) [Reference Zhang14, Reference Zhai and Liu15]. In 1962, the first case of Q fever was definitively confirmed and the first Chinese strain of C. burnetii (Qi Yi) was isolated [Reference Yu16]. Moreover, in the 1960s, five outbreaks of Q fever in abattoir workers, stockyard men and troops from Inner Mongolia, Sichuan, Yunnan and Tibet were investigated using serology and bacterial isolation [Reference Yu, Williams and Thompson17]. Thereafter, there have been many studies and those on animals have indicated C. burnetii is widely distributed in China with several natural foci (Fig. 1). Between 1989 and 2013, there were 29 studies on people and animals (25 seroepidemiological studies, three seroepidemiological/molecular studies and one molecular study) which described C. burnetii infections in 64 cities/municipalities from 19 provinces across China (Tables 1 and 2). The principal animal species studied were cattle, goats, dogs, pigs and mice while horses, deer, marmots, sheep and rabbits were only surveyed once during that period. Nearly half of the studies were performed in the first 20 years while the remainder were conducted in the last 5 years, possibly because of the availability of more simple, sensitive and rapid diagnostic techniques. Initially the CFT was used for serology [Reference Cui18, Reference Yang, Chen and Dou19] but this was replaced by the immunofluorescence assay (IFA) test and most recently with the sensitive enzyme-linked immunosorbent assay (ELISA) which requires a single dilution of sera and can be automated [Reference Sun20, Reference Zhang21]. As substrate the ELISA uses immunodominant antigens of C. burnetii produced using proteomic techniques developed by Chinese scientists [Reference Chao22, Reference Xiong23]. These replaced whole-cell antigens previously used in testing which required sophisticated tissue culture facilities for C. burnetii.

Fig. 1. Sites in China where samples from people or animals were tested and the percentages found positive for anti-C. burnetii antibody. Dots represent sites where samples obtained from people and various animal species were found to be antibody positive against C. burnetii. Seropositive people were found in 14 provinces, mostly in eastern areas. Prevalences were higher in Inner Mongolia and Beijing than in Anhui and Xinjiang. Seropositive animals were found in 16 provinces, mostly those in the east, northeast and northwest of China. Seropositive cattle were more commonly found in the northeastern provinces, Inner Mongolia, Xinjiang, and Henan. Seropositive goats were mostly found in central Anhui province as well as in northeastern provinces and Inner Mongolia.
Table 1. Prevalences of people with evidence of Q fever in different provinces in China, 1989–2013
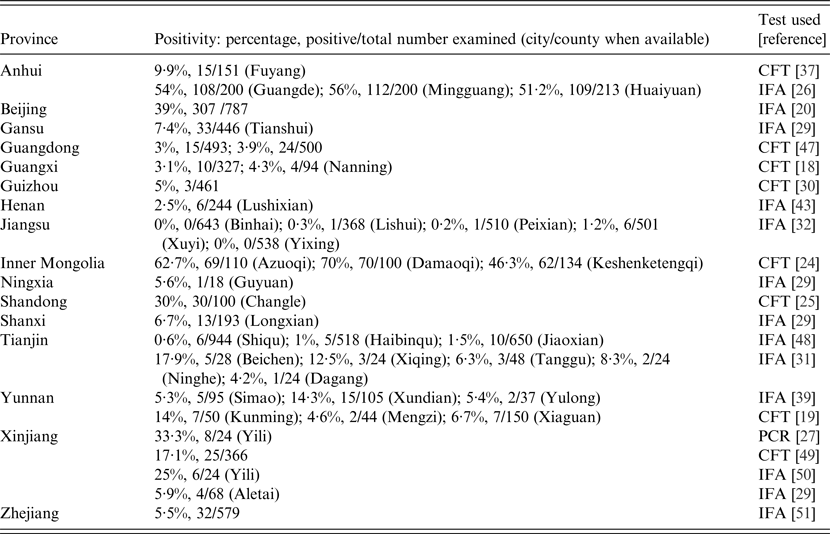
CFT, Complement fixation test; IFA, immunofluorescence assay; PCR, polymerase chain reaction.
Table 2. Prevalences of animals with evidence of Coxiella burnetii infections in different provinces in China, 1989–2013
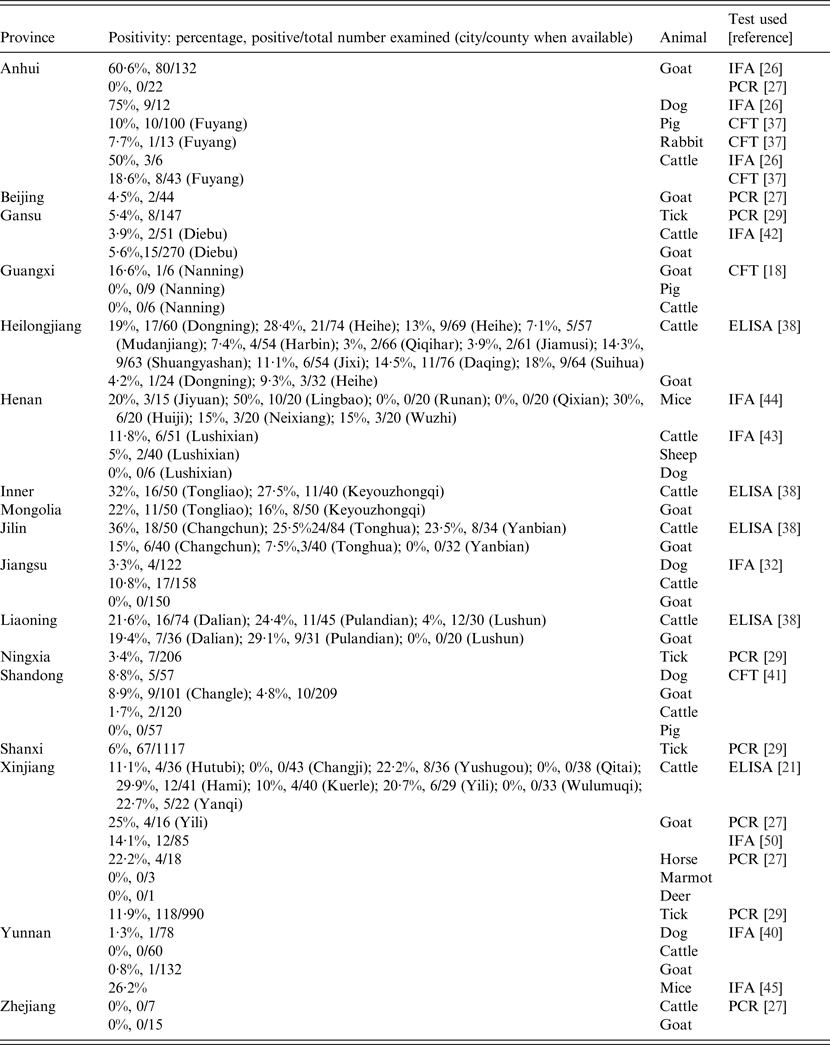
CFT, Complement fixation test; ELISA, enzyme-linked immunosorbent assay; IFA, immunofluorescence assay; PCR, polymerase chain reaction.
The overall prevalence of anti-Coxiella antibodies in the people studied to date in China is 10·2% (1139/11 209) (Table 3). The sensitivities of the CFT and IFA seem to be similar in people with a prevalence of 10·8% (343/3180) by CFT and 9·9% (796/8029) by IFA. The prevalence in cattle from all the studies in China is 15% (288/1918) while 12% (176/1440) of goats have been found positive. ELISA surveys have produced the highest seroprevalences in cattle (17·6%, 250/1423) and IFA the lowest (8·6%, 28/326). In goats the prevalences in IFA surveys and ELISA are similar, i.e. 14% (108/769) and 13·5% (48/355), respectively.
Table 3. Prevalences of anti-Coxiella burnetii antibodies in people, goats and cattle detected by different assays

CFT, Complement fixation test; ELISA, enzyme-linked immunosorbent assay; IFA, immunofluorescence assay.
* Denotes significant difference with other assays in the same row.
Q fever in people
Between 1989 and 2013 there were 19 surveys using serology (CFT or IFA) or polymerase chain reaction (PCR) on almost 10 000 human sera and blood samples from several provinces (Table 1). The majority of these studies were performed between 2010 and 2013. In eight of the 10 seroprevalence studies conducted before 2008, the prevalence rate was under 20%. A 1996 survey showed a 59% seroprevalence in 344 healthy people from three cities in Inner Mongolia (62·7%, in Azuoqi, 70% in Damaoqi, 46·3% in Keshenketengqi) [Reference Niu24] while a study performed a year later showed 30% of middle school students (n = 100) in Shandong were positive [Reference Sun25]. Surveys after 2010 that were conducted in Eastern China also showed generally higher seroprevalences, varying from 56% in Anhui province [Reference Zhang26] to 39% in Beijing [Reference Sun20] (Fig. 2). These increased seroprevalence rates could have been due to the use of the more sensitive IFA test replacing the CFT. In a molecular study in Yili region, Xinjiang province, C. burnetii DNA was detected in eight (33·3%) of 24 human blood samples using the loop-mediated isothermal amplification (LAMP) PCR assay [Reference Pan27].

Fig. 2. Prevalence of antibodies to C. burnetii in China between 1989 and 2013. Dots represent people (blue), cattle (red) and goats (green) that were tested for C. burnetii antibody. Of the 28 studies in the period, 16 were performed in the 20-year period of 1989–2008. Generally, seroprevalence appears to increase throughout the studies with higher detection rates observed during the last 4 years.
Overall, anti-Coxiella antibodies have been detected in people from 32 counties in 15 provinces in China (Table 1). Most infected people had no symptoms or mild febrile disease with influenza-like signs. Seroprevalence varied in different areas but was generally higher in the eastern provinces; Inner Mongolia (58·4%), Anhui (53·7%), Beijing (39%) and Shandong (30%). Lower prevalences were reported from Xinjiang (25%) in the West of China (Table 1, Fig. 1) while the other areas had prevalences of under 10% generally. Possible reasons for these variations in prevalence include geoclimatic differences, numbers of cases examined in different areas, lifestyles in different provinces, prevalence of ticks, socioeconomic status, distribution of domestic animals and their proximity to human dwellings, availability of the diagnostic tests and laboratories, and the sensitivity and specificity of the assay used. The variations seen in the 16S-23S ISR (intergenic spacer region) of seven C. burnetii isolates from China might reflect adaptions to different geographical circumstances [Reference Hu28].
Epidemiological data have shown that most seropositive people in China come from rural areas and have contact with animals [Reference Zhang26, Reference Zhang and Liu29], such as being livestock breeders, being exposed to animal abortion products and assisting with parturitions. In many studies seropositive people were not found to be significantly different with respect to age, sex or occupation [Reference Sun20, Reference Zhang26, Reference Yang, Shi and Guan30, Reference Zhang31]. However, in 2011 an IFA survey of local residents in Northwestern China revealed occupations such as cattle and goat breeders, meat processors and veterinarians were significantly associated with seropositivity [Reference Zhang and Liu29]. Similarly, other studies have shown females have significantly higher prevalences than males in Shandong (31·4% vs. 29·2%) [Reference Sun25], Anhui (58% vs. 49%) [Reference Zhang26] and Jiangsu (0·4% vs. 0·2%) provinces [Reference Tan, Li and Zhang32]. This is contrary to elsewhere in the world where studies have shown males are more likely to be infected [Reference Maurin and Raoult1]. High prevalences in females in China could be related to common activities of women in rural areas such as tending of livestock and tea picking which increases the chance of contact with ticks and exposure to infected soil and animals [Reference Zhang26].
The reported clinical features of Q fever have varied considerably according to geographical location, gender, age, strain, size of inoculum and route of infection. A retrospective study on 500 serum samples of community-acquired pneumonia cases from 12 provincial hospitals collected between November 2001 and June 2002 revealed 44 cases of Q fever pneumonia [Reference Shi-hu33]. On the other hand, hepatitis was considered as the major clinical presentation of acute Q fever cases admitted to E-Da hospital in Kaohsiung County in Southern Taiwan between 2004 and 2007 [Reference Lai34]. As elsewhere, Q fever in China should be suspected in febrile patients with hepatitis or atypical pneumonia who have a history of contact with domestic animals [Reference Bellazreg35].
Q fever is a common cause of fever of unknown origin in the world [Reference Maurin and Raoult1] and in a study of an epidemic of fever of unknown origin in Beijing [Reference Zhu36] anti-Coxiella IgG was detected by IFA in 67% of patients. Healthcare workers should be aware of the possibility of Q fever in febrile patients with non-specific signs, especially those with a history of association with domestic livestock.
It is of note that Q fever is not a notifiable disease in China and thus test facilities are not widely available. Most cases are diagnosed in retrospective and epidemiological studies which means acute cases are often misdiagnosed leading to the greater possibility of chronic infections which have a poor prognosis and high mortality.
C. burnetii infections in cattle
There have been 11 seroprevalence studies on cattle since 1989 with four performed prior to 1996 revealing low seroprevalences of under 20% [Reference Yang, Shi and Guan30, Reference Zhu36, Reference Wang37]. In 2010, the rate was considerably higher, being 50% in cattle from Anhui province [Reference Zhang26] although the sample was very small (n = 6). Increasing seroprevalences were also found in five studies conducted between 2011 and 2013, the highest being 36% in Changchun city, Jilin province [Reference Guo38] (Fig. 2). The highest seroprevalences nationally were found in animals from northern and central provinces. No seropositive cattle were found in studies in Guangxi [Reference Cui18] and Yunnan provinces [Reference Chang39] which are in Southern China (Table 2, Fig. 1). Seropositive animals were associated with abortions and were generally older [Reference Zhang21, Reference Zhang and Liu29, Reference Hong-Bo40]. In the only molecular study to date on cattle in China, C. burnetii DNA was not detected in any of the seven cattle tested from Zhejiang province [Reference Pan27].
C. burnetii infections in goats
Goats are important sources of C. burnetii infection in people and ten serosurveys were conducted on this species in China between 1989 and 2013. The seroprevalences reported varied from 60·61% to 29·1% in Anhui and Liaoning provinces, to 4·8% and 5·6% in Shandong and Gansu provinces, respectively [Reference Zhang26, Reference Guo38, Reference Li, Chen and Huang41, Reference Yang42] (Table 2, Fig. 2). Higher goat seropositivity was reported in animals from the northern and southern provinces of China rather than from the central provinces (Table 2, Fig. 1). The variation in prevalence could be due to small sample sizes or could reflect climatic or agricultural differences between provinces in different areas. Moreover, it may be because of differences between sensitivities of the tests used in the different provinces with the CFT being more insensitive than the more recently introduced ELISA and IFA [Reference Kelly, Coetzer, Thompson and Tustin3]. There is only a single recent molecular study which was conducted on blood from goats from four provinces (Xinjiang, Anhui, Zhejiang, Beijing) using the LAMP PCR assay. Twenty-five percent (4/16) of goats from Xinjiang were positive, as were 5% from Beijing (2/44) [Reference Pan27].
C. burnetii infections in dogs
A total of five seroprevalence studies were performed on dogs between 1996 and 2012 using CFT and IFA (Table 2). Although four of these were performed in closely neighbouring provinces in Eastern China (Anhui, Henan, Shandong, Jiangsu), the prevalences were very different. The highest prevalences were in Anhui (75%), followed by Shandong (9%) [Reference Zhang26, Reference Li, Chen and Huang41], with the lowest in Jiangsu (3%) [Reference Tan, Li and Zhang32], Yunnan (1·28%) [Reference Chang39] and Henan (0/6) [Reference Zhang and LV43] provinces.
C. burnetii infections in pigs
Serology using CFT on pigs from Guangxi, Anhui and Shandong provinces [Reference Cui18, Reference Wang37, Reference Li, Chen and Huang41] revealed that only animals from Anhui were positive (10%, 10/100). Therefore pigs might not play an important role in Q fever in China.
C. burnetii infections in mice
Serosurveillance of mice using IFA has shown positive animals in Henan (19%) in the east of China [Reference Wu44] and in Yuxi city (26%) in Yunnan province in the south of China [Reference Ya45]. Moreover, high copy numbers of C. burnetii DNA were detected using real-time PCR in 32%, 28% and 60% of blood, liver and spleen samples, respectively, of mice in the latter study. The high copy numbers and prevalence of infections suggest mice could play a role in the epidemiology of Q fever in China.
C. burnetii infections in ticks
Ticks are commonly infected with C. burnetii but they are probably not important in the maintenance of infections in livestock and people [Reference Kelly, Coetzer, Thompson and Tustin3]. It is possible, however, that organisms present in tick faeces might contaminate hides and wool and be a source of infection for people and animals. In China, one study [Reference Zhang and Liu29] showed C. burnetii DNA in eight tick species (Dermacentor silvarum, D. niveus, D. nuttalli, Hyalomma detritum, Hy. scupense, Haemaphysalis japonica, H. concinna, H. qinghaiensis) from four provinces (Gansu, Ningxia, Shanxi, Xinjiang) in Northwestern China. Overall, 10% (239/2460) of the ticks studied were positive with significantly higher infection levels in Hy. detritum (84/428), H. concinna (48/356) and D. niveus (46/967) (P < 0·01). Ticks from Xinjiang province were most commonly positive (11·9%), in particular Hy. detritum and Hy. scupenses. A later study [Reference Guo38] showed DNA of C. burnetii in D. silvarum, Ixodes persulcatus, H. conicinna, H. japonica, Boophilus microplus and D. nuttalli from three northeastern provinces (Jilin, Liaoning, Heilongjiang) and Inner Mongolia. Overall 2% of the 475 pooled tick samples (n = 10 for each pool) were positive. Although ticks are infected, as in the rest of the world it has been reported there is no good evidence that Q fever is regularly transmitted to humans by tickbites in China [Reference Wu46].
C. burnetii infections in other animals
In 2013, the only study involving sheep in China revealed 5% of animals (2/40) in Lushixian, Henan province in the east of China, were seropositive by IFA [Reference Yang42]. Although a deer and three marmots from Xinjiang province were negative for C. burnetii in a LAMP PCR assay, DNA of the organism has been detected in 22% (4/18) of horses in the only study on this species in China [Reference Pan27]. Further studies are needed to determine the role these other species might have in the epidemiology of Q fever in China.
CONCLUSION
The overall prevalence of Q fever infections in 29 studies to date since 1989 in China has been 10% (1139/11 209) in people, 15% (288/1918) in cattle and 12% (176/1440) in goats. The seroprevalences reported in cattle and goats are higher than those in other domestic animal species indicating they are major sources of human infection in China, as is the case elsewhere in the world [Reference Kelly, Coetzer, Thompson and Tustin3]. Although human and animal infections are known to occur and be relatively common in China, Q fever is not a reportable disease in the country and clinical cases are probably largely unrecognizable on account of insufficient and inadequate disease surveillance. Of note is the lack of clinical case reports in the country. There is a need for information on the epidemiology of C. burnetii in China as well as many other issues such as distribution, pathogenesis and molecular typing. The data from the studies to date in China provide only a basic picture of Q fever in the country. Active surveillance and further research studies are recommended, particularly with modern and widely applicable molecular methods, to more clearly define the epidemiology and importance of C. burnetii infections in animals and people in China. This will enable the formulation and implementation of locally applicable control methods for Q fever which can be implemented by animal and human healthcare workers.
ACKNOWLEDGEMENTS
This project was supported by grants from the National Natural Science Foundation of China (no. 31272575), by the Jiangsu Co-innovation Center for the Prevention and Control of Important Animal Infectious Diseases and Zoonoses, Yangzhou, Jiangsu, P.R. China, and by the Priority Academic Programme Development of Jiangsu Higher Education Institutions.
DECLARATION OF INTEREST
None.








